| [1] Morisseau C, Hammock BD. Impact of soluble epoxide hydrolase and epoxyeicosanoids on human health. Annu Rev Pharmacol Toxicol. 2013;53(2):37-58. [2] 党海霞,霍欣茹,王福,等.14,15-环氧二十碳三烯酸促进颅骨缺损骨再生[J].口腔医学研究,2016,32(7):692-695.[3] Abdu E, Bruun DA, Yang D, et al. Epoxyeicosatrienoic acids enhance axonal growth in primary sensory and cortical neuronal cell cultures. J Neurochem. 2011;117(4): 632-642. [4] Sander AL, Jakob H, Sommer K, et al. Cytochrome P450-derived epoxyeicosatrienoic acids accelerate wound epithelialization and neovascularization in the hairless mouse ear wound model. Langenbecks Arch Surg. 2011;396(8): 1245-1253. [5] Panigrahy D, Kalish BT, Huang S, et al. Epoxyeicosanoids promote organ and tissue regeneration. Proc Natl Acad Sci U S A. 2013;110(33):13528-13533. [6] Vanella L, Kim DH, Sodhi K, et al. Crosstalk between EET and HO-1 downregulates Bach1 and adipogenic marker expression in mesenchymal stem cell derived adipocytes. Prostaglandins Other Lipid Mediat. 2011;96(1):54-62. [7] Guan H, Zhao L, Cao H, et al. Epoxyeicosanoids suppress steoclastogenesis and prevent ovariectomy-induced bone loss. FASEB J. 2015;29(3):1092-1101. [8] Shahabi P, SiestG, Visvikis-siest S. cytochrome P450 epoxygenase-derived epoxyeicosatrienoic acids. Drug Metab Rev. 2014;46(1):33-56. [9] Buczynski MW, Dumlao DS, Dennis EA. Thematic review series: proteomics. An integrated omics analysis of eicosanoid biology. J Lipid Res. 2009;50(11):1015-1038. [10] Funk CD. Prostaglandins and leukotrienes: advances in eicosanoid biology. Science. 2001;294(5548):1871-1875. [11] Buch PR, Desai I, Balakrishnan S. COX-2 activity and expression pattern during regenerative wound healing of tail in lizard Hemidactylus flaviviridis. Prostaglandins Other Lipid Mediat. 2018;135:11-15. [12] Chiang N, Riley IR, Dalli J, et al. New maresin conjugates in tissue regeneration pathway counters leukotriene D4-stimulated vascular responses. FASEB J. 2018;32(7): 4043-4052. [13] He J, Wang C, Zhu Y, et al. Soluble epoxide hydrolase: A potential target for metabolic diseases. J Diabetes. 2015; 8(3):305-313. [14] Elshenawy OH, Anwar-Mohamed A, El-Kadi AO. 20-Hydroxyeicosatetraenoic acid is a potential therapeutic target in cardiovascular diseases. Curr Drug Metab. 2013; 14(6):706-719. [15] Hoopes SL, Garcia V, Edin ML, et al. Vascular actions of 20-HETE.Prostaglandins Other Lipid Mediat. 2015;120:9-16. [16] Imig JD. Epoxyeicosatrienoic Acids and 20-Hydroxyeicosatetraenoic Acid on Endothelial and Vascular Function. Adv Pharmacol. 2016;77:105-141. [17] Garcia V, Schwartzman ML. Recent developments on the vascular effects of 20-hydroxyeicosatetraenoic acid. Curr Opin Nephrol Hypertens. 