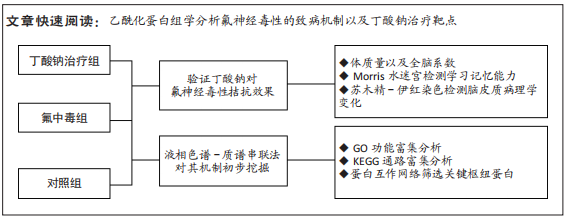
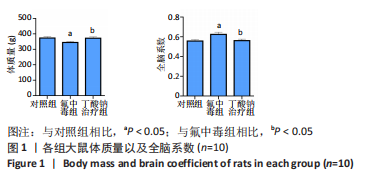
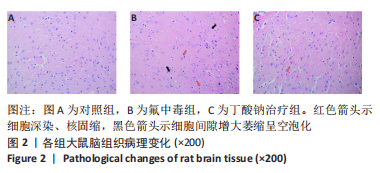
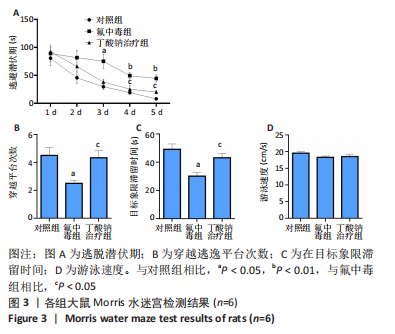
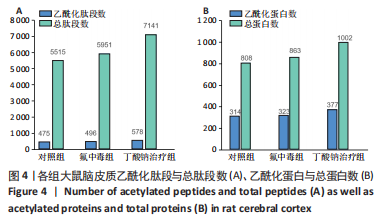
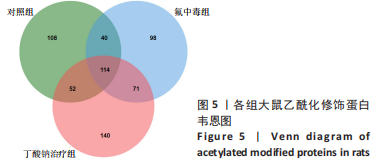
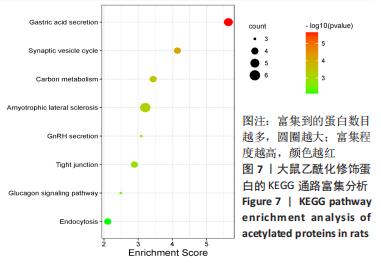
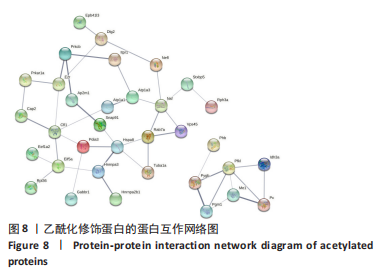
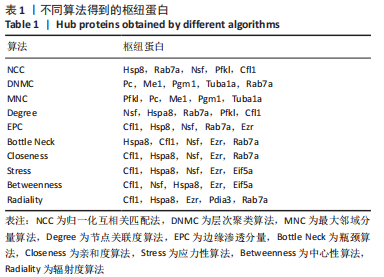
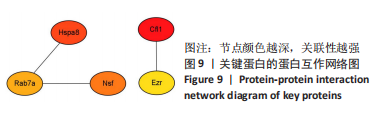
[1] SAEED M, MALIK RN, KAMAL A. Fluorosis and cognitive development among children (6-14 years of age) in the endemic areas of the world: a review and critical analysis. Environmental science and pollution research international. 2020;27(3):2566-2579.
[2] KERDOUN MA, MEKHLOUFI S, ADJAINE OEK, et al. Fluoride concentrations in drinking water and health risk assessment in the south of Algeria. Regul Toxicol Pharmacol. 2022;128:105086.
[3] NING H, LI C, YIN Z, et al. Fluoride exposure decreased neurite formation on cerebral cortical neurons of SD rats in vitro. Environ Sci Pollut Res Int. 2021;28(37):50975-50982.
[4] REN C, ZHANG P, YAO XY, et al. The cognitive impairment and risk factors of the older people living in high fluorosis areas: DKK1 need attention. BMC Public Health. 2021;21(1):2237.
[5] YANI SI, SEWENG A, MALLONGI A, et al. The influence of fluoride in drinking water on the incidence of fluorosis and intelligence of elementary school students in Palu City. Gac Sanit. 2021;35 Suppl 2: S159-S163.
[6] GUPTA R, AMBASTA RK, KUMAR P. Histone deacetylase in neuropathology. Adv Clin Chem. 2021;104:151-231.
