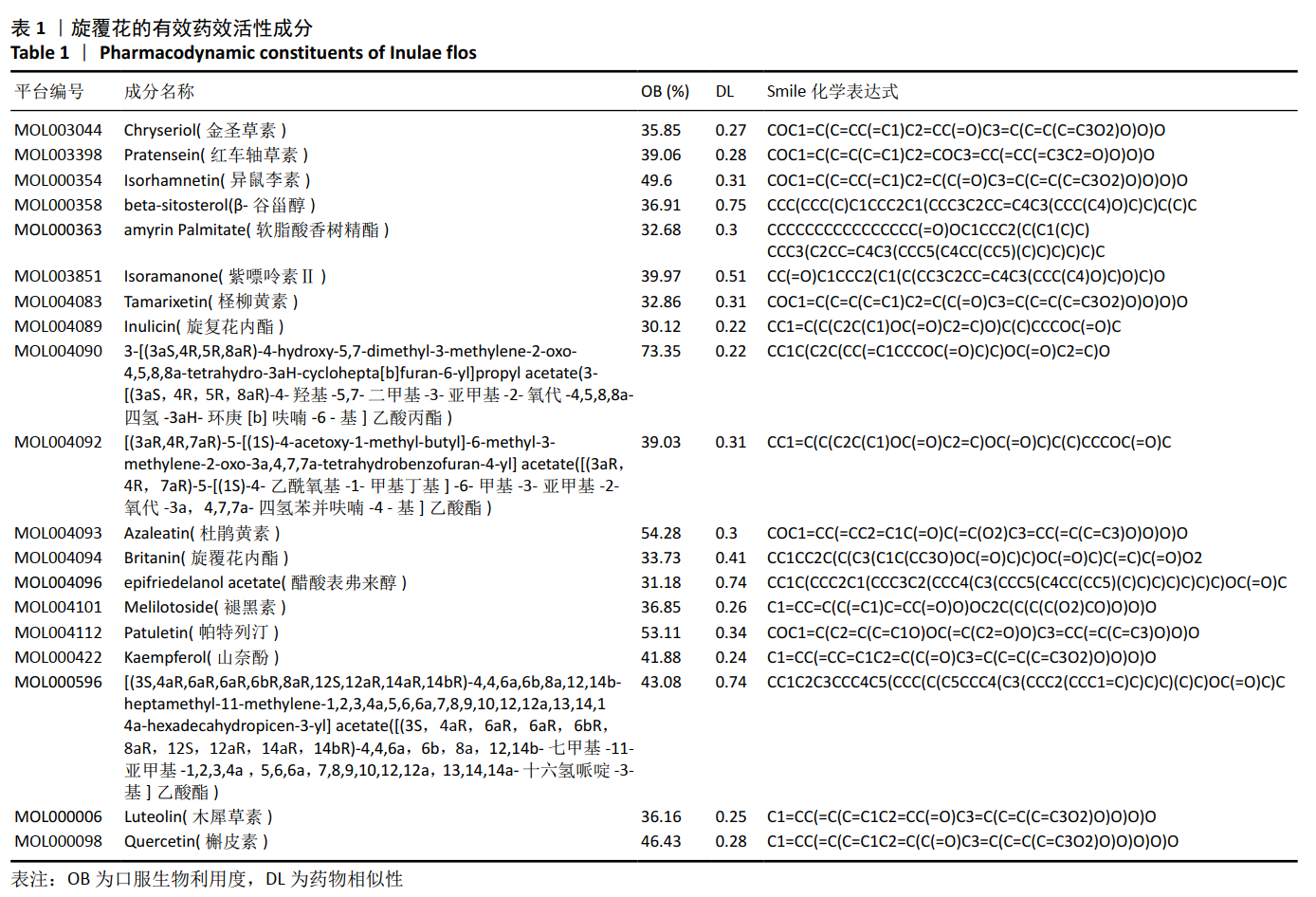
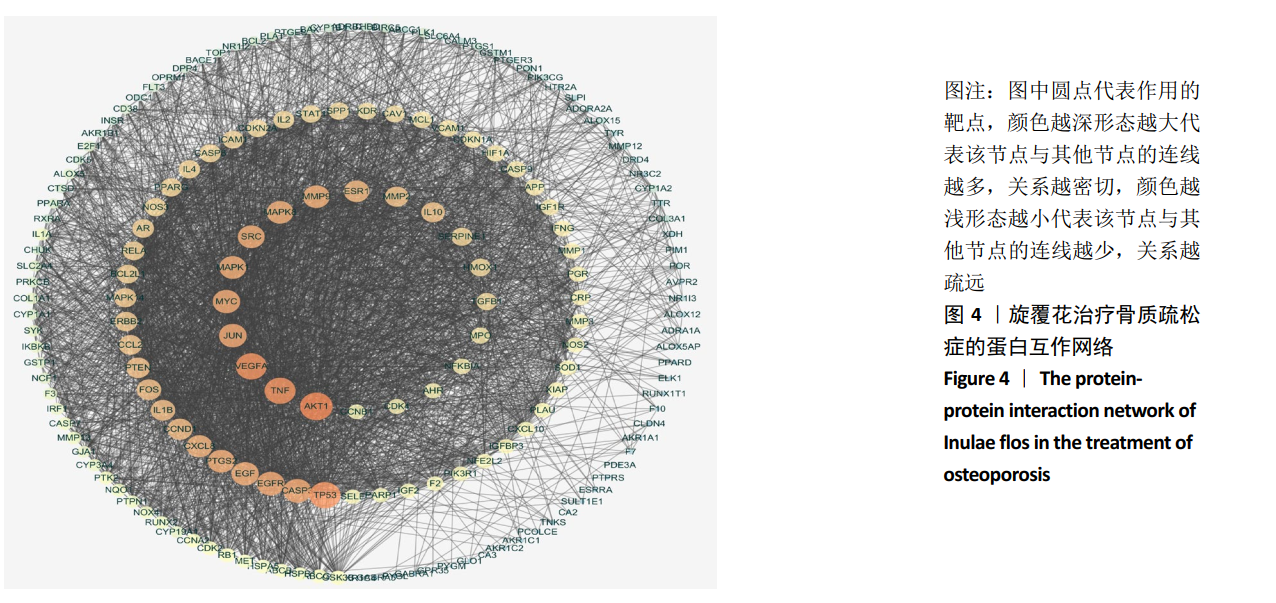
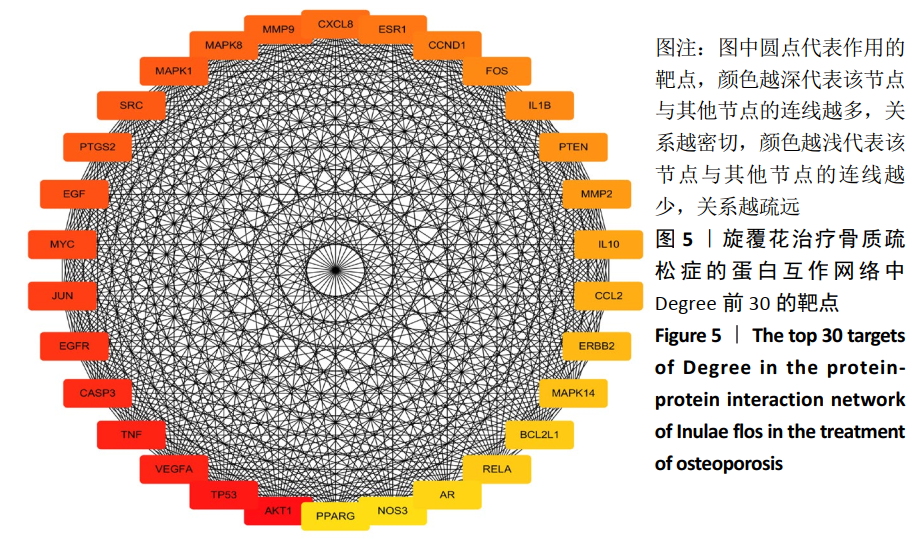
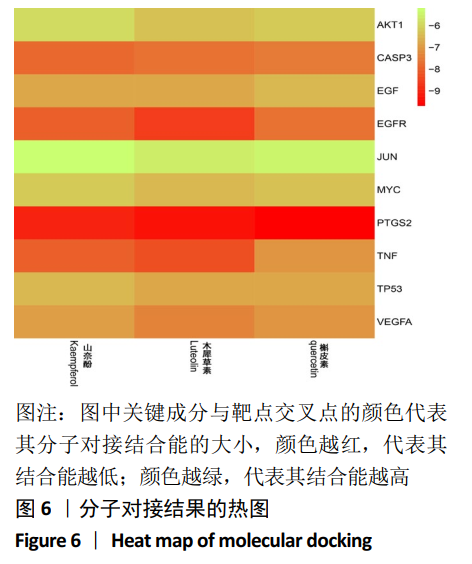
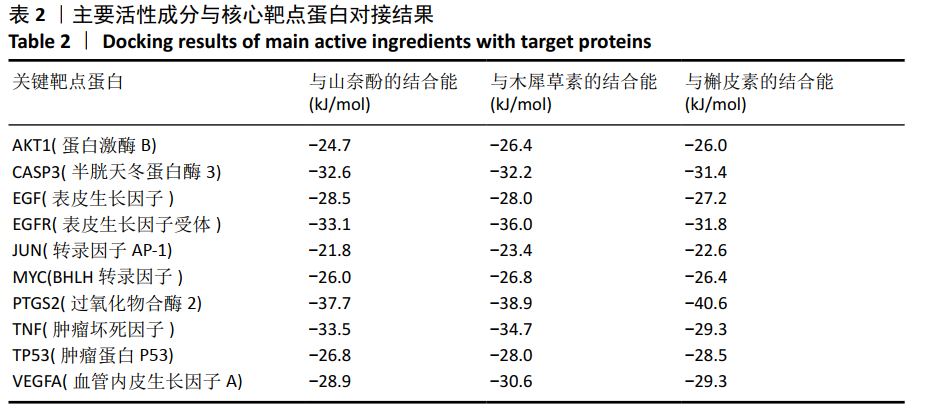
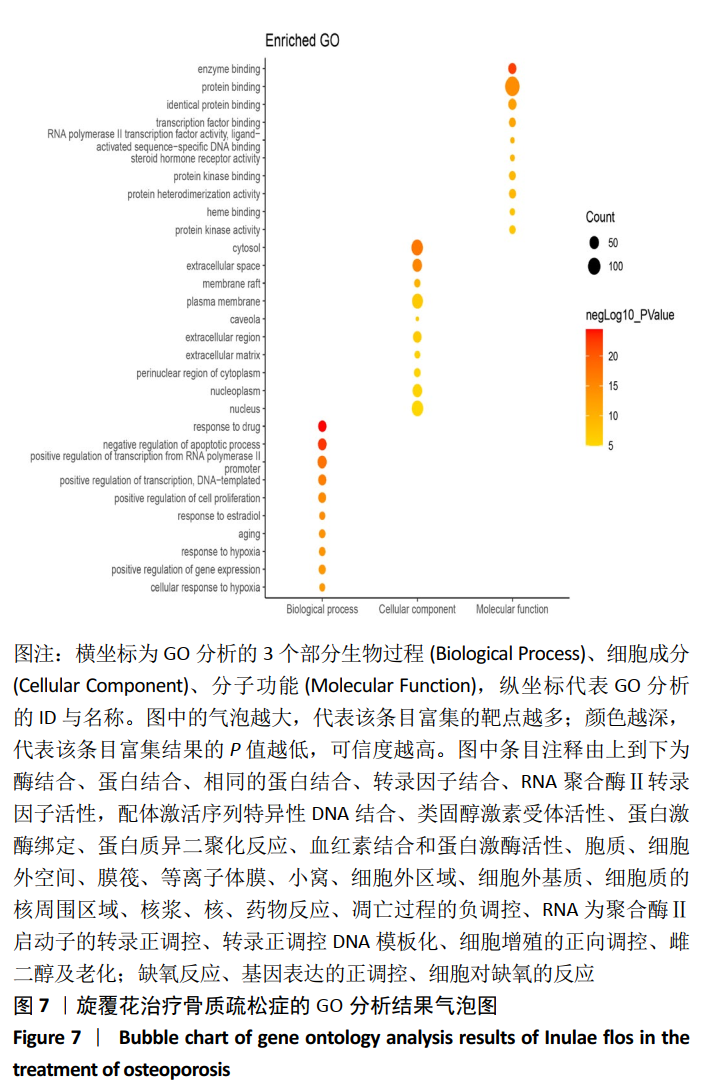
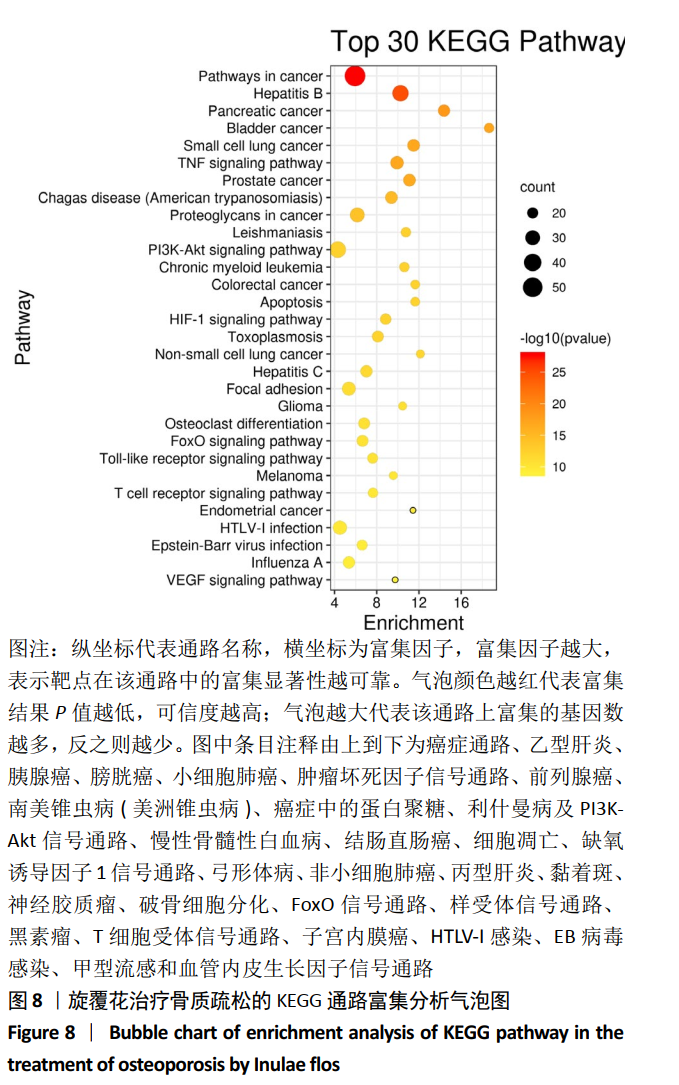
[1] COSMAN F, DE BEUR SJ, LEBOFF MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10): 2359-2381.
[2] ASPRAY TJ, HILL TR. Osteoporosis and the ageing skeleton. Subcell Biochem. 2019;91:453-476.
[3] 中华医学会骨质疏松和骨矿盐疾病分会.中国骨质疏松症流行病学调查及“健康骨骼”专项行动结果发布[J].中华骨质疏松和骨矿盐疾病杂志,2019,12(4):317-318.
[4] BECK BR, DALY RM, SINGH MA, et al. Exercise and Sports Science Australia (ESSA) position statement on exercise prescription for the prevention and management of osteoporosis. J Sci Med Sport. 2017;20(5):438-445.
[5] VANDENBROUCKE A, LUYTEN FP, FLAMAING J, et al. Pharmacological treatment of osteoporosis in the oldest old. Clin Interv Aging. 2017;12: 1065-1077.
[6] MIKAMI Y, MATSUMOTO T, KANO K, et al. Current status of drug therapies for osteoporosis and the search for stem cells adapted for bone regenerative medicine. Anat Sci Int. 2014;89(1):1-10.
