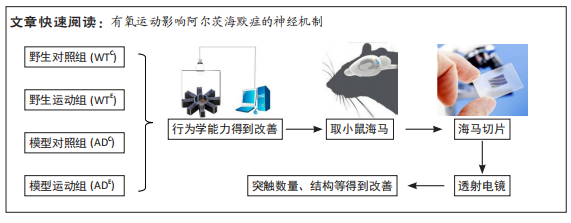
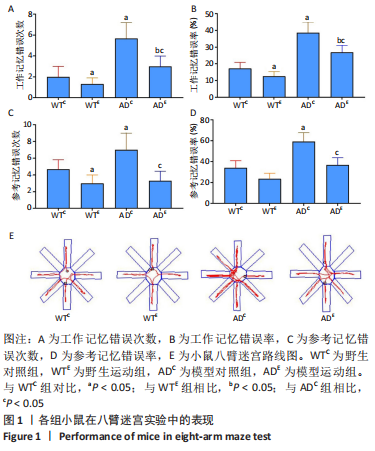
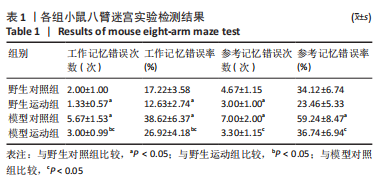
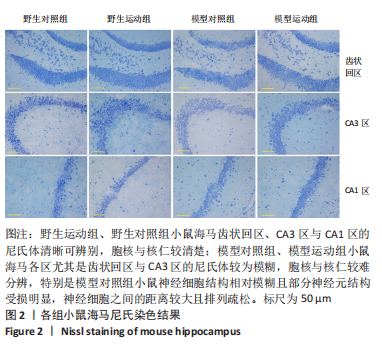
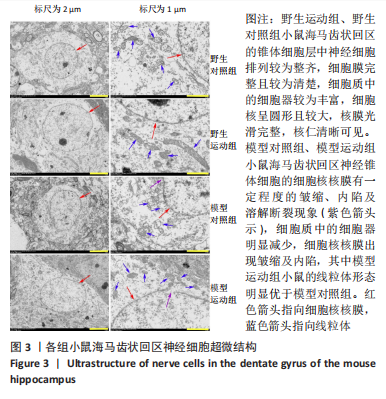
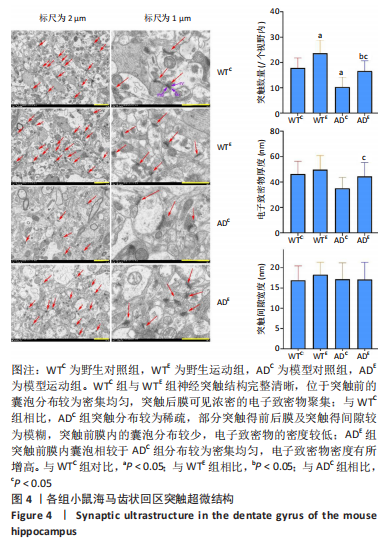
[1] MARTIN P, ANDERS W. World Alzheimer Report 2016. Alzheimer’s Disease International, 2016.
[2] BROOKMEYER R, JOHNSON E, ZIEGLER-GRAHAM K, et al. Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement. 2007; 3(3):186-191.
[3] 赵祥,魏翠兰,张业廷.神经发生和炎性环境在运动条件下的改变与调节[J].中国组织工程研究,2021,25(5):813-820.
[4] BITTNER T, BURGOLD S, DOROSTKAR MM, et al. Amyloid plaque formation precedes dendritic spine loss. Acta Neuropathol. 2012; 124(6):797-807.
[5] TOBIN MK, MUSARACA K, DISOUKY A, et al. Human Hippocampal Neurogenesis Persists in Aged Adults and Alzheimer’s Disease Patients. Cell Stem Cell. 2019;24(6):974-978.
[6] LAUTENSCHLAGER N, COX K, ELLIS K. Physical activity for cognitive health: what advice can we give to older adults with subjective cognitive decline and mild cognitive impairment? Dialogues Clin Neuro. 2019;21(1):61-68.
[7] 张象,张业廷.运动改善阿尔茨海默症模型小鼠病程的剂量效应关系[J].中国组织工程研究,2021,25(17):2761-2766.
[8] KURZINA NP, ARISTOVA IY, VOLNOVA AB, et al. Deficit in working memory and abnormal behavioral tactics in dopamine transporter knockout rats during training in the 8-arm maze. Behav Brain Res. 2020; 390:112642.
[9] MCKHANN GM, KNOPMAN DS, CHERTKOW H, et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):263-269.
[10] WAKHLOO D, OBERHAUSER J, MADIRA A, et al. From cradle to grave: neurogenesis, neuroregeneration and neurodegeneration in Alzheimer’s and Parkinson’s diseases. Neural Regen Res. 2022;17(12): 2606-2614.
[11] VERMUNT L, SIKKES SAM, VAN DEN HOUT A, et al. Duration of preclinical, prodromal, and dementia stages of Alzheimer’s disease in relation to age, sex, and APOE genotype. Alzheimers Dement. 2019; 15(7):888-898.
[12] HANSEN RA, GARTLEHNER G, WEBB AP, et al. Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: A systematic review and meta-analysis. Clin Interv Aging. 2008;3(2):211-225.
[13] MONTGOMERY W, GOREN A, KAHLE-WROBLESKI K, et al. Detection, diagnosis, and treatment of Alzheimer’s disease dementia stratified by severity as reported by caregivers in Japan. Neuropsych Dis Treat. 2018;14:1843-1854.
