[1] CARMELIET P, JAIN RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473(7347):298-307.
[2] GAHARWAR AK, SINGH I, KHADEMHOSSEINI A. Engineered biomaterials for in situ tissue regeneration. Nat Rev Mater. 2020;5(9):686-705.
[3] CAPORARELLO N, D’ANGELI F, CAMBRIA MT, et al. Pericytes in microvessels: From “Mural” function to brain and retina regeneration. Int J Mol Sci. 2019; 20(24):6351.
[4] WON JE, LEE YS, PARK JH, et al. Hierarchical microchanneled scaffolds modulate multiple tissue-regenerative processes of immune-responses, angiogenesis, and stem cell homing. Biomaterials. 2020;227:119548.
[5] JALALI S, TAFAZZOLI-SHADPOUR M, HAGHIGHIPOUR N, et al. Regulation of endothelial cell adherence and elastic modulus by substrate stiffness. Cell Commun Adhes. 2015;22(2-6):79-89.
[6] KRETSCHMER M, RüDIGER D, ZAHLER S. Mechanical aspects of angiogenesis. Cancers (Basel). 2021;13(19):4987.
[7] JANNATBABAEI A, TAFAZZOLI-SHADPOUR M, SEYEDJAFARI E. Effects of substrate mechanics on angiogenic capacity and nitric oxide release in human endothelial cells. Ann N Y Acad Sci. 2020;1470(1):31-43.
[8] WANG Y, ZHANG X, WANG W, et al. Integrin αVβ5/Akt/Sp1 pathway participates in matrix stiffness-mediated effects on VEGFR2 upregulation in vascular endothelial cells. Am J Cancer Res. 2020;10(8):2635-2648.
[9] HUSAIN A, KHADKA A, EHRLICHER A, et al. Substrate stiffening promotes VEGF-A functions via the PI3K/Akt/mTOR pathway. Biochem Biophys Res Commun. 2022;586:27-33.
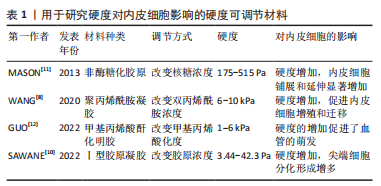
[10] SAWANE M, OGURA Y, NAKAMURA A, et al. Blood vessels sense dermal stiffness via a novel mechanotransducer, APJ. Angiogenesis. 2022;25(2):151-154.
[11] MASON BN, STARCHENKO A, WILLIAMS RM, et al. Tuning three-dimensional collagen matrix stiffness independently of collagen concentration modulates endothelial cell behavior. Acta Biomater. 2013;9(1):4635-4644.
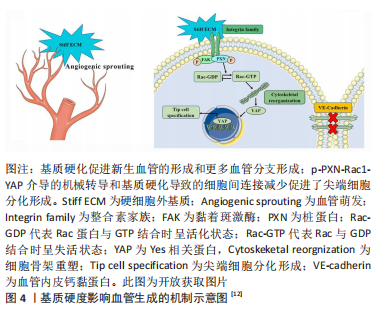
[12] GUO Y, MEI F, HUANG Y, et al. Matrix stiffness modulates tip cell formation through the p-PXN-Rac1-YAP signaling axis. Bioact Mater. 2022;7:364-376.
[13] BETTINGER CJ, LANGER R, BORENSTEIN JT. Engineering substrate topography at the micro- and nanoscale to control cell function. Angew Chem Int Ed Engl. 2009;48(30):5406-5415.
[14] MA C, KUZMA ML, BAI X, et al. Biomaterial-based metabolic regulation in regenerative engineering. Adv Sci. 2019;6(19):1900819.
[15] CHUNG TW, LIU DZ, WANG SY, et al. Enhancement of the growth of human endothelial cells by surface roughness at nanometer scale. Biomaterials. 2003;24(25):4655-4661.
[16] LIU M, WANG D, GU S, et al. Micro/nano materials regulate cell morphology and intercellular communication by extracellular vesicles. Acta Biomater. 2021;124:130-138.
[17] MASCHARAK S, BENITEZ PL, PROCTOR AC, et al. YAP-dependent mechanotransduction is required for proliferation and migration on native-like substrate topography. Biomaterials. 2017;115:155-166.