2017;26(2):74-82. [18] Swardfager W, Hennebelle M, Yu D, et al. Metabolic/ inflammatory/vascular comorbidity in psychiatric disorders; soluble epoxide hydrolase (sEH) as a possible new target. Neurosci Biobehav Rev. 201887:56-66. [19] Lee KS, Liu JY, Wagner KM, et al. Optimized inhibitors of soluble epoxide hydrolase improve in vitro target residence time and in vivo efficacy. J Med Chem. 2014;57(16):7016-7030. [20] Yu Z, Xu F, Huse LM, et al. Soluble epoxide hydrolase regulates hydrolysis of vasoactive epoxyeicosatrienoic acids. Circ Res. 2000;87(11):992-998. [21] Imig JD, Zhao X, Zaharis CZ, et al. An orally active epoxide hydrolase inhibitor lowers blood pressure and provides renal protection in salt-sensitive hypertension. Hypertension. 2005; 46(4):975-981. [22] Luria A, Bettaieb A, Xi Y, et al. Soluble epoxide hydrolase deficiency alters pancreatic islet size and improves glucose homeostasis in a model of insulin resistance. Proc Natl Acad Sci U S A. 2011;108(22):9038-9043. [23] Fleming I. Vascular cytochrome p450 enzymes: physiology and pathophysiology. Trends Cardiovasc Med. 2008;18(1): 20-25. [24] Street J, Bao M, Bunting S, et al. Vascular endothelial growth factor stimulates bone repair by promoting angiogenesis and bone turnover. Proc Natl Acad Sci U S A. 2002;99(15): 9656-9661. [25] Folkman J. Angiogenesis: an organizing principle for drug discovery? . Nat Rev Drug Discov. 2007;6(4):273-286. [26] 鲍小刚,许国华.组织工程骨的快速血管化[J].中国组织工程研究,2015,19(7):1063-1069.[27] Zhang C, Harder DR. Cerebral capillary endothelial cell mitogenesis and morphogenesis induced by astrocytic epoxyeicosatrienoic acid. Stroke. 2002;33(12):2957-2964. [28] Capozzi ME, McCollum GW, Penn JS. The role of cytochrome P450 epoxygenases in retinal angiogenesis. Invest Ophthalmol Vis Sci. 2014;55(7):4253-4260. [29] Pulin Li, Jamie L, Lahvic, et al. Epoxyeicosatrienoic Acids Enhance Embryonic Haematopoiesis and Adult Marrow Engraftment. Nature. 2015;523(7561):468-471. [30] Tassi E, McDonnell K, Gibby KA, et al. Impact of fibroblast growth factor-binding protein–1 expression on angiogenesis and wound healing. Am J Pathol. 2011;179(5):2220-2232. [31] Cheranov SY, Karpurapu M, Wang D, et al. An essential role for SRC-activated STAT-3 in 14, 15-EET–induced VEGF expression and angiogenesis. Blood. 2008;111(12):5581-5591. [32] Michaelis UR, Fisslthaler B, Barbosa-Sicard E, et al. Cytochrome P450 epoxygenases t2C8 and 2C9 are implicated in hypoxia-induced endothelial cell migration and angiogenesis. J Cell Sci. 2005;118(23):5489-5498. [33] Chen JK, Capdevila J, Harris RC. Heparin-binding EGF-like growth factor mediates the biological effects of P450 arachidonate epoxygenase metabolites in epithelial cells. Proc Natl Acad Sci U S A. 2002;99(9):6029-6034. [34] Xu DY, Davis BB, Wang ZH. A potent soluble epoxide hydrolase inhibitor, t-AUCB, acts through PPARγ to modulate the function of endothelial progenitor cells from patients with acute myocardial infarction. Int J Cardiol. 