[1] TEKAVEC E, JÖUD A, RITTNER R, et al. Population-based consultation patterns in patients with shoulder pain diagnoses. BMC Musculoskelet Disord. 2012;13:238.
[2] YOSHIDA M, COLLIN P, JOSSEAUME T, et al. Post-operative rotator cuff integrity, based on Sugayas classification, can reflect abduction muscle strength of the shoulder. Knee Surg Sports Traumatol Arthrosc. 2018;26(1):161-168.
[3] BOORMAN RS, MORE KD, HOLLINSHEAD RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27(3):444-448.
[4] KARPINSKI KATRIN, PLACHEL FABIAN, GERHARDT CHRISTIAN, et al. Different expectations of patients and surgeons with regard to rotator cuff repair. J Shoulder Elbow Surg. 2022;31:1096-1105.
[5] LE BT, WU XL, LAM PH, et al. Factors predicting rotator cuff retears: an analysis of 1000 consecutive rotator cuff repairs. Am J Sports Med. 2014;42(5):1134-1142.
[6] DUQUIN TR, BUYEA C, BISSON LJ. Which method of rotator cuff repair leads to the highest rate of structural healing A systematic review. Am J Sports Med.2010;38(4):835-841.
[7] TEUNIS T, LUBBERTS B, REILLY BT, et al. A systematic review and pooled analysis of the prevalence of rotator cuff disease with increasing age. J Shoulder Elbow Surg. 2014;23(12):1913-1921.
[8] KIM JH, HONG IT, RYU KJ, et al. Retear rate in the late postoperative period after arthroscopic rotator cuff repair. Am J Sports Med. 2014; 42(11):2606-2613.
[9] ROSSETTI L, KUNTZ LA, KUNOLD E, et al . The microstructure and micromechanics of the tendon-bone insertion. Nat Mater. 2017;16(6): 664-670.
[10] CHEN C, WANG Z, ZHENG C, et al. Characterization of zinc and calcium spatial distribution at the fibrocartilage of rabbit patella-patellar tendon complex: A synchrotron radiation micro X-ray fluorescence study. J Orthopaedic Translat. 2014;2(4):242-243.
[11] GENIN GM, THOMOPOULOS S. The tendon-to-bone attachment: Unification through disarray. Nat Mater. 2017;16(6):607-608.
[12] BEDEIR YEHIA H, SCHUMAIER ADAM P, ABU-SHEASHA GHADA et al. Type 2 retear after arthroscopic single-row, double-row and suture bridge rotator cuff repair: a systematic review. Eur J Orthop Surg Traumatol. 2019;29:373-382.
[13] LU H, LIU F, CHEN H, et al. The effect of low intensity pulsed ultrasound on bone tendon junction healing: Initiating after inflammation stage. J Orthop Res. 2016;34(10):1697-1706.
[14] SCHEIBEL MARKUS, BROWN ANNA, WOERTLER KLAUS, et al. Preliminary results after rotator cuff reconstruction augmented with an autologous periosteal flap. Knee Surg Sports Traumatol Arthrosc. 2007;15(3):305-314.
[15] ZHAO Y, DAI H F, LIU S S, et al. Therapeutic effect of autologous periosteum on the healing of tendon-bone interface in rabbit rotator cuff tear. Chinese Journal of Comparative Medicine. 2018;28(2):74-79.
[16] WANG X, ZHAI R, YANG J, et al. Effect of hamstring tendon transfected with adenovirus-mediated transforming growth factor β 1 gene on histomorphology of tendon-bone interface healing after anterior cruciate ligament reconstruction in rabbits. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2015;29(12):1488.
[17] FONT TELLADO S, BALMAYOR ER, VAN GRIENSVEN M. Strategies to engineer tendon/ligament-to-bone interface: Biomaterials,cells and growth factors. Adv Drug DelivRev. 2015;94:126-140.
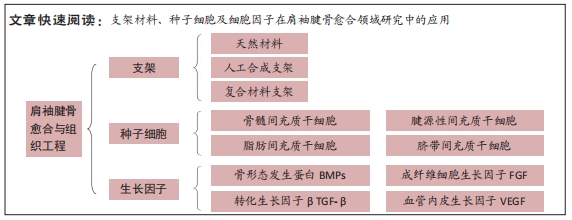
[18] 周勇,朱伟民,彭亮权,等.构建组织工程软骨支架材料:现状及未来[J].中国组织工程研究,2017,21(10):1604-1610.