[18] TANG Z, WANG X, YANG J, et al. Microconvex dot-featured silk fibroin films for promoting human umbilical vein endothelial cell angiogenesis via enhancing the expression of bFGF and VEGF. ACS Biomater Sci Eng. 2021; 7(6):2420-2429.
[19] YANG Y, LIN Y, ZHANG Z, et al. Micro/nano-net guides M2-pattern macrophage cytoskeleton distribution via Src-ROCK signalling for enhanced angiogenesis. Biomater Sci. 2021;9(9):3334-3347.
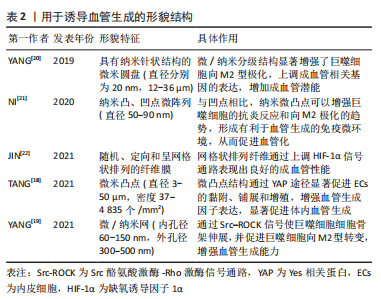
[20] YANG C, ZHAO C, WANG X, et al. Stimulation of osteogenesis and angiogenesis by micro/nano hierarchical hydroxyapatite via macrophage immunomodulation. Nanoscale. 2019;11(38):17699-17708.
[21] NI S, ZHAI D, HUAN Z, et al. Nanosized concave pit/convex dot microarray for immunomodulatory osteogenesis and angiogenesis. Nanoscale. 2020; 12(31):16474-16488.
[22] JIN S, YANG R, CHU C, et al. Topological structure of electrospun membrane regulates immune response, angiogenesis and bone regeneration. Acta Biomater. 2021;129:148-158.
[23] TIAN T, ZHANG T, LIN Y, et al. Vascularization in craniofacial bone tissue engineering. J Dent Res. 2018;97(9):969-976.
[24] GRIFFON DJ, SEDIGHI MR, SCHAEFFER DV, et al. Chitosan scaffolds: interconnective pore size and cartilage engineering. Acta Biomater. 2006; 2(3):313-320.
[25] CHOI SW, ZHANG Y, MACEWAN MR, et al. Neovascularization in biodegradable inverse opal scaffolds with uniform and precisely controlled pore sizes. Adv Healthc Mater. 2013;2(1):145-154.
[26] BAI F, WANG Z, LU J, et al. The correlation between the internal structure and vascularization of controllable porous bioceramic materials in vivo: a quantitative study. Tissue Eng Part A. 2010;16(12):3791-3803.
[27] YIN Y, HE XT, WANG J, et al. Pore size-mediated macrophage M1-to-M2 transition influences new vessel formation within the compartment of a scaffold. Appl Mater Today. 2020;18:100466.
[28] WANG WY, KENT RN 3RD, HUANG SA, et al. Direct comparison of angiogenesis in natural and synthetic biomaterials reveals that matrix porosity regulates endothelial cell invasion speed and sprout diameter. Acta Biomater. 2021;135:260-273.
[29] JIA G, HUANG H, NIU J, et al. Exploring the interconnectivity of biomimetic hierarchical porous Mg scaffolds for bone tissue engineering: Effects of pore size distribution on mechanical properties, degradation behavior and cell migration ability. J Magnes Alloy. 2021;9(6):1954-1966.
[30] GUPTE MJ, SWANSON WB, HU J, et al. Pore size directs bone marrow stromal cell fate and tissue regeneration in nanofibrous macroporous scaffolds by mediating vascularization. Acta Biomater. 2018;82:1-11.
[31] ALI D, SEN S. Finite element analysis of mechanical behavior, permeability and fluid induced wall shear stress of high porosity scaffolds with gyroid and lattice-based architectures. J Mech Behav Biomed Mater. 2017;75:262-270.
[32] MEHDIZADEH H, BAYRAK ES, LU C, et al. Agent-based modeling of porous scaffold degradation and vascularization: optimal scaffold design based on architecture and degradation dynamics. Acta Biomater. 2015;27:167-178.
[33] WANG S, HASHEMI S, STRATTON S, et al. The effect of physical cues of biomaterial scaffolds on stem cell behavior. Adv Healthc Mater. 2021;10(3): e2001244.
[34] LIU J, LONG H, ZEUSCHNER D, et al. Synthetic extracellular matrices with tailored adhesiveness and degradability support lumen formation during angiogenic sprouting. Nat Commun. 2021;12(1):3402.