2013;167(4):1298-1304. [35] Revermann M, Schloss M, Barbosa-Sicard E. Soluble epoxide hydrolase deficiency attenuates neointima formation in the femoral cuff model of hyperlipidemic mice. Arterioscler Thromb Vasc Biol. 2010;30(5):909-914. [36] Davis BB, Thompson DA, Howard LL. Inhibitors of soluble epoxide hydrolase attenuate vascular smooth muscle cell proliferation. Proc Natl Acad Sci U S A. 2002;99(4):2222-2227. [37] Ng VY, Morisseau C, Falck JR, et al. Inhibition of smooth muscle proliferation by urea-based alkanoic acids via peroxisome proliferator-activated receptor alpha-dependent repression of cyclin D1. Arterioscler Thromb Vasc Biol. 2006; 26(11):2462-2468. [38] Kim HS, Kim SK, Kang KW. Differential Effects of sEH Inhibitors on the Proliferation and Migration of Vascular Smooth Muscle Cells. Int J Mol Sci. 2017;18(12):2683. [39] Carmeliet P. Mechanisms of angiogenesis and arteriogenesis. Nat Med. 2000;6(4):389-395. [40] Fleming I, Michaelis UR, Bredenkötter D, et al. Endothelium-derived hyperpolarizing factor synthase (Cytochrome P450 2C9) is a functionally significant source of reactive oxygen species in coronary arteries. Circ Res. 2001; 88(1):44-51[41] Eming SA, Wynn TA, Martin P. Inflammation and metabolism in tissue repair and regeneration. Science. 2017;356(6342): 1026-1030. [42] Liu H, Li D, Zhang Y, et al. Inflammation, mesenchymal stem cells and bone regeneration. Histochem Cell Biol. 2018;149 (4): 393-404. [43] Runyan CM, Gabrick KS. Biology of Bone Formation, Fracture Healing, and Distraction Osteogenesis. J Craniofac Surg. 2017;28(5):1380-1389. [44] Chen FM, Lu H, Wu LA, et al. Surface-engineering of glycidyl methacrylated dextran/gelatin microcapsules with thermo-responsive poly(N-isopropylacrylamide) gates for controlled delivery of stromal cell-derived factor-1α. Biomaterials. 2013;34(27):6515-6527. [45] Godot V, Arock M, Garcia G, et al. H4 histamine receptor mediates optimal migration of mast cell precursors to CXCL12. J Allergy Clin Immunol. 2007;120(4):827-834. [46] Melief SM, Zwaginga JJ, Fibbe WE, et al. Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cells Transl Med. 2013;2(6):455-463. [47] Kim KS, Kim HS, Park JM, et al. Long-term immunomodulatory effect of amniotic stem cells in an Alzheimer's disease model. Neurobiol Aging. 2013;34(10): 2408-20. [48] Liu D, Xu J, Liu O, et al. Mesenchymal stem cells derived from inflamed periodontal ligaments exhibit impaired immunomodulation. J Clin Periodontol. 2012;39:1174-1182. [49] Liu Y, Wang L, Kikuiri T, et al. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via IFN-γ and TNF-α. Nat Med. 2011;17:1594-1601. [50] Vanella L, Kim D H, Sodhi K, Barbagallo I, Burgess AP, Falck JR, Schwartzman ML, Abraham NG. Crosstalk between EET and HO-1 downregulates Bach1 and adipogenic marker expression in mesenchymal stem cell derived adipocytes. Prostaglandins Other Lipid Mediat. 2011;96(1):54-62. [51] Harada S, Rodan GA. Control of osteoblast function and regulation of bone mass. Nature. 