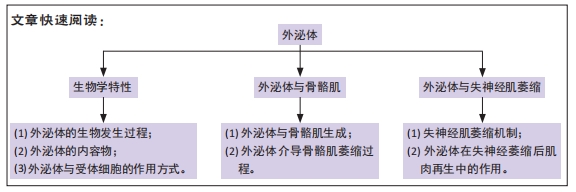
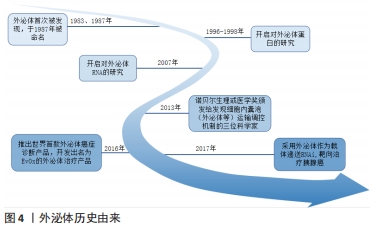
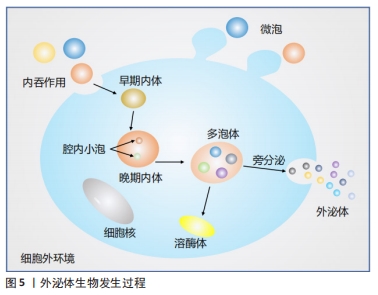
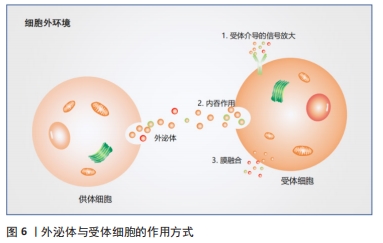
[1] NEUBRECH F, HEIDER S, HARHAUS L, et al. Chitosan nerve tube for primary repair of traumatic sensory nerve lesions of the hand without a gap: study protocol for a randomized controlled trial. Trials. 2016;17:48.
[2] ROSÉN B, VIKSTRÖM P, TURNER S, et al. Enhanced early sensory outcome after nerve repair as a result of immediate post-operative re-learning: a randomized controlled trial. J Hand Surg Eur Vol. 2015; 40(6):598-606.
[3] 钟声,章明星,刘建卫,等.电针治疗失神经肌萎缩机制研究进展[J].中国中医药现代远程教育,2018,16(19):146-147.
[4] TIELAND M, TROUWBORST I, CLARK BC. Skeletal muscle performance and ageing. J Cachexia Sarcopenia Muscle. 2018;9(1):3-19.
[5] VIJAYAVENKATARAMAN S. Nerve guide conduits for peripheral nerve injury repair: A review on design, materials and fabrication methods. Acta Biomater. 2020;106:54-69.
[6] 杨东丽,杨琼,罗嘉,等.外泌体参与骨骼肌机能调控研究进展[J].动物医学进展,2018,39(1):103-108.
[7] ZHANG Y, CHOPP M, LIU XS, et al. Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons. Mol Neurobiol. 2017;54(4):2659-2673.
[8] HE C, ZHENG S, LUO Y, et al. Exosome Theranostics: Biology and Translational Medicine. Theranostics. 2018;8(1):237-255.
[9] HARDING C, HEUSER J, STAHL P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329-339.
[10] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977.
[11] HENNE WM, STENMARK H, EMR SD. Molecular mechanisms of the membrane sculpting ESCRT pathway. Cold Spring Harb Perspect Biol. 2013;5(9):a016766.
[12] STUFFERS S, SEM WEGNER C, STENMARK H, et al. Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic. 2009;10(7): 925-937.
[13] HESSVIK NP, ØVERBYE A, BRECH A, et al. PIKfyve inhibition increases exosome release and induces secretory autophagy. Cell Mol Life Sci. 2016;73(24):4717-4737.
[14] XU J, CAMFIELD R, GORSKI SM. The interplay between exosomes and autophagy - partners in crime. J Cell Sci. 2018;131(15):jcs215210.
[15] KORKUT C, ATAMAN B, RAMACHANDRAN P, et al. Trans-synaptic transmission of vesicular Wnt signals through Evi/Wntless. Cell. 2009; 139(2):393-404.
