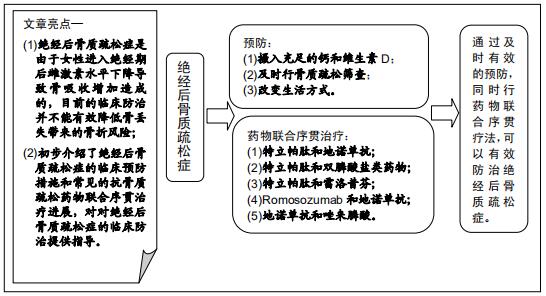
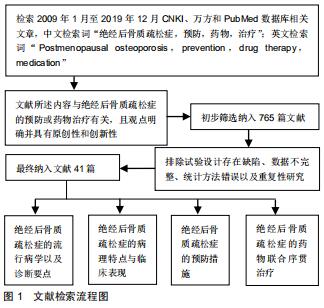
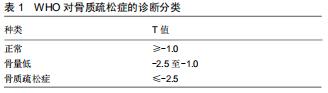
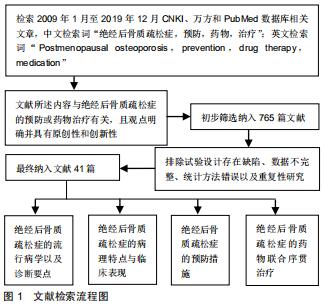
|
[1] YAN G, HUANG Y, CAO H, et al. Association of breastfeeding and postmenopausal osteoporosis in Chinese women: a community-based retrospective study. BMC Womens Health. 2019;19(1):110.
[2] SHEN Y, GRAY DL, MARTINEZ DS. Combined pharmacologic therapy in postmenopausal osteoporosis. Endocrinol Metab Clin North Am. 2016;46(1):193-206.
[3] THOR U, LIS S, JENS B. Bone matrix levels of dickkopf and sclerostin are positively correlated with bone mass and strength in postmenopausal osteoporosis. Int J Mol Sci. 2019; 20(12):2896.
[4] TANG D, JU C, LIU Y, et al. Therapeutic effect of icariin combined with stem cells on postmenopausal osteoporosis in rats. J Bone Miner Metabol. 2018;36(2):180-188.
[5] LIU H, LI B. Prospect of combination and sequential therapies for postmenopausal osteoporosis. Curr Pharm Design. 2017; 23(41):6251-6255.
[6] CHAN DC, LEE YS, WU YJ, et al. A 12-year ecological study of hip fracture rates among older Taiwanese adults. Calcif Tissue Int. 2013;93(5):397-404.
[7] MILLER DP. Management of severe osteoporosis. Exp Opin Pharmacother. 2016: 14656566.
[8] HILIGSMANN ML, EVERS SM, BEN SEDRINE W, et al. A systematic review of cost-effectiveness analyses of drugs for postmenopausal osteoporosis. Pharmacoeconomics. 2015; 33(3):205-224.
[9] MIGNOT MA, TAISNE N, LEGROUX I, et al. Bisphosphonate drug holidays in postmenopausal osteoporosis: effect on clinical fracture risk. Osteoporosis Int. 2017;28(12): 3431-3438.
[10] JACKSON RD, MYSIW WJ. Insights into the epidemiology of postmenopausal osteoporosis: the Women's Health Initiative. Semin Reprod Med. 2014;32(6):454-462.
[11] PRENTICE RL, PETTINGER MB, JACKSON RD, et al. Health risks and benefits from calcium and vitamin D supplementation: Women's Health Initiative clinical trial and cohort study. Osteoporosis Int. 2013;24(2):567-580.
[12] VERBRUGGE FH, EVELIEN G, KOEN M, et al. Who should receive calcium and vitamin D supplementation? Age Ageing. 2012; (5): 5.
[13] 中华医学会骨质疏松和骨矿盐疾病分会. 原发性骨质疏松症诊疗指南(2017)[J].中国骨质疏松杂志,2019,25(3):281-309.
[14] BISCHOFF-FERRARI HA, WILLETT WC, ORAV EJ, et al. A Pooled Analysis of Vitamin D Dose Requirements for Fracture Prevention. Obstetr Gynecol Surv. 2013;367(1):40-49.
[15] 王建华.骨质疏松症治疗药物的分类与用药选择[J].中华老年骨科与康复电子杂志,2019,5(5):297-300.
[16] ROSEN CJ, ADAMS JS, BIKLE DD, et al. The nonskeletal effects of Vitamin D: an endocrine society scientific statement. End Rev. 2012;33(3):456-492.
[17] DIAB DL, WATTS NB. Diagnosis and treatment of osteoporosis in older adults. Endocrinol Metab Clin North Am. 2013;42(2):305-317.
[18] US PREVENTIVE SERVICES TASK FORCE, CURRY SJ, KRIST AH, et al. Screening for osteoporosis to prevent fractures: us preventive services task force recommendation statement. JAMA. 2018;319(24):2521-2531.
[19] BONNICK SL, HARRIS ST, KENDLER DL, et al. Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):25-54.
[20] EVANS RK, NEGUS CH, CENTI AJ, et al. Peripheral QCT sector analysis reveals early exercise-induced increases in tibial bone mineral density. J Musculoskel Neur Int. 2012; 12(3):155-164.