2003;15; 423(6937): 349-355. [52] Zhu XB, Lin WJ, Lv C, et al. MicroRNA-539 promotes osteoblast proliferation and differentiation and osteoclast apoptosis through the AXNA-dependent Wnt signaling pathway in osteoporotic rats. J Cell Biochem. 2018 Jun 12. [53] Bao Y, Gao Y, Du M, et al. opical Treatment with Xiaozheng Zhitong Paste (XZP) Alleviates Bone Destruction and Bone Cancer Pain in a Rat Model of Prostate Cancer-Induced Bone Pain by Modulating the RANKL/RANK/OPG Signaling. Evid Based Complement Alternat Med. 2015;2015:215892. [54] Rangaswami H, Schwappacher R, Tran T, et al. Protein kinase G and focal adhesion kinase converge on Src/Akt/ β-catenin signaling module in osteoblast mechanotransduction. J Biol Chem. 2012;287(25): 21509-21519. [55] Liang D, Yang M, Guo B, et al. Zinc inhibits H(2)O(2)-induced MC3T3-E1 cells apoptosis via MAPK and PI3K/AKT pathways. Biol Trace Elem Res. 2012;148(3):420-429. [56] Oni-Orisan A, Deng Y, Schuck RN, et al. Dual modulation of cyclooxygenase and CYP epoxygenase metabolism and acute vascular inflammation in mice. Prostaglandins Other Lipid Mediat. 2013;104-105:67-73. [57] Wang H, Lin L, Jiang J, et al. Up-regulation of endothelial nitric-oxide synthase by endothelium-derived hyperpolarizing factor involves mitogen-activated protein kinase and protein kinase C signaling pathways. J Pharmacol Exp Ther. 2003; 307(2):753–764. [58] Abraham NG, Sodhi K, Silvis AM, et al. CYP2J2 targeting to endothelial cells attenuates adiposity and Vascular Dysfunction in Mice Fed a High-Fat Diet by Reprogramming Adipocyte Phenotype. Hypertension. 2014;64(6):1352–1361. [59] Duflot T, Roche C, Lamoureux F, et al. Design and discovery of soluble epoxide hydrolase inhibitors for the treatment of cardiovascular diseases. Expert Opin Drug Discov. 2014;9(3): 229-243. [60] Lazaar AL, Yang L, Boardley RL, et al. Pharmacokinetics, pharmacodynamics and adverse event profile of GSK2256294, a novel soluble epoxide hydrolase inhibitor. Br J Clin Pharmacol. 2016;81(5):971-979. [61] Chen D, Whitcomb R, MacIntyre E, et al. Pharmacokinetics and pharmacodynamics of AR9281, an inhibitor of soluble epoxide hydrolase, in single- and multiple-dose studies in healthy human subjects. J Clin Pharmacol. 2012;52(3): 319-328. [62] Bellien J, Iacob M, Remy-Jouet I, et al. Epoxyeicosatrienoic acids contribute with altered nitric oxide and endothelin-1 pathways to conduit artery endothelial dysfunction in essential hypertension. Circulation. 2012;125(10):1266-1275. [63] Noonan JE, Dusting GJ, Nguyen TT, et al. Flicker light-induced retinal vasodilation is unaffected by inhibition of epoxyeicosatrienoic acids and prostaglandins in humans. Invest Ophthalmol Vis Sci. 2014;55(10):7007-7013. [64] Liu JY, Park SH, Morisseau C, et al. Sorafenib has soluble epoxide hydrolase inhibitory activity, which contributes to its effect profile in vivo. Mol Cancer Ther. 2009;8(8):2193-2203. [65] Kim JH, Cho CW, Tai BH, et al. Soluble Epoxide Hydrolase Inhibitory Activity of Selaginellin Derivatives from Selaginella tamariscina. Molecules. 