[16] ANEL A, GALLEGO-LLEYDA A, DE MIGUEL D, et al. Role of Exosomes in the Regulation of T-cell Mediated Immune Responses and in Autoimmune Disease. Cells. 2019;8(2):154.
[17] ZHAO J, LI X, HU J, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019;115(7):1205-1216.
[18] WANG X, ZHOU Y, GAO Q, et al. The Role of Exosomal microRNAs and Oxidative Stress in Neurodegenerative Diseases. Oxid Med Cell Longev. 2020;2020:3232869.
[19] ZHANG L, YU D. Exosomes in cancer development, metastasis, and immunity. Biochim Biophys Acta Rev Cancer. 2019;1871(2):455-468.
[20] YANG L, PENG X, LI Y, et al. Long non-coding RNA HOTAIR promotes exosome secretion by regulating RAB35 and SNAP23 in hepatocellular carcinoma. Mol Cancer. 2019;18(1):78.
[21] PEGTEL DM, GOULD SJ. Exosomes. Annu Rev Biochem. 2019;88:487-514.
[22] SCHNEIDER A, SIMONS M. Exosomes: vesicular carriers for intercellular communication in neurodegenerative disorders. Cell Tissue Res. 2013; 352(1):33-47.
[23] LARIOS J, MERCIER V, ROUX A, et al. ALIX- and ESCRT-III-dependent sorting of tetraspanins to exosomes. J Cell Biol. 2020;219(3): e201904113.
[24] WEI D, ZHAN W, GAO Y, et al. RAB31 marks and controls an ESCRT-independent exosome pathway. Cell Res. 2021;31(2):157-177.
[25] VALADI H, EKSTRÖM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-659.
[26] GIBBINGS DJ, CIAUDO C, ERHARDT M, et al. Multivesicular bodies associate with components of miRNA effector complexes and modulate miRNA activity. Nat Cell Biol. 2009;11(9):1143-1149.
[27] BATAGOV AO, KUROCHKIN IV. Exosomes secreted by human cells transport largely mRNA fragments that are enriched in the 3’-untranslated regions. Biol Direct. 2013;8:12.
[28] VILLARROYA-BELTRI C, GUTIÉRREZ-VÁZQUEZ C, SÁNCHEZ-CABO F, et al. Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat Commun. 2013;4:2980.
[29] SANTANGELO L, GIURATO G, CICCHINI C, et al. The RNA-Binding Protein SYNCRIP Is a Component of the Hepatocyte Exosomal Machinery Controlling MicroRNA Sorting. Cell Rep. 2016;17(3):799-808.
[30] TRAN N. Cancer Exosomes as miRNA Factories. Trends Cancer. 2016; 2(7):329-331.
[31] SKOTLAND T, HESSVIK NP, SANDVIG K, et al. Exosomal lipid composition and the role of ether lipids and phosphoinositides in exosome biology. J Lipid Res. 2019;60(1):9-18.
[32] OUWENEEL AB, THOMAS MJ, SORCI-THOMAS MG. The ins and outs of lipid rafts: functions in intracellular cholesterol homeostasis, microparticles, and cell membranes: Thematic Review Series: Biology of Lipid Rafts. J Lipid Res. 2020;61(5):676-686.
[33] PETERSEN SV, POULSEN NB, LINNEBERG MATTHIESEN C, et al. Novel and Converging Ways of NOX2 and SOD3 in Trafficking and Redox Signaling in Macrophages. Antioxidants (Basel). 2021;10(2):172.
[34] BADAWY SMM, OKADA T, KAJIMOTO T, et al. Extracellular α-synuclein drives sphingosine 1-phosphate receptor subtype 1 out of lipid rafts, leading to impaired inhibitory G-protein signaling. J Biol Chem. 2018;293(21):8208-8216.
[35] ZHANG B, WANG M, GONG A, et al. HucMSC-Exosome Mediated-Wnt4 Signaling Is Required for Cutaneous Wound Healing. Stem Cells. 2015;33(7):2158-2168.
