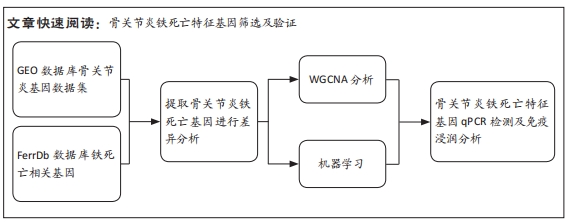
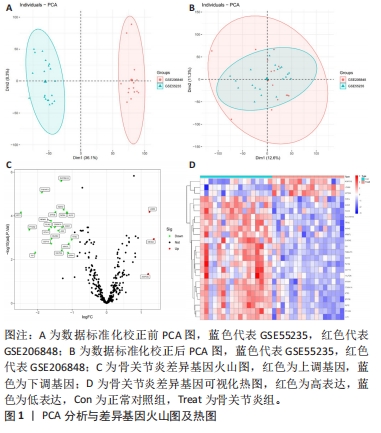
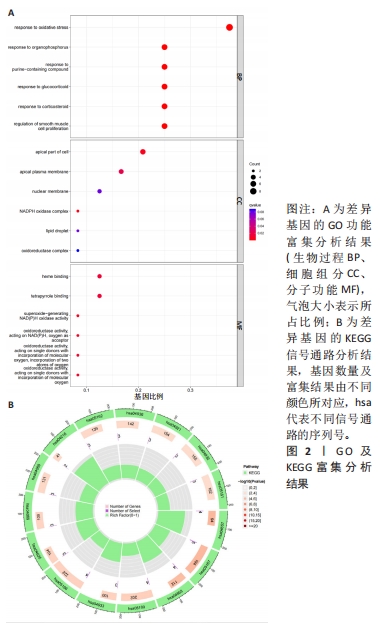
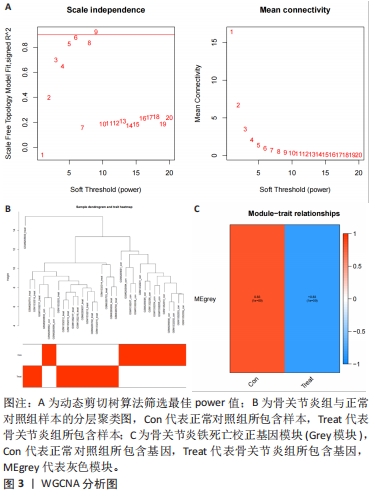
[1] TANG X, WANG S, ZHAN S, et al. The Prevalence of Symptomatic Knee Osteoarthritis in China: Results From the China Health and Retirement Longitu dinal Study. Arthritis Rheumatol. 2016;68(3):648-653.
[2] ZHANG Y, YANG J, YU T, et al. Epidemiological Characteristics of Osteoarthritis in Yichang City-Hubei Province, China, 2017-2018. China CDC Wkly. 2021;3(16): 335-339.
[3] CHAILIER E, DEROYER C, CIREGIA F, et al.Chondrocyte dedifferentiation and osteoarthritis(OA). Bioc hem Pharmacol. 2019;165:49-65.
[4] JIANG X, STOCKWELL BR, CONRAD M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22(4):266-282.
