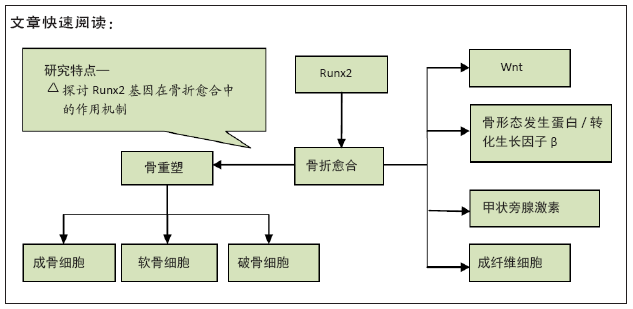
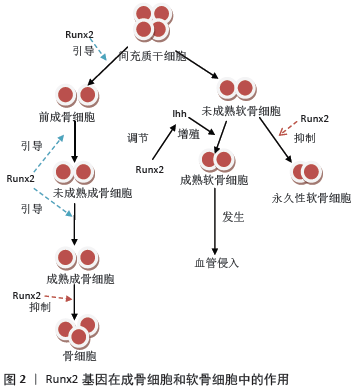
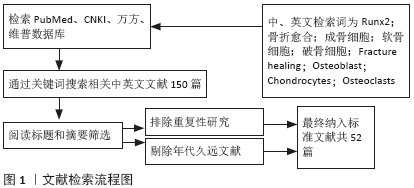
[1] TZIOUPIS C, GIANNOUDIS PV.Prevalence of long-bone non-unions.Injury. 2007;38 Suppl 2:S3-9.
[2] HAK DJ, FITZPATRICK D, BISHOP JA,et al. Delayed union and nonunions: epidemiology, clinical issues, and financial aspects.Injury. 2014;45 Suppl 2:S3-7.
[3] WYSOKINSKI D, BLASIAK J, PAWLOWSKA E.Role of RUNX2 in Breast Carcinogenesis.Int J Mol Sci.2015;16: 20969-20993.
[4] YOKOMIZO-NAKANO T,KUBOTA S,BAI J,et al.Overexpression of RUNX3 represses RUNX1 to drive transformation of myelodysplastic syndrome.Cancer Res.2020;80(12):2523-2536.
[5] OTÁLORA-OTÁLORA BA, HENRÍQUEZ B, LÓPEZ-KLEINE L,et al.RUNX family: Oncogenes or tumor suppressors (Review). Oncol Rep. 2019;42: 3-19.
[6] 黄耀星,余丹纯,孙小娟等.CD74与RUNX3在人胰腺癌细胞内存在相互作用[J].分子影像学杂志,2019,42(3):393-396.
[7] Ito Y, Bae SC, Chuang LS. The RUNX family: developmental regulators in cancer. Nat Rev Cancer. 2015;15:81-95.
[8] Bae SC,Lee YH.Phosphorylation, acetylation and ubiquitination: The molecular basis of RUNX regulation. Gene. 2006;366(1):58-66.
[9] NIEKE S, YASMIN N, KAKUGAWA K, et al.Unique N-terminal sequences in two Runx1 isoforms are dispensable for Runx1 function. BMC Dev Biol. 2017;17(1):14.
[10] OKURA H, SATO S, KISHIKAWA S,et al.Runx2-I isoform contributes to fetal bone formation even in the absence of specific N-terminal amino acids. PLoS ONE. 2014;9:e108294.
[11] TAKAHASHI A, DE ANDRÉS MC, HASHIMOTO K, et al.DNA methylation of the RUNX2 P1 promoter mediates MMP13 transcription in chondrocytes. Sci Rep. 2017;7:7771.
[12] KIM HJ, KIM WJ, RYOO HM. Post-Translational Regulations of Transcriptional Activity of RUNX2. Moleculer Cells. 2019;43(2):160-167.
[13] TAKARADA T, HINOI E, NAKAZATO R, et al. An analysis of skeletal development in osteoblast-specific and chondrocyte-specific runt-related transcription factor-2 (Runx2) knockout mice. J Bone Miner Res. 2013;28:2064-2069.
[14] ADHAMI MD, RASHID H, CHEN H, et al.Loss of Runx2 in committed osteoblasts impairs postnatal skeletogenesis. J Bone Miner Res. 2015;30:71-82.