[7] XIAO Y, LI K, WANG Z, et al. Pectolinarigenin prevents bone loss in ovariectomized mice and inhibits RANKL-induced osteoclastogenesis via blocking activation of MAPK and NFATc1 signaling. J Cell Physiol. 2019;234(8):13959-13968.
[8] ZHAN Y, LIANG J, TIAN K, et al. Vindoline inhibits RANKL-induced osteoclastogenesis and prevents ovariectomy-induced bone loss in mice. Front Pharmacol. 2020;10:1587.
[9] JIN HZ, LEE D, LEE JH, et al. New sesquiterpene dimers from Inula britannica inhibit NF-kappaB activation and NO and TNF-alpha production in LPS-stimulated RAW264.7 cells. Planta Med. 2006;72(1):40-45.
[10] DONG M, HONG T, LIU S, et al. Hepatoprotective effect of the flavonoid fraction isolated from the flower of Inula britannica against D-Galactosamine-induced hepatic injury. Mol Med Rep. 2013;7(6):1919-1923.
[11] HAN M, WEN JK, ZHENG B, et al. Acetylbritannilatone suppresses NO and PGE2 synthesis in RAW 264.7 macrophages through the inhibition of iNOS and COX-2 gene expression. Life Sci. 2004;75(6):675-684.
[12] 李祖浩,王辰宇,王中汉,等.骨质疏松性骨缺损的治疗进展:支架植入与局部药物递送[J].中国组织工程研究,2018,22(18):2939-2945.
[13] RU J, LI P, WANG J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform. 2014;6:13.
[14] KIM S, SHOEMAKER BA, BOLTON EE, et al. Finding Potential Multitarget Ligands Using PubChem. Methods Mol Biol. 2018;1825:63-91.
[15] STELZER G, ROSEN N, PLASCHKES I, et al. The GeneCards suite: from gene data mining to disease genome sequence analyses. Curr Protoc Bioinformatics. 2016;54:1.30.1-1.30.33.
[16] WISHART DS, FEUNANG YD, GUO AC, et al. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2018;46(D1):D1074-D1082.
[17] SHANNON P, MARKIEL A, OZIER O, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003;13(11):2498-2504.
[18] SZKLARCZYK D, MORRIS JH, COOK H, et al. The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 2017;45(D1):D362-D368.
[19] TROTT O, OLSON AJ. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem. 2010;31(2):455-461.
[20] HUANG DA W, SHERMAN BT, LEMPICKI RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4(1):44-57.
[21] KHOSLA S, HOFBAUER LC. Osteoporosis treatment: recent developments and ongoing challenges. Lancet Diabetes Endocrinol. 2017; 5(11):898-907.
[22] LEDER BZ. Parathyroid hormone and parathyroid hormone-related protein analogs in osteoporosis therapy. Curr Osteoporos Rep. 2017;15(2): 110-119.
[23] LEVIN VA, JIANG X, KAGAN R. Estrogen therapy for osteoporosis in the modern era. Osteoporos Int. 2018;29(5):1049-1055.
[24] 国家药典委员会.中华人民共和国药典.一部[S].北京:中国医药科技出版社,2020:339.
[25] YUAN Z, MIN J, ZHAO Y, et al. Quercetin rescued TNF-alpha-induced impairments in bone marrow-derived mesenchymal stem cell osteogenesis and improved osteoporosis in rats. Am J Transl Res. 2018;10(12):4313-4321.
[26] DERAKHSHANIAN H, DJALALI M, DJAZAYERY A, et al. Quercetin prevents experimental glucocorticoid-induced osteoporosis: a comparative study with alendronate. Can J Physiol Pharmacol. 2013;91(5):380-385.
[27] FARR JN, XU M, WEIVODA MM, et al. Targeting cellular senescence prevents age-related bone loss in mice. Nat Med. 2017;23(9):1072-1079.
[28] SUH KS, CHON S, CHOI EM. Luteolin alleviates methylglyoxal-induced cytotoxicity in osteoblastic MC3T3-E1 cells. Cytotechnology. 2016;68(6):2539-2552.