[35] TRAPPMANN B, BAKER BM, POLACHECK WJ, et al. Matrix degradability controls multicellularity of 3D cell migration. Nat Commun. 2017;8(1):371.
[36] 贺利贞.功能化介孔二氧化硅纳米载药体系在肿瘤诊疗中的应用与机制研究[D].广州:暨南大学,2020.
[37] GUERRERO PA, MCCARTY JH. Integrins in vascular development and pathology. Adv Pharmacol. 2018;81:129-153.
[38] MELCHIORRI AJ, HIBINO N, YI T, et al. Contrasting biofunctionalization strategies for the enhanced endothelialization of biodegradable vascular grafts. Biomacromolecules. 2015;16(2):437-446.
[39] MOULISOVá V, GONZALEZ-GARCíA C, CANTINI M, et al. Engineered microenvironments for synergistic VEGF-Integrin signalling during vascularization. Biomaterials. 2017;126:61-74.
[40] PARK TY, MAENG SW, JEON EY, et al. Adhesive protein-based angiogenesis-mimicking spatiotemporal sequential release of angiogenic factors for functional regenerative medicine. Biomaterials. 2021;272:120774.
[41] BAI Y, BAI L, ZHOU J, et al. Sequential delivery of VEGF, FGF-2 and PDGF from the polymeric system enhance HUVECs angiogenesis in vitro and CAM angiogenesis. Cell Immunol. 2018;323:19-32.
[42] TEIXEIRA SP, DOMINGUES RM, SHEVCHUK M, et al. Biomaterials for sequestration of growth factors and modulation of cell behavior. Adv Funct Mater. 2020;30(44):1909011
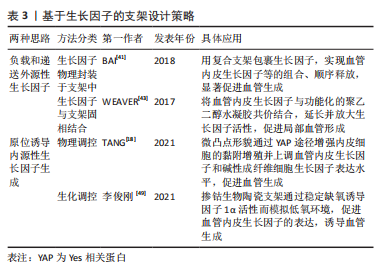
[43] WEAVER JD, HEADEN DM, AQUART J, et al. Vasculogenic hydrogel enhances islet survival, engraftment, and function in leading extrahepatic sites. Sci Adv. 2017;3(6):e1700184.
[44] MARTINO MM, BRIQUEZ PS, Güç E, et al. Growth factors engineered for super-affinity to the extracellular matrix enhance tissue healing. Science. 2014;343(6173):885-888.
[45] QU M, JIANG X, ZHOU X, et al. Stimuli-responsive delivery of growth factors for tissue engineering. Adv Healthc Mater. 2020;9(7):e1901714.
[46] JOSHI RV, NELSON CE, POOLE KM, et al. Dual pH- and temperature-responsive microparticles for protein delivery to ischemic tissues. Acta Biomater. 2013;9(5):6526-6534.
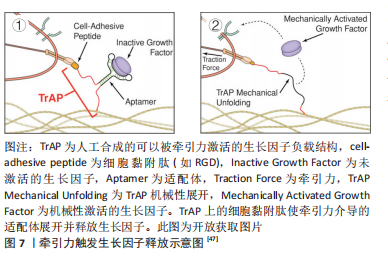
[47] STEJSKALOVá A, OLIVA N, ENGLAND FJ, et al. Biologically inspired, cell-selective release of aptamer-trapped growth factors by traction forces. Adv Mater. 2019;31(7):e1806380.
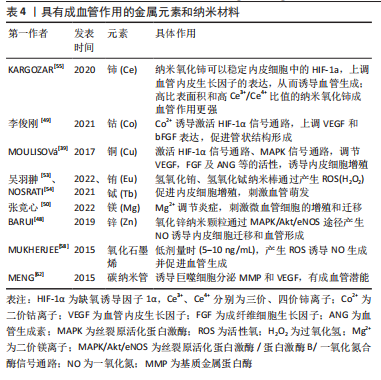
[48] BARUI AK, NETHI SK, HAQUE S, et al. Recent development of metal nanoparticles for angiogenesis study and their therapeutic applications. ACS Appl Bio Mater. 2019;2(12):5492-5511.
[49] 李俊刚.3D打印掺钴生物陶瓷支架介导成骨-成血管偶联促进骨修复的作用研究[D].福州:福建医科大学,2021.