[21] LIU GF, WANG ZQ, LIU L, et al. A network meta-analysis on the short-term efficacy and adverse events of different anti-osteoporosis drugs for the treatment of postmenopausal osteoporosis. J Cell Biochem.2018; 119(6):4469-4481.
[22] HSIAO FY, HSU WW. Comparative risks for cancer associated with use of calcitonin, bisphosphonates or selective estrogen receptor modulators among osteoporosis patients: a population-based cohort study. Jpn J Clin Oncol. 2017;47(10):935-941.
[23] BEAUDOIN C, JEAN S, BESSETTE L, et al. Denosumab compared to other treatments to prevent or treat osteoporosis in individuals at risk of fracture: a systematic review and meta-analysis. Osteoporos Int. 2016; 27(9): 2835-2844.
[24] MA YL, MARIN F, STEPAN J, et al. Comparative effects of teriparatide and strontium ranelate in the periosteum of iliac crest biopsies in postmenopausal women with osteoporosis. Bone. 2011;48(5):972-978.
[25] LI X, OMINSKY MS, WARMINGTON KS, et al. Sclerostin antibody treatment increases bone formation, bone mass, and bone strength in a rat model of postmenopausal osteoporosis. J Bone Miner Res. 2009;24(4):578-588.
[26] APPELMAN-DIJKSTRA NM, PAPAPOULOS SE. Sclerostin inhibition in the management of osteoporosis. Calcif Tissue Int. 2016;98(4):370-380.
[27] BLACK DM, REID IR, CAULEY JA, et al. The effect of 6 versus 9 years of zoledronic acid treatment in osteoporosis: a randomized second extension to the HORIZON-Pivotal Fracture Trial (PFT). J Bone Miner Res. 2015;30(5):934-944.
[28] GENNARI L, ROTATORI S, BIANCIARDI S, et al. Treatment needs and current options for postmenopausal osteoporosis. Exp Opin Pharmacother. 2016;17(8):1141-1152.
[29] TSAI JN, UIHLEIN AV, HANG L, et al. Teriparatide and denosumab, alone or combined, in women with postmenopausal osteoporosis: The DATA study randomised trial. Lancet. 2013; 382(9886):50-56.
[30] TSAI J, UIHLEIN A, BURNETT-BOWIE S, et al. Effects of two years of teriparatide, denosumab, or both on bone microarchitecture and strength (DATA-HRpQCT study). J Clin Endocrinol Metab.2016:20161160.
[31] LEDER BZ, TSAI JN, UIHLEIN AV, et al. Denosumab and teriparatide transitions in postmenopausal osteoporosis (the DATA-Switch study): extension of a randomised controlled trial. Lancet (London, England). 2015; 386(9999): 1147-1155.
[32] LEDER BZ, TSAI JN, NEER RM, et al. Response to therapy with teriparatide, denosumab, or both in postmenopausal women in the DATA (denosumab and teriparatide administration) study randomized controlled trial. J Clin Densitom. 2016;19(3):346-351.
[33] FINKELSTEIN JS, WYLAND JJ, HANG L, et al. Effects of teriparatide, alendronate, or both in women with postmenopausal osteoporosis. J Clin Endocrinol Metab. 2010;95(4):1838-1845.
[34] EASTELL R, NICKELSEN T, MARIN F, et al. Sequential treatment of severe postmenopausal osteoporosis after teriparatide: final results of the randomized, controlled European Study of Forsteo (EUROFORS). J Bone Miner Res. 2009; 24(4):726-736.
[35] MUSCHITZ C, KOCIJAN R, FAHRLEITNE-PAMMER A, et al. Overlapping and continued alendronate or raloxifene administration in patients on teriparatide: effects on areal and volumetric bone mineral density the CONFORS study. J Bone Miner Res. 2014;29(8):1777-1785.
[36] MCCLUNG MR, BROWN JP, DIEZ-PEREZ A, et al. Effects of 24 months of treatment with romosozumab followed by 12 months of denosumab or placebo in postmenopausal women with low bone mineral density: a randomized, double-blind, phase 2, parallel group study. J Bone Miner Res. 2018;33(8): 1397-1406.
[37] ANASTASILAKIS AD, POLYZOS SA, EFSTATHIADOU ZA, et al. Denosumab in treatment-naïve and pre-treated with zoledronic acid postmenopausal women with low bone mass: Effect on bone mineral density and bone turnover markers. Metabolism. 2015;64(10): 1291-1297.
[38] ANASTASILAKIS AD, POLYZOS SA, GKIOMISI A, et al. Denosumab versus zoledronic acid in patients previously treated with zoledronic acid. Osteoporosis Int. 2015; 26(10): 2521-2527.
[39] LIN J, ZHU J, WANG Y, et al. Chinese single herbs and active ingredients for postmenopausal osteoporosis: From preclinical evidence to action mechanism. Biosci Trends. 2017;11(5): 496-506.
[40] KOMM BS, MORGENSTERN D, YAMAMOTO LA, et al. The safety and tolerability profile of therapies for the prevention and treatment of osteoporosis in postmenopausal women. Exp Rev Clini Pharmacol. 2015;8(6):1-16.
[41] MANDEMA JW, ZHENG J, LIBANATI C, et al. Time course of bone mineral density changes with denosumab compared with other drugs in postmenopausal osteoporosis: a dose-response–based meta-analysis. J Clin Endocrinol Metab. 2014;99(10):3746-3755.
|