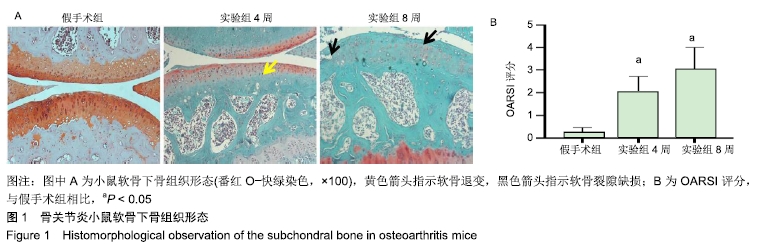
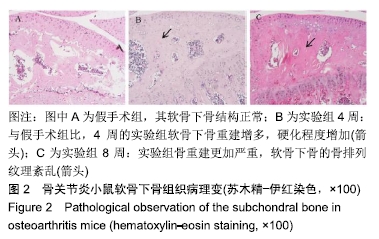
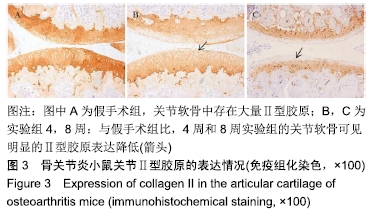
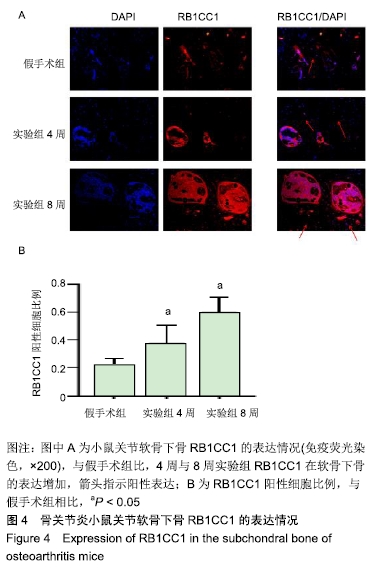
[1] CROSS M, SMITH E, HOY D, et al. The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis. 2014;73(7):1323-1330.
[2] HUNTER DJ, SCHOFIELD D, CALLANDER E. The individual and socioeconomic impact of osteoarthritis. Nat Rev Rheumatol. 2014;10(7): 437-441.
[3] LAMBOVA SN, MÜLLER-LADNER U. Osteoarthritis - Current Insights in Pathogenesis, Diagnosis and Treatment. Curr Rheumatol Rev. 2018; 14(2): 91-97.
[4] NELSON AE, ALLEN KD, GOLIGHTLY YM, et al. A systematic review of recommendations and guidelines for the management of osteoarthritis: The chronic osteoarthritis management initiative of the U.S. bone and joint initiative. Semin Arthritis Rheum. 2014;43(6):701-712.
[5] MURPHY L, HELMICK CG. The impact of osteoarthritis in the United States: a population-health perspective: A population-based review of the fourth most common cause of hospitalization in U.S. adults. Orthop Nurs. 2012;31(2):85-91.
[6] MURAOKA T, HAGINO H, OKANO T, et al. Role of subchondral bone in osteoarthritis development: a comparative study of two strains of guinea pigs with and without spontaneously occurring osteoarthritis. Arthritis Rheum. 2007;56(10):3366-3374.
[7] DORE DA. The role of subchondral bone in osteoarthritis. Hobart, Australia: University of Tasmania. 2011.
[8] MARUOTTI N, CORRADO A, CANTATORE FP. Osteoblast role in osteoarthritis pathogenesis. J Cell Physiol. 2017;232(11):2957-2963.
[9] OCHI Y, CHANO T, IKEBUCHI K, et al. RB1CC1 activates the p16 promoter through the interaction with hSNF5. Oncol Rep. 2011;26(4):805-812.
[10] LI L, WANG G, HU JS, et al. RB1CC1-enhanced autophagy facilitates PSCs activation and pancreatic fibrogenesis in chronic pancreatitis. Cell Death Dis. 2018;9(10):952.
[11] LI S, QIANG Q, SHAN H, et al. MiR-20a and miR-20b negatively regulate autophagy by targeting RB1CC1/FIP200 in breast cancer cells. Life Sci. 2016;147:143-152.
[12] CARAMÉS B, HASEGAWA A, TANIGUCHI N, et al. Autophagy activation by rapamycin reduces severity of experimental osteoarthritis. Ann Rheum Dis. 2012;71(4):575-581.