[15] KOMORI T. Animal models for osteoporosis. Eur J Pharmacol. 2015;759: 287-294.
[16] MORIISHI T, FUKUYAMA R, ITO M, et al. Osteocyte network; a negative regulatory system for bone mass augmented by the induction of Rankl in osteoblasts and Sost in osteocytes at unloading. PLoS One. 2012;7(6):e40143.
[17] KOMORI T. Roles of Runx2 in Skeletal Development. Adv Exp Med Biol. 2017;962:83-93.
[18] KOMORI T. Regulation of Proliferation, Differentiation and Functions of Osteoblasts by Runx2. Int J Mol Sci. 2019;20(7):1694.
[19] NISHIMURA R, WAKABAYASHI M, HATA K, et al. Osterix Regulates Calcification and Degradation of Chondrogenic Matrices through Matrix Metalloproteinase 13 (MMP13) Expression in Association with Transcription Factor Runx2 during Endochondral Ossification. J Biol Chem. 2012;287(40):33179-33190.
[20] KONG L, ZUO R, WANG M, et al.Silencing MicroRNA-137-3p, which Targets RUNX2 and CXCL12 Prevents Steroid-induced Osteonecrosis of the Femoral Head by Facilitating Osteogenesis and Angiogenesis. Int J Biol Sci. 2020;16(4):655-670.
[21] ZHU W, HE X, HUA Y, et al. The E3 ubiquitin ligase WWP2 facilitates RUNX2 protein transactivation in a mono-ubiquitination manner during osteogenic differentiation. J Biol Chem. 2017;292:11178-11188.
[22] Komori T. Runx2, an inducer of osteoblast and chondrocyte differentiation. Histochem Cell Biol. 2018;149:313-323.
[23] Kurio N, Saunders C, Bechtold TE, et al. Roles of Ihh signaling in chondroprogenitor function in postnatal condylar cartilage. Matrix Biol. 2018;67:15-31.
[24] Liao L, Zhang S, Zhou GQ, et al. Deletion of Runx2 in condylar chondrocytes disrupts TMJ tissue homeostasis. J Cell Physiol. 2019;234: 3436-3444.
[25] Liao L, Jiang H, Fan Y, et al. Runx2 is required for postnatal intervertebral disc tissue growth and development. J Cell Physiol. 2019; 234(5):6679-6687.
[26] Provot S, Schipani E. Molecular mechanisms of endochondral bone development. Biochem Biophys Res Commun. 2005;328(3):658-665.
[27] Jiang Q, Qin X, Yoshida CA, et al.Antxr1, Which is a Target of Runx2, Regulates Chondrocyte Proliferation and Apoptosis. Int J Mol Sci. 2020;21(7):2425.
[28] ENOMOTO H, ENOMOTO-IWAMOTO M, IWAMOTO M, et al. Cbfa1 Is a Positive Regulatory Factor in Chondrocyte Maturation. J Biol Chem. 2000;275(12):8695-8702.
[29] CATHELINE SE, HOAK D, CHANG M, et al.Chondrocyte-Specific RUNX2 Overexpression Accelerates Post-traumatic Osteoarthritis Progression in Adult Mice. J Bone Miner Res. 2019;34(9):1676-1689.
[30] UETA C, IWAMOTO M, KANATANI N, et al. Skeletal malformations caused by overexpression of Cbfa1 or its dominant negative form in chondrocytes. J Cell Biol. 2001;153:87-100.
[31] HIMENO M, ENOMOTO H, LIU W, et al. Impaired vascular invasion of Cbfa1-deficient cartilage engrafted in the spleen.J Bone Miner Res. 2002;17:1297-305.
[32] HOFBAUER LC, KÜHNE CA, VIERECK V. The OPG/RANKL/RANK system in metabolic bone diseases. J Musculoskelet Neuronal Interact. 2004; 4(3):268-275.
[33] MARTIN A, XIONG J, KOROMILA T, et al. Estrogens antagonize RUNX2-mediated osteoblast-driven osteoclastogenesis through regulating RANKL membrane association. Bone. 2015;75: 96-104.
[34] ENOMOTO H, SHIOJIRI S, HOSHI K, et al. Induction of osteoclast differentiation by Runx2 through receptor activator of nuclear factor-kappa B ligand (RANKL) and osteoprotegerin regulation and partial rescue of osteoclastogenesis in Runx2-/- mice by RANKL transgene. J Biol Chem. 2003;278:23971-23977.