[36] COSSETTI C, IRACI N, MERCER TR, et al. Extracellular vesicles from neural stem cells transfer IFN-γ via Ifngr1 to activate Stat1 signaling in target cells. Mol Cell. 2014;56(2):193-204.
[37] LÄSSER C. Exosomes in diagnostic and therapeutic applications: biomarker, vaccine and RNA interference delivery vehicle. Expert Opin Biol Ther. 2015;15(1):103-117.
[38] VALCHEVA-KUZMANOVA S, ZHELYAZKOVA-SAVOVA M. Anxiolytic-like effect of Aronia melanocarpa fruit juice in rats. Methods Find Exp Clin Pharmacol. 2009;31(10):651-654.
[39] YU B, KIM HW, GONG M, et al. Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol. 2015;182:349-360.
[40] CHRISTIANSON HC, SVENSSON KJ, VAN KUPPEVELT TH, et al. Cancer cell exosomes depend on cell-surface heparan sulfate proteoglycans for their internalization and functional activity. Proc Natl Acad Sci U S A. 2013;110(43):17380-17385.
[41] CAPONNETTO F, MANINI I, SKRAP M, et al. Size-dependent cellular uptake of exosomes. Nanomedicine. 2017;13(3):1011-1020.
[42] ROME S, FORTERRE A, MIZGIER ML, et al. Skeletal Muscle-Released Extracellular Vesicles: State of the Art. Front Physiol. 2019;10:929.
[43] GUESCINI M, GUIDOLIN D, VALLORANI L, et al. C2C12 myoblasts release micro-vesicles containing mtDNA and proteins involved in signal transduction. Exp Cell Res. 2010;316(12):1977-1984.
[44] ASWAD H, FORTERRE A, WIKLANDER OP, et al. Exosomes participate in the alteration of muscle homeostasis during lipid-induced insulin resistance in mice. Diabetologia. 2014;57(10):2155-2164.
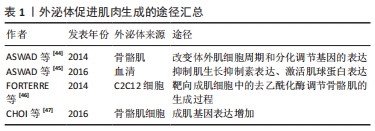
[45] ASWAD H, JALABERT A, ROME S. Depleting extracellular vesicles from fetal bovine serum alters proliferation and differentiation of skeletal muscle cells in vitro. BMC Biotechnol. 2016;16:32.
[46] FORTERRE A, JALABERT A, CHIKH K, et al. Myotube-derived exosomal miRNAs downregulate Sirtuin1 in myoblasts during muscle cell differentiation. Cell Cycle. 2014;13(1):78-89.
[47] CHOI JS, YOON HI, LEE KS, et al. Exosomes from differentiating human skeletal muscle cells trigger myogenesis of stem cells and provide biochemical cues for skeletal muscle regeneration. J Control Release. 2016;222:107-115.
[48] NIE Y, SATO Y, GARNER RT, et al. Skeletal muscle-derived exosomes regulate endothelial cell functions via reactive oxygen species-activated nuclear factor-κB signalling. Exp Physiol. 2019;104(8):1262-1273.
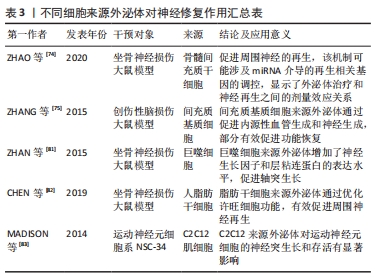
[49] MADISON RD, MCGEE C, RAWSON R, et al. Extracellular vesicles from a muscle cell line (C2C12) enhance cell survival and neurite outgrowth of a motor neuron cell line (NSC-34). J Extracell Vesicles. 2014;3(1): 22865.
[50] CID-DÍAZ T, SANTOS-ZAS I, GONZÁLEZ-SÁNCHEZ J, et al. Obestatin controls the ubiquitin-proteasome and autophagy-lysosome systems in glucocorticoid-induced muscle cell atrophy. J Cachexia Sarcopenia Muscle. 2017;8(6):974-990.