2015;20(12):21405-21414. [66] Kitamura S, Morisseau C, Inceoglu B, et al. Potent natural soluble epoxide hydrolase inhibitors from Pentadiplandra brazzeana baillon: synthesis, quantification, and measurement of biological activities in vitro and in vivo. PLoS One. 2015;6:10(2):e0117438. [67] Bai MM, Shi W, Tian JM, et al. Soluble epoxide hydrolase inhibitory and anti-inflammatory components from the leaves of Eucommia ulmoides Oliver (duzhong). J Agric Food Chem. 2015;63(8):2198-2205. [68] Sun YN, Li W, Song SB, et al. A new phenolic derivative with soluble epoxide hydrolase and nuclear factor-kappaB inhibitory activity from the aqueous extract of Acacia catechu. Nat Prod Res. 2016;30(18):2085-2092. [69] Sun YN,Kim JH,Li W,et al. Soluble epoxide hydrolase inhibitory activity of anthraquinone components from Aloe. Bioorg Med Chem. 2015;23(20):6659-6665. |
.jpg) 文题释义:
血管生成:是从已有的毛细血管或毛细血管后静脉发展而形成的,首先是血管基底膜降解和血管内皮细胞被激活,进而血管内皮细胞发生增殖和迁移,最后重建形成新的血管和血管网。血管形成是促血管形成因子和抑制因子多种生物因子协调作用的复杂过程,正常情况下二者处于平衡状态,一旦该平衡打破,血管系统即被激活,使血管生成过度或抑制血管系统使血管退化。
血管内皮生长因子:是高度保守的同源二聚体糖蛋白,是一种强有力的血管生成因子,由于mRNA不同的剪切方式,产生多种蛋白形式,其中血管内皮生长因子121,145,165是分泌型可溶性蛋白,能直接作用于血管内皮细胞促进血管内皮细胞增殖,增加血管通透性。
文题释义:
血管生成:是从已有的毛细血管或毛细血管后静脉发展而形成的,首先是血管基底膜降解和血管内皮细胞被激活,进而血管内皮细胞发生增殖和迁移,最后重建形成新的血管和血管网。血管形成是促血管形成因子和抑制因子多种生物因子协调作用的复杂过程,正常情况下二者处于平衡状态,一旦该平衡打破,血管系统即被激活,使血管生成过度或抑制血管系统使血管退化。
血管内皮生长因子:是高度保守的同源二聚体糖蛋白,是一种强有力的血管生成因子,由于mRNA不同的剪切方式,产生多种蛋白形式,其中血管内皮生长因子121,145,165是分泌型可溶性蛋白,能直接作用于血管内皮细胞促进血管内皮细胞增殖,增加血管通透性。.jpg) 文题释义:
血管生成:是从已有的毛细血管或毛细血管后静脉发展而形成的,首先是血管基底膜降解和血管内皮细胞被激活,进而血管内皮细胞发生增殖和迁移,最后重建形成新的血管和血管网。血管形成是促血管形成因子和抑制因子多种生物因子协调作用的复杂过程,正常情况下二者处于平衡状态,一旦该平衡打破,血管系统即被激活,使血管生成过度或抑制血管系统使血管退化。
血管内皮生长因子:是高度保守的同源二聚体糖蛋白,是一种强有力的血管生成因子,由于mRNA不同的剪切方式,产生多种蛋白形式,其中血管内皮生长因子121,145,165是分泌型可溶性蛋白,能直接作用于血管内皮细胞促进血管内皮细胞增殖,增加血管通透性。
文题释义:
血管生成:是从已有的毛细血管或毛细血管后静脉发展而形成的,首先是血管基底膜降解和血管内皮细胞被激活,进而血管内皮细胞发生增殖和迁移,最后重建形成新的血管和血管网。血管形成是促血管形成因子和抑制因子多种生物因子协调作用的复杂过程,正常情况下二者处于平衡状态,一旦该平衡打破,血管系统即被激活,使血管生成过度或抑制血管系统使血管退化。
血管内皮生长因子:是高度保守的同源二聚体糖蛋白,是一种强有力的血管生成因子,由于mRNA不同的剪切方式,产生多种蛋白形式,其中血管内皮生长因子121,145,165是分泌型可溶性蛋白,能直接作用于血管内皮细胞促进血管内皮细胞增殖,增加血管通透性。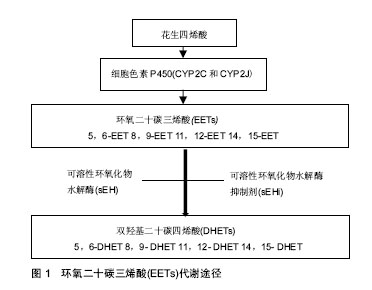

.jpg) 文题释义:
血管生成:是从已有的毛细血管或毛细血管后静脉发展而形成的,首先是血管基底膜降解和血管内皮细胞被激活,进而血管内皮细胞发生增殖和迁移,最后重建形成新的血管和血管网。血管形成是促血管形成因子和抑制因子多种生物因子协调作用的复杂过程,正常情况下二者处于平衡状态,一旦该平衡打破,血管系统即被激活,使血管生成过度或抑制血管系统使血管退化。
血管内皮生长因子:是高度保守的同源二聚体糖蛋白,是一种强有力的血管生成因子,由于mRNA不同的剪切方式,产生多种蛋白形式,其中血管内皮生长因子121,145,165是分泌型可溶性蛋白,能直接作用于血管内皮细胞促进血管内皮细胞增殖,增加血管通透性。
文题释义:
血管生成:是从已有的毛细血管或毛细血管后静脉发展而形成的,首先是血管基底膜降解和血管内皮细胞被激活,进而血管内皮细胞发生增殖和迁移,最后重建形成新的血管和血管网。血管形成是促血管形成因子和抑制因子多种生物因子协调作用的复杂过程,正常情况下二者处于平衡状态,一旦该平衡打破,血管系统即被激活,使血管生成过度或抑制血管系统使血管退化。
血管内皮生长因子:是高度保守的同源二聚体糖蛋白,是一种强有力的血管生成因子,由于mRNA不同的剪切方式,产生多种蛋白形式,其中血管内皮生长因子121,145,165是分泌型可溶性蛋白,能直接作用于血管内皮细胞促进血管内皮细胞增殖,增加血管通透性。