[13] ALMONTE-BECERRIL M, NAVARRO-GARCIA F, GONZALEZ-ROBLES A, et al. Cell death of chondrocytes is a combination between apoptosis and autophagy during the pathogenesis of Osteoarthritis within an experimental model. Apoptosis. 2010;15(5):631-638.
[14] FANG H, BEIER F. Mouse models of osteoarthritis: modelling risk factors and assessing outcomes. Nat Rev Rheumatol. 2014;10(7):413-421.
[15] GLASSON SS, ASKEW R, SHEPPARD B, et al. Characterization of and osteoarthritis susceptibility in ADAMTS-4-knockout mice. Arthritis Rheum. 2004;50(8):2547-2558.
[16] VON DER MARK K, KIRSCH T, NERLICH A, et al. Type X collagen synthesis in human osteoarthritic cartilage. Indication of chondrocyte hypertrophy. Arthritis Rheum. 1992;35(7):806-811.
[17] KOBAYASHI M, SQUIRES GR, MOUSA A, et al. Role of interleukin-1 and tumor necrosis factor alpha in matrix degradation of human osteoarthritic cartilage. Arthritis Rheum. 2005;52(1):128-135.
[18] CHOI MC, MARUYAMA T, CHUN CH, et al. Alleviation of Murine Osteoarthritis by Cartilage-Specific Deletion of IκBζ. Arthritis Rheumatol. 2018;70(9):1440-1449.
[19] BURR DB. Increased biological activity of subchondral mineralized tissues underlies the progressive deterioration of articular cartilage in osteoarthritis. J Rheumatol. 2005;32(6):1156-1159.
[20] LI G, YIN J, GAO J, et al. Subchondral bone in osteoarthritis: insight into risk factors and microstructural changes. Arthritis Res Ther. 2013;15(6): 223.
[21] FUNCK-BRENTANO T, COHEN-SOLAL M. Subchondral bone and osteoarthritis. Curr Opin Rheumatol. 2015;27(4):420-426.
[22] RADIN EL, PAUL IL, ROSE RM. Role of mechanical factors in pathogenesis of primary osteoarthritis. Lancet. 1972;1(7749):519-522.
[23] CHAN PMB, WEN C, YANG WC, et al. Is subchondral bone cyst formation in non-load-bearing region of osteoarthritic knee a vascular problem?. Med Hypotheses. 2017;109:80-83.
[24] LIN C, SHAO Y, ZENG C, et al. Blocking PI3K/AKT signaling inhibits bone sclerosis in subchondral bone and attenuates post-traumatic osteoarthritis [published correction appears in J Cell Physiol. 2019 Jun;234(6):9873]. J Cell Physiol. 2018;233(8):6135-6147.
[25] FANG H, HUANG L, WELCH I, et al. Early Changes of Articular Cartilage and Subchondral Bone in The DMM Mouse Model of Osteoarthritis. Sci Rep. 2018;8(1):2855.
[26] XUE J, XUE J, ZHANG J, et al. miR-130b-3p/301b-3p negatively regulated Rb1cc1 expression on myogenic differentiation of chicken primary myoblasts. Biotechnol Lett. 2017;39(11):1611-1619.
[27] CHEN J, YAN L, WANG H, et al. ZBTB38, a novel regulator of autophagy initiation targeted by RB1CC1/FIP200 in spinal cord injury. Gene. 2018; 678:8-16.
[28] CORONA VELAZQUEZ AF, JACKSON WT. So Many Roads: the Multifaceted Regulation of Autophagy Induction. Mol Cell Biol. 2018;38(21): e00303-18.
[29] NISHIMURA I, CHANO T, KITA H, et al. RB1CC1 protein suppresses type II collagen synthesis in chondrocytes and causes dwarfism. J Biol Chem. 2011;286(51):43925-43932.
[30] BOULEFTOUR W, GRANITO RN, VANDEN-BOSSCHE A, et al. Bone Shaft Revascularization After Marrow Ablation Is Dramatically Accelerated in BSP-/- Mice, Along With Faster Hematopoietic Recolonization. J Cell Physiol. 2017;232(9):2528-2537.
[31] BOULEFTOUR W, JUIGNET L, VERDIÈRE L, et al. Deletion of OPN in BSP knockout mice does not correct bone hypomineralization but results in high bone turnover. Bone. 2019;120:411-422.
[32] LI Z, WANG L, WEI J, et al. Bone-strengthening pill (BSP) promotes bone cell and chondrocyte repair, and the clinical and experimental study of BSP in the treatment of osteonecrosis of the femoral head. Oncotarget. 2017;8(57):97079-97089.
