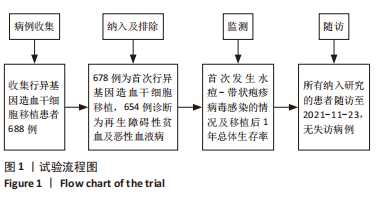
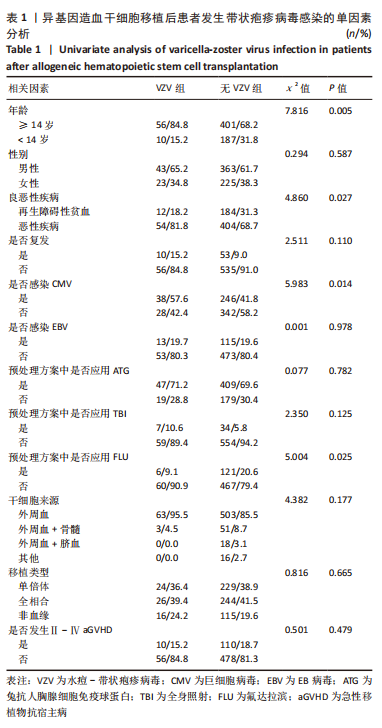
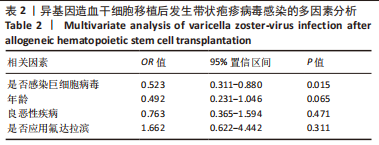
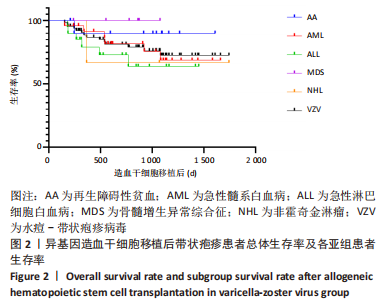
[1] 王昱,黄晓军.造血干细胞移植在血液疾病中的应用进展[J].中华血液学杂志,2019,40(8):704-708.
[2] SAHIN U, TOPRAK SK, ATILLA PA, et al. An overview of infectious complications after allogeneic hematopoietic stem cell transplantation. J Infect Chemother. 2016;22(8):505-514.
[3] HELDMAN MR, AAGAARD KM, HILL JA. Assessing and restoring adaptive immunity to HSV, VZV, and HHV-6 in solid organ and hematopoietic cell transplant recipients. Clin Microbiol Infect. 2022; 28(10):1345-1350.
[4] 魏伟,明英姿.浅谈实体器官移植后水痘-带状疱疹病毒感染[J].器官移植,2018,9(5):399-401.
[5] DÜVER F, WEIßBRICH B, EYRICH M, et al. Viral reactivations following hematopoietic stem cell transplantation in pediatric patients - A single center 11-year analysis. PLoS One. 2020;15(2):e0228451.
[6] 黄胜萍,廖烈兰,方润平.带状疱疹并发症的诊治与预防的研究进展[J].内科,2022,17(3):301-304.
[7] DADWAL SS. Herpes Virus Infections Other than Cytomegalovirus in the Recipients of Hematopoietic Stem Cell Transplantation. Infect Dis Clin North Am. 2019;33(2):467-484.
[8] LJUNGMAN P, LÖNNQVIST B, GAHRTON G, et al. Clinical and subclinical reactivations of varicella-zoster virus in immunocompromised patients. J Infect Dis. 1986;153(5):840-847.
[9] CAUDA R, GROSSI CE, WHITLEY RJ, et al. Analysis of immune function in herpes zoster patients: demonstration and characterization of suppressor cells. J Immunol. 1987;138(4):1229-1233.
[10] BOCCARD M, CONRAD A, MOUTON W, et al. A Simple-to-Perform ifn-γ mRNA Gene Expression Assay on Whole Blood Accurately Appraises Varicella Zoster Virus-Specific Cell-Mediated Immunity After Allogeneic Hematopoietic Stem Cell Transplantation. Front Immunol. 2022;13:919806.
[11] BAUMRIN E, CHENG MP, KANJILAL S, et al. Severe Herpes Zoster Requiring Intravenous Antiviral Treatment in Allogeneic Hematopoietic Cell Transplantation Recipients on Standard Acyclovir Prophylaxis. Biol Blood Marrow Transplant. 2019;25(8):1642-1647.
[12] KOC Y, MILLER KB, SCHENKEIN DP, et al. Varicella zoster virus infections following allogeneic bone marrow transplantation: frequency, risk factors, and clinical outcome. Biol Blood Marrow Transplant. 2000; 6(1):44-49.
[13] BUUS-GEHRIG C, BOCHENNEK K, HENNIES MT, et al. Systemic viral infection in children receiving chemotherapy for acute leukemia. Pediatr Blood Cancer. 2020;67(12):e28673.
[14] ERARD V, GUTHRIE KA, VARLEY C, et al. One-year acyclovir prophylaxis for preventing varicella-zoster virus disease after hematopoietic cell transplantation: no evidence of rebound varicella-zoster virus disease after drug discontinuation. Blood. 2007;110(8):3071-3077.
[15] SEO HM, KIM YS, BANG CH, et al. Antiviral prophylaxis for preventing herpes zoster in hematopoietic stem cell transplant recipients: A systematic review and meta-analysis. Antiviral Res. 2017;140:106-115.