[50] 张竞心,刘林枫,张士文,等.镁离子促进骨再生的分子机制[J].中国组织工程研究,2022,26(33):5384-5392.
[51] AUGUSTINE R, DALVI YB, YADU NATH VK, et al. Yttrium oxide nanoparticle loaded scaffolds with enhanced cell adhesion and vascularization for tissue engineering applications. Mater Sci Eng C Mater Biol Appl. 2019; 103:109801.
[52] AUGUSTINE R, HASAN A, PATAN NK, et al. Titanium nanorods loaded pcl meshes with enhanced blood vessel formation and cell migration for wound dressing applications. Macromol Biosci. 2019;19(7):e1900058.
[53] 吴羽翀,彭旭,余喜讯.掺铕聚磷酸钙骨组织工程支架可促成骨,血管生成及抗无菌性松动[J].中国组织工程研究,2022,26(28):4458-4465.
[55] NOSRATI H, ARAMIDEH KHOUY R, NOSRATI A, et al. Nanocomposite scaffolds for accelerating chronic wound healing by enhancing angiogenesis. J Nanobiotechnol. 2021;19(1):1-21.
[55] KARGOZAR S, BAINO F, HAMZEHLOU S, et al. Nanotechnology for angiogenesis: opportunities and challenges. Chem Soc Rev. 2020;49(14): 5008-5057.
[56] LAU P, BIDIN N, ISLAM S, et al. Influence of gold nanoparticles on wound healing treatment in rat model: Photobiomodulation therapy. Lasers Surg Med. 2017;49(4):380-386.
[57] CHEN Y, WU Y, GAO J, et al. Transdermal vascular endothelial growth factor delivery with surface engineered gold nanoparticles. ACS Appl Mater Interfaces. 2017;9(6):5173-5180.
[58] MUKHERJEE S, SRIRAM P, BARUI AK, et al. Graphene oxides show angiogenic properties. Adv Healthc Mater. 2015;4(11):1722-1732.
[59] QIAN Y, SONG J, ZHAO X, et al. 3D Fabrication with integration molding of a graphene oxide/polycaprolactone nanoscaffold for neurite regeneration and angiogenesis. Adv Sci (Weinh). 2018;5(4):1700499.
[60] HUANG S, LIU H, LIAO K, et al. Functionalized GO nanovehicles with nitric oxide release and photothermal activity-based hydrogels for bacteria-infected wound healing. ACS Appl Mater Interfaces. 2020;12(26): 28952-28964.
[61] MASOTTI A, MILLER MR, CELLUZZI A, et al. Regulation of angiogenesis through the efficient delivery of microRNAs into endothelial cells using polyamine-coated carbon nanotubes. Nanomed. 2016;12(6):1511-1522.
[62] MENG J, LI X, WANG C, et al. Carbon nanotubes activate macrophages into a M1/M2 mixed status: recruiting naïve macrophages and supporting angiogenesis. ACS Appl Mater Interfaces. 2015;7(5):3180-3188. |
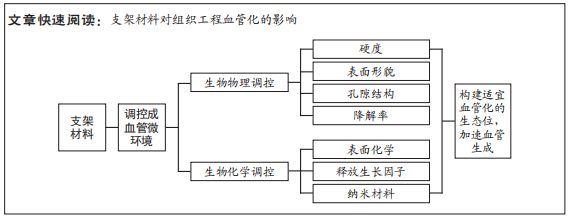

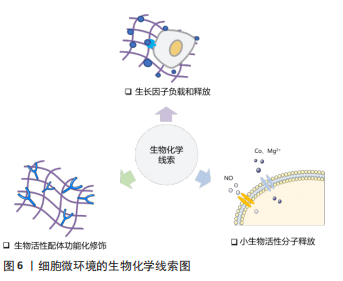
 促进血管生成对于组织修复疗法的成功和组织工程构建体的命运至关重要。虽然许多生化信号分子已经被使用,但由于半衰期短和活性差等问题,它们在体内的生物学功能非常有
促进血管生成对于组织修复疗法的成功和组织工程构建体的命运至关重要。虽然许多生化信号分子已经被使用,但由于半衰期短和活性差等问题,它们在体内的生物学功能非常有