[33] BOLAMPERTI S, VILLA I, SPINELLO A, et al. Evidence for Altered Canonical Wnt Signaling in the Trabecular Bone of Elderly Postmenopausal Women with Fragility Femoral Fracture. Biomed Res Int. 2016;2016:8169614.
[34] SHAH M, GBURCIK V, REILLY P, et al. Local origins impart conserved bone type-related differences in human osteoblast behaviour. Eur Cell Mater. 2015;29:155-176.
[35] PESESSE L, SANCHEZ C, WALSH DA, et al. Bone sialoprotein as a potential key factor implicated in the pathophysiology of osteoarthritis. Osteoarthritis Cartilage. 2014;22(4):547-556.
[36] CUI Z, CRANE J, XIE H, et al. Halofuginone attenuates osteoarthritis by inhibition of TGF-β activity and H-type vessel formation in subchondral bone. Ann Rheum Dis. 2016;75(9):1714-1721.
[37] IKEBUCHI K, CHANO T, OCHI Y, et al. RB1CC1 activates the promoter and expression of RB1 in human cancer. Int J Cancer. 2009;125(4):861-867.
[38] SURANENI MV, MOORE JR, ZHANG D, et al. Tumor-suppressive functions of 15-Lipoxygenase-2 and RB1CC1 in prostate cancer. Cell Cycle. 2014;13(11):1798-1810.
[39] LI X, WAN X, CHEN H, et al. Identification of miR-133b and RB1CC1 as independent predictors for biochemical recurrence and potential therapeutic targets for prostate cancer. Clin Cancer Res. 2014;20(9): 2312-2325.
[40] LI J, FU Z, JIANG H, et al. IGF2-derived miR-483-3p contributes to macrosomia through regulating trophoblast proliferation by targeting RB1CC1. Mol Hum Reprod. 2018;24(9):444-452.
[41] CORONA VELAZQUEZ A, CORONA AK, KLEIN KA, et al. Poliovirus induces autophagic signaling independent of the ULK1 complex. Autophagy. 2018;14(7):1201-1213.
[42] GRUNWALD DS, OTTO NM, PARK JM, et al. GABARAPs and LC3s have opposite roles in regulating ULK1 for autophagy induction. Autophagy. 2019. doi:10.1080/15548627.2019.1632620.
[43] MORSELLI E, SHEN S, RUCKENSTUHL C, et al. p53 inhibits autophagy by interacting with the human ortholog of yeast Atg17, RB1CC1/FIP200. Cell Cycle. 2011;10(16):2763-2769.
[44] GUO M, MU Y, YU D, et al. Comparison of the expression of TGF-β1, E-cadherin, N-cadherin, TP53, RB1CC1 and HIF-1α in oral squamous cell carcinoma and lymph node metastases of humans and mice. Oncol Lett. 2018;15(2):1639-1645.
[45] YAO J, JIA L, KHAN N, et al. Deletion of autophagy inducer RB1CC1 results in degeneration of the retinal pigment epithelium. Autophagy. 2015;11(6):939-953.
[46] ZHANG Y, VASHEGHANI F, LI YH, et al. Cartilage-specific deletion of mTOR upregulates autophagy and protects mice from osteoarthritis. Ann Rheum Dis. 2015;74(7):1432-1440.
[47] CARAMÉS B, TANIGUCHI N, OTSUKI S, et al. Autophagy is a protective mechanism in normal cartilage, and its aging-related loss is linked with cell death and osteoarthritis. Arthritis Rheum. 2010;62(3): 791-801.
[48] ZHU X, YANG S, LIN W, et al. Roles of Cell Cyle Regulators Cyclin D1, CDK4, and p53 in Knee Osteoarthritis. Genet Test Mol Biomarkers. 2016;20(9):529-534.
[49] ZHONG G, LONG H, MA S, et al. miRNA-335-5p relieves chondrocyte inflammation by activating autophagy in osteoarthritis. Life Sci. 2019; 226:164-172.
[50] CHENG NT, MENG H, MA LF, et al. Role of autophagy in the progression of osteoarthritis: The autophagy inhibitor, 3-methyladenine, aggravates the severity of experimental osteoarthritis. Int J Mol Med. 2017;39(5): 1224-1232.
[51] XUE JF, SHI ZM, ZOU J, et al. Inhibition of PI3K/AKT/mTOR signaling pathway promotes autophagy of articular chondrocytes and attenuates inflammatory response in rats with osteoarthritis. Biomed Pharmacother. 2017;89:1252-1261.
|