[16] UMEZAWA Y, KAKIHANA K, OSHIKAWA G, et al. Clinical features and risk factors for developing varicella zoster virus dissemination following hematopoietic stem cell transplantation. Transpl Infect Dis. 2014;16(2):195-202.
[17] BAUMRIN E, IZAGUIRRE NE, BAUSK B, et al. Safety and reactogenicity of the recombinant zoster vaccine after allogeneic hematopoietic cell transplantation. Blood Adv. 2021;5(6):1585-1593.
[18] XUE E, XIE H, LEISENRING WM, et al. High Incidence of Herpes Zoster After Cord Blood Hematopoietic Cell Transplant Despite Longer Duration of Antiviral Prophylaxis. Clin Infect Dis. 2021;72(8):1350-1357.
[19] 沈悌,赵永强.血液病诊断及疗效标准[M].4版.北京:科学出版社,2018.
[20] 中华医学会血液学分会干细胞应用学组.中国异基因造血干细胞移植治疗血液系统疾病专家共识(Ⅲ)——急性移植物抗宿主病(2020年版)[J].中华血液学杂志,2020,41(7):529-536.
[21] SCHOEMANS HM, LEE SJ, FERRARA JL, et al. EBMT-NIH-CIBMTR Task. Force position statement on standardized terminology & guidance for graft-versus-host disease assessment. Bone Marrow Transplant. 2018; 53(11):1401-1415.
[22] CHUN JY, KIM K, LEE MK, et al. Immunogenicity and safety of a live herpes zoster vaccine in hematopoietic stem cell transplant recipients. BMC Infect Dis. 2021;21(1):117.
[23] STOREK J, DAWSON MA, STORER B, et al. Immune reconstitution after allogeneic marrow transplantation compared with blood stem cell transplantation. Blood. 2001;97(11):3380-3389.
[24] 郝新健,周健,房佰俊,等.异基因造血干细胞移植后带状疱疹的临床分析[J].中华实验和临床感染病杂志(电子版),2012,6(3):221-224.
[25] BEYAR-KATZ O, BITTERMAN R, ZUCKERMAN T, et al. Anti-herpesvirus prophylaxis, pre-emptive treatment or no treatment in adults undergoing allogeneic transplant for haematological disease: systematic review and meta-analysis. Clin Microbiol Infect. 2020;26(2):189-198.
[26] HAN SB, KIM SK, LEE JW, et al. Varicella zoster virus infection after allogeneic hematopoietic cell transplantation in children using a relatively short duration of acyclovir prophylaxis: A retrospective study. Medicine (Baltimore). 2017;96(14):e6546.
[27] ASANO-MORI Y, KANDA Y, OSHIMA K, et al. Long-term ultra-low-dose acyclovir against varicella–zoster virus reactivation after allogeneic hematopoietic stem cell transplantation. Am J Hematol 2008;83:472-476.
[28] MASCARENHAS K, TEH JB, PENG K, et al. Efficacy of low-dose zoster prophylaxis in patients undergoing allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2020;55(8):1662-1664.
[29] BLENNOW O, FJAERTOFT G, WINIARSKI J, et al. Varicella-zoster reactivation after allogeneic stem cell transplantation without routine prophylaxis--the incidence remains high. Biol Blood Marrow Transplant. 2014;20(10):1646-1649.
[30] LIZZI J, HILL T, JAKUBOWSKI J. Varicella Zoster Virus Encephalitis. Clin Pract Cases Emerg Med. 2019;3(4):380-382.
[31] OH JH, TUMMALA S, HUSNAIN MG. Disseminated herpes zoster with acute encephalitis in an immunocompetent elderly man. BMJ Case Rep. 2020;13(6):e232928.
[32] GRATAMA JW, WEILAND HT, HEKKER AC, et al. Herpes virus immunity and acute graft-versus-host disease. Transplant Proc. 1987;19(1 Pt 3): 2680-2682.
[33] ARVIN A, ABENDROTH A. VZV: immunobiology and host response//ARVIN A, CAMPADELLI-FIUME G, MOCARSKI E, et al. editors. Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis. Cambridge: Cambridge University Press. 2007:Chapter 39.
[34] KENNEDY PGE, GERSHON AA. Clinical Features of Varicella-Zoster Virus Infection. Viruses. 2018;10(11):609.
[35] LEWIS DJ, SCHLICHTE MJ, DAO H JR. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100(5):321,324,330.
[36] 宋微,马良娟.移植与带状疱疹相关性新进展[J].中国麻风皮肤病杂志,2021,37(4):260-262.
[37] KANG M, ASLAM S. Varicella zoster virus encephalitis in solid organ transplant recipients: Case series and review of literature. Transpl Infect Dis. 2019;21(2):e13038.
[38] NABI S, KAHLON P, GOGGINS M, et al. VZV encephalitis following successful treatment of CMV infection in a patient with kidney transplant. BMJ Case Rep. 2014;2014:bcr2014206655.
[39] 王卓,何岳林,廖建云等.异基因造血干细胞移植术后患儿带状疱疹病毒感染的临床研究[J].中华实用儿科临床杂志,2019,34(12): 930-933.
[40] DOKI N, MIYAWAKI S, TANAKA M, et al. Visceral varicella zoster virus infection after allogeneic stem cell transplantation.Transpl Infect Dis. 2013;15(3):314-318.
|