[19] BONANI WALTER,SINGHATANADGIGE WEERASAK,PORNANONG ARAMWIT, et al. Natural Origin Materials for Osteochondral Tissue Engineering. Adv Exp Med Biol. 2018;1058:3-30.
[20] 刘嘉鑫,安丽萍,张广瑞,等.促进肩袖止点腱骨愈合的研究进展[J].中国骨伤,2020,33(7):684-688.
[21] SABETKISH S, KAJBAFZADEH AM, SABETKISH N, et al. Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix liver scaffolds. J Biomed Mater Res A. 2015;103(4): 1498-1508.
[22] LIAO J, XU B, ZHANG R, et al. Applications of decellularized materials in tissue engineering: advantages, drawbacks and current improvements, and future perspectives. J Mater Chem B. 2020;8:10023-10049.
[23] LU H, TANG Y, LIU F, et al. Comparative Evaluation of the Book-Type Acellular Bone Scaffold and Fibrocartilage Scaffold for Bone-Tendon Healing. J Orthop Res. 2019;37:1709-1722.
[24] 廖昊燃,余伟林,胡庆翔,等.生物学方法促进肩袖腱骨愈合研究进展[J].中华肩肘外科电子杂志,2021,9(2):183-186.
[25] ENCALADA-DIAZ I, COLE BJ, MACGILLIVRAY JD, et al . Rotator cuff repair augmentation using a novel polycarbonate polyurethane patch: preliminary results at 12 months’ follow-up. J Shoulder Elbow Surg. 2011;20(5):788-7894.
[26] CHEN P, CUI L, FU S, et al. The 3D-Printed PLGA Scaffolds Loaded with Bone Marrow-Derived Mesenchymal Stem Cells Augment the Healing of Rotator Cuff Repair in the Rabbits. Cell Transplant. 2020;29: 963689720973647.
[27] ATESOK K, DORAL MN, KARLSSON J, et al. Multilayer scaffolds in orthopaedic tissue engineering. Knee Surg Sports Traumatol Arthrosc. 2016;24(7):2365-2373.
[28] LI X, CHENG R, SUN Z, et al. Flexible bipolar nanofibrous membranes for improving gradient microstructure in tendon-to-bone healing. Acta Biomater. 2017;61: 204-216.
[29] TARAFDER S, BRITO JA, MINHAS S, et al. In situ tissue engineering of the tendon-to-bone interface by endogenous stem/progenitor cells. Biofabrication. 2019;12(1):015008.
[30] BHARDWAJ N, DEVI D, MANDAL BB. Tissue-engineered cartilage: the crossroads of biomaterials, cells and stimulating factors. Macromol Biosci. 2015;15:153-182.
[31] LEONG DANIEL J, SUN HUI B. Mesenchymal stem cells in tendon repair and regeneration: basic understanding and translational challenges. Ann N Y Acad Sci. 2016;1383:88-96.
[32] ZEINEDDINE HA , FRUSH TJ, SALEH ZM, et al. Applications of Tissue Engineering in Joint Arthroplasty: Current Concepts Update. Orthopedic Clinics of North America. 2017;48(3):275.
[33] BEITZEL K, SOLOVYOVA O, COTE MP, et al. The future role of mesenchymal stem cells in the management of shoulder disorders. Arthroscopy. 2013;29(10):1702-1711.
[34] HERNIGOU P, FLOUZAT LACHANIETTE CH, DELAMBRE J, et al. Biologic augmentation of rotator cuff repair with mesenchymalstem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop 2014;38(9):1811-1818.
[35] KIDA Y, MORIHARA T, MATSUDA K, et al. Bone marrow-derived cells from the footprint infiltrate into the repaired rotator cuff. J Shoulder Elbow Surg. 2013;22(2):197-205.
[36] CHEN HS, SU YT, CHAN TM, et al. Human adipose-derived stem cells accelerate the restoration of tensile strength of tendon and alleviate the progression of rotator cuff injury in a rat model. Cell Transplant. 2015;24(3):509-520.
[37] KIM YS, SUNG CH, CHUNG SH, et al. Does an injection of adipose-derived mesenchymal stem cells loaded in fibringlue influence rotator cuff repair outcomes? A clinical and magnetic resonance imaging study. Am J Sports Med. 2017;45:2010-2018.