[29] KIM TH, JUNG JW, HA BG, et al. The effects of luteolin on osteoclast differentiation, function in vitro and ovariectomy-induced bone loss. J Nutr Biochem. 2011;22(1):8-15.
[30] JING Z, WANG C, YANG Q, et al. Luteolin attenuates glucocorticoid-induced osteoporosis by regulating ERK/Lrp-5/GSK-3β signaling pathway in vivo and in vitro. J Cell Physiol. 2019; 234(4):4472-4490.
[31] NOWAK B, MATUSZEWSKA A, NIKODEM A, et al. Oral administration of kaempferol inhibits bone loss in rat model of ovariectomy-induced osteopenia. Pharmacol Rep. 2017;69(5):1113-1119.
[32] KIM CJ, SHIN SH, KIM BJ, et al. The effects of kaempferol-inhibited autophagy on osteoclast formation. Int J Mol Sci. 2018;19(1):125.
[33] SHARMA AR, NAM JS. Kaempferol stimulates WNT/β-catenin signaling pathway to induce differentiation of osteoblasts. J Nutr Biochem. 2019;74:108228.
[34] WONG SK, CHIN KY, IMA-NIRWANA S. The osteoprotective effects of kaempferol: the evidence from in vivo and in vitro studies. Drug Des Devel Ther. 2019;13:3497-3514.
[35] KOMORI T. Cell death in chondrocytes, osteoblasts, and osteocytes. Int J Mol Sci. 2016; 17(12):2045.
[36] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature. 2003; 423(6937):337-342.
[37] RAISZ LG. Pathogenesis of osteoporosis: concepts, conflicts, and prospects. J Clin Invest. 2005;115(12):3318-3325.
[38] LANE NE. Epidemiology, etiology, and diagnosis of osteoporosis. Am J Obstet Gynecol. 2006;194 (2 Suppl):S3-S11.
[39] DOMAZETOVIC V, MARCUCCI G, IANTOMASI T, et al. Oxidative stress in bone remodeling: role of antioxidants. Clin Cases Miner Bone Metab. 2017;14(2):209-216.
[40] XI JC, ZANG HY, GUO LX, et al. The PI3K/AKT cell signaling pathway is involved in regulation of osteoporosis. J Recept Signal Transduct Res. 2015;35(6):640-645.
[41] ZHU J, TANG Y, WU Q, et al. HIF-1α facilitates osteocyte-mediated osteoclastogenesis by activating JAK2/STAT3 pathway in vitro. J Cell Physiol. 2019;234(11):21182-21192.
[42] SHAW AT, GRAVALLESE EM. Mediators of inflammation and bone remodeling in rheumatic disease. Semin Cell Dev Biol. 2016;49:2-10.
[43] LOCANTORE P, DEL GATTO V, GELLI S, et al. The interplay between immune system and microbiota in osteoporosis. Mediators Inflamm. 2020;2020:3686749.
[44] ALONSO-PÉREZ A, FRANCO-TREPAT E, GUILLÁN-FRESCO M, et al. Role of Toll-Like Receptor 4 on Osteoblast Metabolism and Function. Front Physiol. 2018;9:504.
[45] CHUNG HY, LEE EK, CHOI YJ, et al. Molecular inflammation as an underlying mechanism of the aging process and age-related diseases. J Dent Res. 2011;90(7):830-840.
[46] KOUSTENI S. FoxOs: Unifying links between oxidative stress and skeletal homeostasis. Curr Osteoporos Rep. 2011;9(2):60-66.
[47] MA X, SU P, YIN C, et al. The roles of FoxO transcription factors in regulation of bone cells function. Int J Mol Sci. 2020;21(3):692.
[48] CHEN G, GOEDDEL DV. TNF-R1 signaling: a beautiful pathway. Science. 2002;296(5573): 1634-1635.
[49] ZHA L, HE L, LIANG Y, et al. TNF-α contributes to postmenopausal osteoporosis by synergistically promoting RANKL-induced osteoclast formation. Biomed Pharmacother. 2018;102:369-374. |