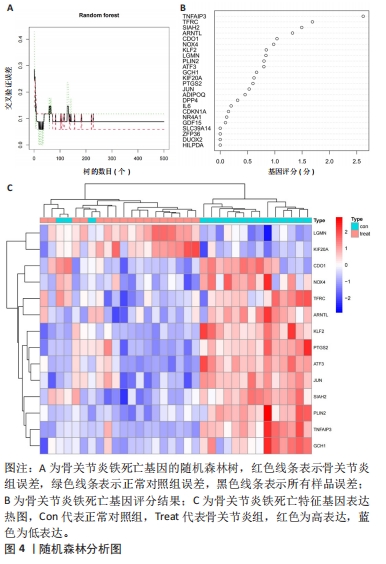
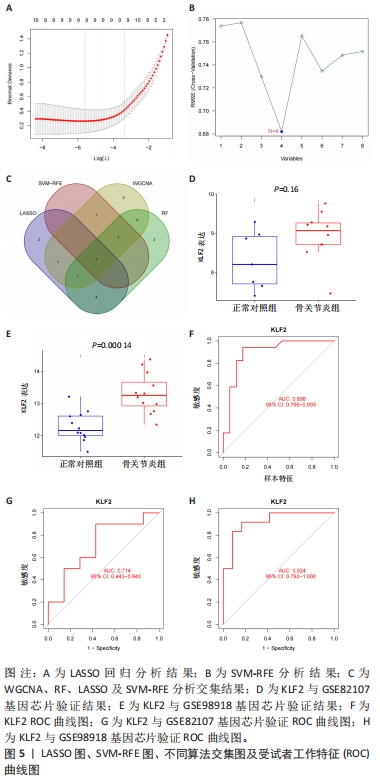
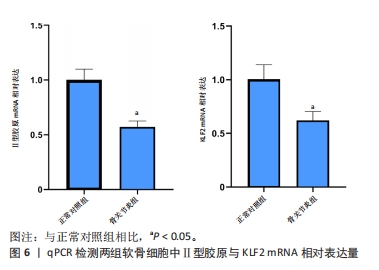
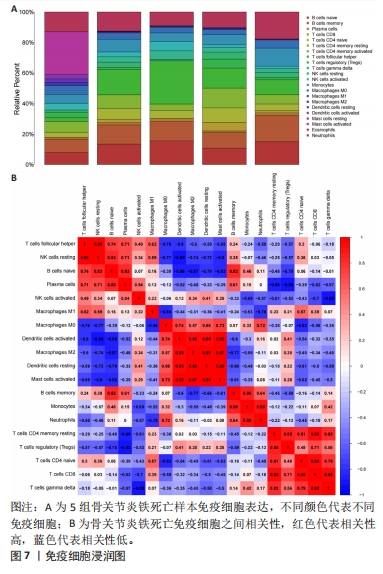
[5] 李哲,袁长深,官岩兵,等.骨关节炎中铁死亡的生物信息学分析与实验验证[J].中国组织工程研究,2023,27(17):2637-2643.
[6] CAMACHO DM, COLLINS KM, POWERS RK,et al. Next-Generation Machine Learning for Biological Networks. Cell. 2018;173(7):1581-1592.
[7] LIU W, LI L, YE H,et al. Weighted gene co-expression network analysis in biomedicine research. Sheng Wu Gong Cheng Xue Bao. 2017;33(11):1791-1801.
[8] WANG F, SU Q, LI C. Identification of cuproptosis-related asthma diagnostic genes by WGCNA analysis and machine learning. J Asthma. 2023;14:1-12.
[9] CHEN L, ZHANG YH, WANG S, et al. Prediction and analysis of essential genes using the enrichments of gene ontology and KEGG pathways. PLoS One. 2017; 12(9):0184129.
[10] KANEHISA M, FURUMICHI M, TANABE M, et al. KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017;45(1):353-361.
[11] LANGFELDER P, HORVATH S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008;9:559.
[12] CHEN X, ISHWARAN H. Random forests for genomic data analysis. Genomics. 2012;99(6):323-329.
[13] LI Z, SILLANPAA MJ. Overview of LASSO-related penalized regression methods for quantitative trait mapping and genomic selection. Theor Appl Genet. 2012; 125(3):419-435.
[14] GAO J, KWAN PW, SHI D. Sparse kernel learning with LASSO and Bayesian inference algorithm. Neural Netw. 2010;23(2):257-264.
[15] LIVAK KJ, SCHMITTGEN TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25(4):402-408.
[16] CHEN D, SHEN J, ZHAO W, et al. Osteoarthritis: toward a comprehensive understanding of pathological mechanism. Bone Res. 2017;5:16044.
[17] SACITHARAN PK. Ageing and Osteoarthritis. Subcell Biochem. 2019;91:123-159.
[18] HAWKER GA, KING LK. The Burden of Osteoarthritis in Older Adults. Clin Geriatr Med. 2022;38(2):181-192.