[38] ZHENG CX, SUI BD, LIU N, et al. Adipose mesenchymal stem cells from osteoporotic donors preserve functionality and modulate systemic inflammatory microenvironment in osteoporotic cytotherapy. Sci Rep. 2018;8:5215.
[39] LI CY, WU XY, TONG JB, et al. Comparative analysis of human mesenchymal stem cells from bone marrow and adipose tissue under xeno-free conditions for cell therapy. Stem Cell Res Ther. 2015;6:55.
[40] VALENCIA MORA M, ANTUÑA ANTUÑA S, GARCÍA ARRANZ M, et al. Application of adipose tissue-derived stem cells in a rat rotator cuff repair model. Injury. 2014;45 Suppl 4:S22-S27.
[41] GIMBLE JM, KATZ AJ, BUNNELL BA. Adipose-derived stem cells for regenerative medicine. Circ Res. 2007;100:1249-1260
[42] NAGURA I, KOKUBU T, MIFUNE Y, et al. Characterization of progenitor cells derived from torn human rotator cuff tendons by gene expression patterns of chondrogenesis, osteogenesis, and adipogenesis. J Orthop Surg Res. 2016;11:40.
[43] SHEN W, CHEN J, YIN Z, et al. Allogenous tendon stem/progenitor cells in silk scaffold for functional shoulder repair. Cell Transplant. 2012; 21(5):943-958.
[44] HUANG K, CHEN CA, DONG SK, et al. Differentiation Ability of Tendon-Derived Stem Cells and Histological Characteristics of Rotator Cuff Remnant on the Greater Tuberosity Degenerated with Age and Chronicity. Arthroscopy. 2022;38(4):1037-1048.
[45] TAN Q, LUI PP, RUI YF, et al. Comparison of potentials of stem cells isolated from tendon and bone marrow for musculoskeletal tissue engineering. Tissue Eng Part A. 2012;18(7-8):840-851.
[46] BI Y, EHIRCHIOU D, KILTS TM, et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med. 2007;13(10):1219-1227.
[47] BAI L, LI D, LI J, et al. Bioactive molecules derived from umbilicalcord mesenchymal stem cells. Acta Histochem. 2016;118(8):761-769.
[48] PARK GY, KWON DR, LEE SC. Regeneration of Full-Thickness Rotator Cuff Tendon Tear After Ultrasound-Guided Injection With Umbilical Cord Blood-Derived Mesenchymal Stem Cells in a Rabbit Model. Stem Cells Transl Med. 2015;4:1344-1351.
[49] KWON DR, KIM KL, MOON YS. Regeneration of Chronic Rotator Cuff Tear in a Rabbit Model: Synergetic Benefits of Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells, Polydeoxyribonucleotides, and Microcurrent Therapy. Biomed Res Int. 2022;2022:6496773
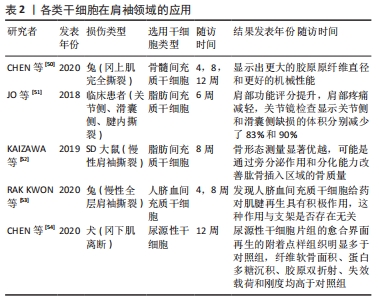
[50] CHEN P, CUI L, FU SC, et al. The 3D-Printed PLGA Scaffolds Loaded with Bone Marrow-Derived Mesenchymal Stem Cells Augment the Healing of Rotator Cuff Repair in the Rabbits. Cell Transplant. 2020;29: 963689720973647.
[51] JO CH, CHAI JW, JEONG EC, et al. Intratendinous Injection of Autologous Adipose Tissue-Derived Mesenchymal Stem Cells for the Treatment of Rotator Cuff Disease: A First-In-Human Trial. Stem Cells. 2018;36(9):1441-1450.
[52] KAIZAWA Y, FRANKLIN A, LEYDEN J, et al. Augmentation of chronic rotator cuff healing using adipose-derived stem cell-seeded human tendon-derived hydrogel. J Orthop Res. 2019;37(4):877-886.
[53] RAK KWON D, JUNG S, JANG J, et al. A 3-Dimensional Bioprinted Scaffold With Human Umbilical Cord Blood-Mesenchymal Stem Cells Improves Regeneration of Chronic Full-Thickness Rotator Cuff Tear in a Rabbit Model. Am J Sports Med. 2020;48(4):947-958.