[35] GENG YM, LIU CX, LU WY, et al. LAPTM5 is transactivated by RUNX2 and involved in RANKL trafficking in osteoblastic cells. Mol Med Rep. 2019;20(5):4193-4201.
[36] MOHAMMED MK, SHAO C, WANG J, et al. Wnt/β-catenin signaling plays an ever-expanding role in stem cell self-renewal, tumorigenesis and cancer chemoresistance. Genes Dis. 2016;3:11-40.
[37] HUYBRECHTS Y, MORTIER G, BOUDIN E, et al.WNT Signaling and Bone: Lessons From Skeletal Dysplasias and Disorders. Front Endocrinol (Lausanne). 2020;11:165.
[38] CHEN Y, HU Y, YANG L, et al. Runx2 alleviates high glucose-suppressed osteogenic differentiation via PI3K/AKT/GSK3β/β-catenin pathway. Cell Biol Int. 2017;41:822-832.
[39] WU M, WANG Y, SHAO JZ, et al. Cbfβ governs osteoblast-adipocyte lineage commitment through enhancing β-catenin signaling and suppressing adipogenesis gene expression. Proc Natl Acad Sci U.S.A. 2017;114:10119-10124.
[40] YU X, SHEN G, REN H, et al. TGFβ-induced factor homeobox 2 blocks osteoblastic differentiation through targeting pSmad3/HDAC4/H4ac/Runx2 axis. J Cell Physiol. 2019;234:21284-21293.
[41] YANG B, LIN X, TAN J, et al. Root bark of Sambucus Williamsii Hance promotes rat femoral fracture healing by the BMP-2/Runx2 signaling pathway. J Ethnopharmacol. 2016;191:107-114.
[42] LIU DD, ZHANG JC, ZHANG Q, et al. TGF‐β/BMP signaling pathway is involved in cerium‐promoted osteogenic differentiation of mesenchymal stem cells. J Cell Biochem. 2013;114(5):1105-1114.
[43] YAN J, LI J, HU J, et al. Smad4 deficiency impairs chondrocyte hypertrophy via the Runx2 transcription factor in mouse skeletal development. J Biol Chem. 2018;293:9162-9175.
[44] KOPF J, PAARMANN P, HIEPEN C, et al. BMP growth factor signaling in a biomechanical context. Biofactors. 2014;40:171-187.
[45] QIN L, RAGGATT LJ, PARTRIDGE NC. Parathyroid hormone: a double-edged sword for bone metabolism. Trends Endocrinol Metab. 2004;15: 60-65.
[46] 庄颖,庞希瑶,蔡琴,等.rhPTH(1-34)间断刺激对人根尖牙乳头干细胞增殖及成牙成骨分化的影响[J].口腔生物医学,2018,9(3): 125-128.
[47] LAI K, XI Y, MIAO X, et al. PTH coatings on titanium surfaces improved osteogenic integration by increasing expression levels of BMP-2/Runx2/Osterix. Rsc Advances. 2017;7(89):56256-56265.
[48] QIN X, JIANG Q, MIYAZAKI T, et al. Runx2 regulates cranial suture closure by inducing hedgehog, Fgf, Wnt and Pthlh signaling pathway gene expressions in suture mesenchymal cells.Hum Mol Genet. 2019;28:896-911.
[49] Kim JM, Yang YS, Park KH, et al. The ERK MAPK Pathway Is Essential for Skeletal Development and Homeostasis.Int J Mol Sci. 2019;20(8): 1803.
[50] Li Y, Ge C, Franceschi RT. MAP Kinase-Dependent RUNX2 Phosphorylation Is Necessary for Epigenetic Modification of Chromatin During Osteoblast Differentiation. J Cell Physiol. 2017;232(9): 2427-2435.
[51] KIM HJ, KIM JH, BAE SC, et al. The protein kinase C pathway plays a central role in the fibroblast growth factor-stimulated expression and transactivation activity of Runx2. J Biol Chem. 2003;278(1):319-326.
[52] KAWANE T, QIN X, JIANG Q, et al. Runx2 is required for the proliferation of osteoblast progenitors and induces proliferation by regulating Fgfr2 and Fgfr3. Sci Rep. 2018;8(1):13551.
|