[51] LI S, HAO M, LI B, et al. CACNA1H downregulation induces skeletal muscle atrophy involving endoplasmic reticulum stress activation and autophagy flux blockade. Cell Death Dis. 2020;11(4):279.
[52] WANG M, HU R, WANG Y, et al. Atractylenolide III Attenuates Muscle Wasting in Chronic Kidney Disease via the Oxidative Stress-Mediated PI3K/AKT/mTOR Pathway. Oxid Med Cell Longev. 2019;2019:1875471.
[53] O’NEILL BT, BHARDWAJ G, PENNIMAN CM, et al. FoxO Transcription Factors Are Critical Regulators of Diabetes-Related Muscle Atrophy. Diabetes. 2019;68(3):556-570.
[54] HUDSON MB, RAHNERT JA, ZHENG B, et al. miR-182 attenuates atrophy-related gene expression by targeting FoxO3 in skeletal muscle. Am J Physiol Cell Physiol. 2014;307(4):C314-C319.
[55] HUDSON MB, WOODWORTH-HOBBS ME, ZHENG B, et al. miR-23a is decreased during muscle atrophy by a mechanism that includes calcineurin signaling and exosome-mediated export. Am J Physiol Cell Physiol. 2014;306(6):C551-C558.
[56] MIAO C, ZHANG W, FENG L, et al. Cancer-derived exosome miRNAs induce skeletal muscle wasting by Bcl-2-mediated apoptosis in colon cancer cachexia. Mol Ther Nucleic Acids. 2021;24:923-938.
[57] LI C, WU Q, LI Z, et al. Exosomal microRNAs in cancer-related sarcopenia: Tumor-derived exosomal microRNAs in muscle atrophy. Exp Biol Med (Maywood). 2021;246(10):1156-1166.
[58] COENEN-STASS AML, WOOD MJA, ROBERTS TC. Biomarker Potential of Extracellular miRNAs in Duchenne Muscular Dystrophy. Trends Mol Med. 2017;23(11):989-1001.
[59] CISTERNA BA, CARDOZO C, SÁEZ JC. Neuronal involvement in muscular atrophy. Front Cell Neurosci. 2014;8:405.
[60] PAUDYAL A, SLEVIN M, MAAS H, et al. Time course of denervation-induced changes in gastrocnemius muscles of adult and old rats. Exp Gerontol. 2018;106:165-172.
[61] SIU PM, ALWAY SE. Mitochondria-associated apoptotic signalling in denervated rat skeletal muscle. J Physiol. 2005;565(Pt 1):309-323.
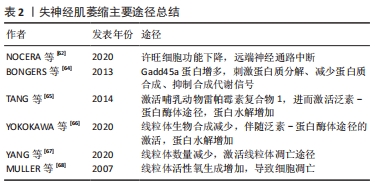
[62] NOCERA G, JACOB C. Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol Life Sci. 2020;77(20): 3977-3989.
[63] SACHECK JM, HYATT JP, RAFFAELLO A, et al. Rapid disuse and denervation atrophy involve transcriptional changes similar to those of muscle wasting during systemic diseases. FASEB J. 2007;21(1):140-155.
[64] BONGERS KS, FOX DK, EBERT SM, et al. Skeletal muscle denervation causes skeletal muscle atrophy through a pathway that involves both Gadd45a and HDAC4. Am J Physiol Endocrinol Metab. 2013;305(7): E907-915.
[65] TANG H, INOKI K, LEE M, et al. mTORC1 promotes denervation-induced muscle atrophy through a mechanism involving the activation of FoxO and E3 ubiquitin ligases. Sci Signal. 2014;7(314):ra18.
[66] YOKOKAWA T, MORI R, SUGA T, et al. Muscle denervation reduces mitochondrial biogenesis and mitochondrial translation factor expression in mice. Biochem Biophys Res Commun. 2020;527(1):146-152.
[67] YANG X, XUE P, CHEN H, et al. Denervation drives skeletal muscle atrophy and induces mitochondrial dysfunction, mitophagy and apoptosis via miR-142a-5p/MFN1 axis. Theranostics. 2020;10(3):1415-1432.