[14] PARANJPE MD, WANG JK, ZHOU Y. Sex, ApoE4 and Alzheimer’s disease: rethinking drug discovery in the era of precision medicine. Neural Regen Res. 2021;16(9):1764-1765.
[15]HOFFMANN K, SOBOL NA, FREDERIKSEN KS, et al.Moderate-to-High Intensity Physical Exercise in Patients with Alzheimer’s Disease: A Randomized Controlled Trial. J Alzheimers Dis. 2016;50(2):443-453.
[16] KE HC, HUANG HJ, LIANG KC, et al. Selective improvement of cognitive function in adult and aged APP/PS1 transgenic mice by continuous non-shock treadmill exercise. Brain Res. 2011;1403:1-11.
[17] LIU HL, ZHAO G, ZHANG H, et al. Long-term treadmill exercise inhibits the progression of Alzheimer’s disease-like neuropathology in the hippocampus of APP/PS1 transgenic mice. Behav Brain Res. 2013;256:261-272.
[18] SCARMEAS N, LUCHSINGER JA, SCHUPF N, et al. Physical Activity, Diet, and Risk of Alzheimer Disease. JAMA. 2009;302(6):627-637.
[19] SMITH JC, NIELSON KA, WOODARD JL, et al. Physical activity and brain function in older adults at increased risk for Alzheimer’s disease. Brain Sci. 2013;3(1):54-83.
[20] LAUTENSCHLAGER NT, COX KL, FLICKER L, et al. Effect of physical activity on cognitive function in older adults at risk for Alzheimer disease: a randomized trial. JAMA. 2008 ;300(9):1027-1037.
[21] ETGEN T, SANDER D, HUNTGEBURTH U, et al. Physical Activity and Incident Cognitive Impairment in Elderly Persons The INVADE Study. Arch Intern Med. 2010;170(2):186-190.
[22] KNOBLAUCH A, KÖRNER E, KÖRNER U, et al. Structural synaptic plasticity has high memory capacity and can explain graded amnesia, catastrophic forgetting, and the spacing effect. PloS One. 2014;9(5): e96485.
[23] ALONSO-NANCLARES L, MERINO-SERRAIS P, GONZALEZ S, et al. Synaptic changes in the dentate gyrus of APP/PS1 transgenic mice revealed by electron microscopy. J Neuropath Exp Neur. 2013;72(5): 386-395.
[24] SHENG C, XU P, ZHOU K, et al.βIcariin Attenuates Synaptic and Cognitive Deficits in an A-Induced Rat Model of Alzheimer’s Disease. Biomed Res Int. 2017;2017:7464872.
[25] YANG X, YAO C, TIAN T, et al. A novel mechanism of memory loss in Alzheimer’s disease mice via the degeneration of entorhinal-CA1 synapse. Mol Psychiatry. 2018;23(2):199-210.
[26] GROSSBERG S. Acetylcholine Neuromodulation in Normal and Abnormal Learning and Memory: Vigilance Control in Waking, Sleep, Autism, Amnesia and Alzheimer’s Disease. Front Neural Circuits. 2017;11:82.
[27] LI X, GUAN J, SUN T, et al. Circadian learning and memory changes in Abeta1-42 induced Alzheimer’s mice. Metab Brain Dis. 2020;35(3):463-471.
[28] SAEEDI M, RASHIDY-POUR A. Association between chronic stress and Alzheimer’s disease: Therapeutic effects of Saffron. Biomed Pharmacother. 2021;133:110995. doi: 10.1016/j.biopha.2020.110995.
[29] GÓMEZ-GALLEGO M, GÓMEZ-GARCÍA J. Stress and verbal memory in patients with Alzheimer’s disease: different role of cortisol and anxiety. Aging Ment Health. 2019;23(11):1496-1502.
[30] MORENO-JIMENEZ EP, FLOR-GARCIA M, TERREROS-RONCAL J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25(4):554.
[31] CHEN X, WU X, WU H, et al. Phase separation at the synapse. Nat Neurosci. 2020;23(3):301-310.
[32] KENNEDY M. The Protein Biochemistry of the Postsynaptic Density in Glutamatergic Synapses Mediates Learning in Neural Networks. Biochemistry. 2018;57(27):4005-4009.
[33] ZENG M, CHEN X, GUAN D, et al. Reconstituted Postsynaptic Density as a Molecular Platform for Understanding Synapse Formation and Plasticity. Cell. 2018;174(5):1172-1187.e16.
[34] ZENG M, SHANG Y, ARAKI Y, et al. Phase Transition in Postsynaptic Densities Underlies Formation of Synaptic Complexes and Synaptic Plasticity. Cell. 2016;166(5):1163-1175.e12.
[35] BUONAGURO EF, MORLEY-FLETCHER S, AVAGLIANO C, et al. Glutamatergic postsynaptic density in early life stress programming: Topographic gene expression of mGlu5 receptors and Homer proteins. Prog Neuropsychopharmacol Biol Psychiatry. 2020;96:109725.
[36] SCHAEFER ML, PEREZ PJ, WANG M, et al. Neonatal Isoflurane Anesthesia or Disruption of Postsynaptic Density-95 Protein Interactions Change Dendritic Spine Densities and Cognitive Function in Juvenile Mice. Anesthesiology. 2020;133(4):812-823.
[37] DE BARTOLOMEIS A, AVAGLIANO C, VELLUCCI L, et al. Translating preclinical findings in clinically relevant new antipsychotic targets: focus on the glutamatergic postsynaptic density. Implications for treatment resistant schizophrenia. Neurosci Biobehav Rev. 2019;107:795-827. |