[54] CHEN Y, XU Y, LI M, et al. Application of Autogenous Urine-Derived Stem Cell Sheet Enhances Rotator Cuff Healing in a Canine Model. Am J Sports Med. 2020;48(14):3454-3466.
[55] WÜRGLER HAURI CC, DOURTE LM, BARADET TC, et al. Temporal expression of 8 growth factors in tendon to bone healing in a rat supraspinatus model. J Shoulder Elbow Surg. 2007;16(5 Suppl): S198-S203.
[56] KABUTO Y, MORIHARA T, SUKENARI T, et al. Stimulation of rotator cuff repair by sustained release of bone morphogenetic protein-7 using a gelatinhydrogel sheet. Tissue Eng Part A.2015;21(13-14):2025-2033.
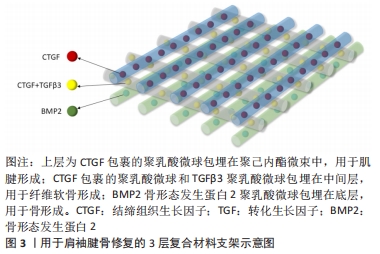
[57] CHEN P, CUI L, CHEN G, et al. The application of BMP-12-overexpressing mesenchymal stem cells loaded 3D-printed PLGA scaffolds in rabbit rotator cuff repair. Int J Biol Macromol. 2019;138:79-88.
[58] LAMPLOT JD, ANGELINE M, ANGELES J, et al. Distinct effects of platelet-rich plasma and BMP13 on rotator cuff tendon injury healing in a rat model. Am J Sports Med. 2014;42(12):2877-2887.
[59] HAN B, JONES IA, YANG Z, et al . Repair of rotator cuff tendon defects in aged rats using a growth factor injectable gel scaffold. Arthroscopy. 2020;36(3):629-637.
[60] YAMAKADO K, KITAOKA K, YAMADA H, et al . The influence of mechanical stress on graft healing in a bone tunnel. Arthroscopy. 2002; 18(1):82-90.
[61] WANG R, XU B, XU HG. Up-regulation of TGF-β promotes tendon- to- bone healing after anterior cruciate ligament reconstruction using bone marrow-derived mesenchymal stem cells through the TGF-β/MAPK signaling pathway in a New Zealand white rabbit model. Cell Physiol Biochem. 2017;41(1):213-226.
[62] SHAH M, FOREMAN DM, FERGUSON MW. Neutralisation of TGF-beta 1 and TGF-beta 2 or exogenous addition of TGF-beta 3 to cutaneous rat wounds reduces scarring. J Cell Sci. 1995;108(Pt 3):985-1002.
[63] MANNING CN, KIM HM, SAKIYAMA-ELBERT S, et al. Sustained delivery of transforming growth factor beta three enhances tendon- to- bone healing in a rat model. J Orthop Res. 2011;29(7):1099-1105.
[64] ZHAO S, ZHAO J, DONG S, et al. Biological augmentation of rotator cuff repair using bFGF-loaded electrospun poly (lactide-co-glycolide) fibrous membranes. Int J Nanomedicine. 2014;9:2373-2385.
[65] YONEMITSU R, TOKUNAGA T, SHUKUNAMI C, et al. Fibroblast Growth Factor 2 Enhances Tendon-to-Bone Healing in a Rat Rotator Cuff Repair of Chronic Tears. Am J Sports Med.2019;47:1701-1712.
[66] LU LY, MA M, CAI JF, et al. Effects of Local Application of Adipose-Derived Stromal Vascular Fraction on Tendon-Bone Healing after Rotator Cuff Tear in Rabbits. Chin Med J (Engl). 2018;131(21):2620-2622.
[67] HUANG Y, PAN M, SHU H, et al . Vascular endothelial growth factor enhances tendon-bone healing by activating Yes-associated protein for angiogenesis induction and rotator cuff reconstruction in rats. J Cell Biochem. 2020;121(3):2343-2353
[68] KORNTNER S, LEHNER C, GEHWOLF R, et al. Limiting angiogenesis to modulate scar formation. Adv Drug Deliv Rev. 2019;146:170-189
[69] JÄRVINEN TA. Neovascularisation in tendinopathy: from eradication to stabilisation? Br J Sports Med. 2020;54(1):1-2.
|