[19] OO WM. Prospects of Disease-Modifying Osteoarthritis Drugs. Clin Geriatr Med. 2022;38(2):397-432.
[20] YAO X, SUN K, YU S, et al. Chondrocyte ferroptosis contribute to the progressi on of osteoarthritis. J Orthop Translat. 2020;27:33-43.
[21] STOCKWELL BR, FRIEDMANN ANGELI JP, BBYIR H, et al. Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease. Cell. 2017; 171(2):273-285.
[22] JING X, LIN J, DU T, et al. Iron Overload Is Associated With Accelerated Progression of Osteoarthritis: The Role of DMT1 Mediated Iron Homeostasis. Front Cell Dev Biol. 2021;8:594509.
[23] JING X, DU T, LI T, et al.The detrimental effect of iron on OA chondrocytes: Importance of pro-inflammatory cytokines induced iron influx and oxidative stress. J Cell Mol Med. 2021;25(12):5671-5680.
[24] ZAHAN OM, SERBAN O, GHERMAN C, et al. The evaluation of oxidative stress in osteoarthritis. Med Pharm Rep. 2020;93(1):12-22.
[25] BONJOUR JP. Calcium and phosphate: a duet of ions playing for bone health. J Am Coll Nutr. 2011;30(5):438-448.
[26] NA HS, PARK JS, CHO KH, et al. Interleukin-1-Interleukin-17 Signaling Axis Induces Cartilage Destruction and Promotes Experimental Osteoarthritis. Front ImMunol. 2020;11:730.
[27] CAI P, JIANG T, LI B, et al. Comparison of rheumatoid arthritis (RA) and osteoarthritis (OA) based on microarray profiles of human joint fibroblast-like synoviocytes. Cell Biochem Funct. 2019;37(1):31-41.
[28] WITTNER J, SCHUH W. Krüppel-like Factor 2 (KLF2) in Immune Cell Migration. Vaccines (Basel). 2021;9(10):1171.
[29] KAWATA M, TERAMURA T, ORDOUKHANIAN P, et al. Krüppel-like factor-4 and Krüppel-like factor-2 are important regulators of joint tissue cells and protect against tissue destruction and inflammation in osteoarthritis. Ann Rheum Dis. 2022;9:221867.
[30] GAO X, JIANG S, DU Z, et al. KLF2 Protects against Osteoarthritis by Repressing Oxidative Response through Activation of Nrf2/ARE Signaling In Vitro and In Vivo. Oxid Med Cell Longev. 2019;19:8564681
[31] YUAN Y, TAN H, DAI P. Krüppel-Like Factor 2 Regulates Degradation of Type II Collagen by Suppressing the Expression of Matrix Metalloproteinase (MMP)-13. Cell Physiol Biochem. 2017;42(6):2159-2168.
[32] LAHA D, DEB M, DAS H. KLF2 (kruppel-like factor 2 [lung]) regulates osteoclastogenesis by modulating autophagy. Autophagy. 2019;15(12):2063-2075.
[33] HOU Z, WANG Z, TAO Y, et al. KLF2 regulates osteoblast differentiation by targeting of Runx2. Lab Invest. 2019;99(2):271-280.
[34] RAGIPOGLU D, DUDECK A, HAFFNER-LUNTZER M, et al. The Role of Mast Cells in Bone Metabolism and Bone Disorders. Front Immunol. 2020;11:163.
[35] GERMIC N, FRANGEZ Z, YOUSEFI S, et al. Regulation of the innate immune system by autophagy: monocytes, macrophages, dendritic cells and antigen presentation. Cell Death Differ. 2019;26(4):715-727.
[36] CAI W, LI H, ZHANG Y, et al. Identification of key biomarkers and immune infiltration in the synovial tissue of osteoarthritis by bioinformatics analysis. Peer J. 2020,8:8390.
[37] FERNANDES JC, MARTEL-PELLETIER J, PELLETIER JP. The role of cytokines in osteoarthritis pathophysiology. Biorheology. 2002;39:237-246.
|