[68] MULLER FL, SONG W, JANG YC, et al. Denervation-induced skeletal muscle atrophy is associated with increased mitochondrial ROS production. Am J Physiol Regul Integr Comp Physiol. 2007;293(3): R1159-R1168.
[69] ADHIHETTY PJ, O’LEARY MF, CHABI B, et al. Effect of denervation on mitochondrially mediated apoptosis in skeletal muscle. J Appl Physiol (1985). 2007;102(3):1143-1151.
[70] SCHAAKXS D, KALBERMATTEN DF, RAFFOUL W, et al. Regenerative cell injection in denervated muscle reduces atrophy and enhances recovery following nerve repair. Muscle Nerve. 2013;47(5):691-701.
[71] 刘晓光,肖卫华,赵淋淋,等.间充质干细胞移植治疗骨骼肌损伤的研究进展[J].中国康复医学杂志,2015,30(12):1313-1317.
[72] JOANISSE S, NEDERVEEN JP, SNIJDERS T, et al. Skeletal Muscle Regeneration, Repair and Remodelling in Aging: The Importance of Muscle Stem Cells and Vascularization. Gerontology. 2017;63(1):91-100.
[73] PHINNEY DG, PITTENGER MF. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells. 2017;35(4):851-858.
[74] ZHAO J, DING Y, HE R, et al. Dose-effect relationship and molecular mechanism by which BMSC-derived exosomes promote peripheral nerve regeneration after crush injury. Stem Cell Res Ther. 2020;11(1): 360.
[75] ZHANG Y, CHOPP M, MENG Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;122(4):856-867.
[76] WEN D, PENG Y, LIU D, et al. Mesenchymal stem cell and derived exosome as small RNA carrier and Immunomodulator to improve islet transplantation. J Control Release. 2016;238:166-175.
[77] SHABBIR A, COX A, RODRIGUEZ-MENOCAL L, et al. Mesenchymal Stem Cell Exosomes Induce Proliferation and Migration of Normal and Chronic Wound Fibroblasts, and Enhance Angiogenesis In Vitro. Stem Cells Dev. 2015;24(14):1635-1647.
[78] XU JF, YANG GH, PAN XH, et al. Altered microRNA expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS One. 2014;9(12):e114627.
[79] SHAO L, ZHANG Y, LAN B, et al. MiRNA-Sequence Indicates That Mesenchymal Stem Cells and Exosomes Have Similar Mechanism to Enhance Cardiac Repair. Biomed Res Int. 2017;2017:4150705.
[80] FURUTA T, MIYAKI S, ISHITOBI H, et al. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem Cells Transl Med. 2016;5(12):1620-1630.
[81] ZHAN C, MA CB, YUAN HM, et al. Macrophage-derived microvesicles promote proliferation and migration of Schwann cell on peripheral nerve repair. Biochem Biophys Res Commun. 2015;468(1-2):343-348.
[82] CHEN J, REN S, DUSCHER D, et al. Exosomes from human adipose-derived stem cells promote sciatic nerve regeneration via optimizing Schwann cell function. J Cell Physiol. 2019;234(12):23097-23110.
[83] MADISON RD, MCGEE C, RAWSON R, et al. Extracellular vesicles from a muscle cell line (C2C12) enhance cell survival and neurite outgrowth of a motor neuron cell line (NSC-34). J Extracell Vesicles. 2014;3.
[84] DE GASPERI R, HAMIDI S, HARLOW LM, et al. Denervation-related alterations and biological activity of miRNAs contained in exosomes released by skeletal muscle fibers. Sci Rep. 2017;7(1):12888.
[85] HUANG QK, QIAO HY, FU MH, et al. MiR-206 Attenuates Denervation-Induced Skeletal Muscle Atrophy in Rats Through Regulation of Satellite Cell Differentiation via TGF-β1, Smad3, and HDAC4 Signaling. Med Sci Monit. 2016;22:1161-1170.
|