[7] DAI Y, WEI T, SHEN Z, et al. Classical HDACs in the regulation of neuroinflammation. Neurochem Int. 2021;150:105182.
[8] PAO PC, TSAI LH. Histone Deacetylases 1 and 2 in Memory Function. ACS Chem Neurosci. 2022;13(7):848-858.
[9] YU Z, HAN J, CHEN H, et al. Oral Supplementation With Butyrate Improves Myocardial Ischemia/Reperfusion Injury via a Gut-Brain Neural Circuit. Front Cardiovasc Med. 2021;8:718674.
[10] WANG C, ZHENG D, WENG F, et al. Sodium butyrate ameliorates the cognitive impairment of Alzheimer’s disease by regulating the metabolism of astrocytes. Psychopharmacology (Berl). 2022;239(1): 215-227.
[11] LI X, FAN X, YUAN X, et al. The Role of Butyric Acid in Treatment Response in Drug-Naïve First Episode Schizophrenia. Front Psychiatry. 2021;12:724664.
[12] GAO Y, YA B, LI X, et al. Myricitrin ameliorates cognitive deficits in MCAO cerebral stroke rats via histone acetylation-induced alterations of brain-derived neurotrophic factor. Mol Cell Biochem. 2021;476(2): 609-617.
[13] JAWORSKA J, ZALEWSKA T, SYPECKA J, et al. Effect of the HDAC Inhibitor, Sodium Butyrate, on Neurogenesis in a Rat Model of Neonatal Hypoxia-Ischemia: Potential Mechanism of Action. Mol Neurobiol. 2019;56(9):6341-6370.
[14] 王正东,黄娜,陈婧娴,等.丁酸钠抑制氟中毒可诱导小胶质细胞活化及炎症因子表达增多[J]. 中国组织工程研究,2021,25(7):1075-1080.
[15] JIANG Y, LI K, LI X, et al. Sodium butyrate ameliorates the impairment of synaptic plasticity by inhibiting the neuroinflammation in 5XFAD mice. Chem Biol Interact. 2021;341:109452.
[16] STILLING RM, VAN DE WOUW M, CLARKE G, et al. The neuropharmacology of butyrate: The bread and butter of the microbiota-gut-brain axis? Neurochem Int. 2016;99:110-132.
[17] CODA DM, GRÄFF J. Neurogenetic and Neuroepigenetic Mechanisms in Cognitive Health and Disease. Front Mol Neurosci. 2020;13:205.
[18] ZHANG H, ELEFANT F. Exploring the Alzheimer’s disease neuroepigenome: recent advances and future trends. Neural Regen Res. 2022;17(2):325-327.
[19] CAMPBELL RR, WOOD MA. How the epigenome integrates information and reshapes the synapse. Nat Rev Neurosci. 2019;20(3):133-147.
[20] BURNS AM, GRÄFF J. Cognitive epigenetic priming: leveraging histone acetylation for memory amelioration. Curr Opin Neurobiol. 2021;67:75-84.
[21] HECKLAU K, MUELLER S, KOCH SP, et al. The Effects of Selective Inhibition of Histone Deacetylase 1 and 3 in Huntington’s Disease Mice. Front Mol Neurosci. 2021;14:616886.
[22] BI N, GU X, FAN A, et al. Bisphenol-A exposure leads to neurotoxicity through upregulating the expression of histone deacetylase 2 in vivo and in vitro. Toxicology. 2022;465:153052.
[23] JEONG H, THEN F, MELIA TJ, et al. Acetylation targets mutant huntingtin to autophagosomes for degradation. Cell. 2009;137(1):60-72.
[24] KUMAR V, KUNDU S, SINGH A, et al. Understanding the Role of Histone Deacetylase and their Inhibitors in Neurodegenerative Disorders: Current Targets and Future Perspective. Curr Neuropharmacol. 2022; 20(1):158-178.
[25] KITAHARA M, INOUE T, MANI H, et al. Exercise and pharmacological inhibition of histone deacetylase improves cognitive function accompanied by an increase of gene expressions crucial for neuronal plasticity in the hippocampus. Neurosci Lett. 2021;749:135749.
[26] JIANG P, LI G, ZHOU X, et al. Chronic fluoride exposure induces neuronal apoptosis and impairs neurogenesis and synaptic plasticity: Role of GSK-3β/β-catenin pathway. Chemosphere. 2019;214:430-435.
[27] MAITY S, FARRELL K, NAVABPOUR S, et al. Epigenetic Mechanisms in Memory and Cognitive Decline Associated with Aging and Alzheimer’s Disease. Int J Mol Sci. 2021;22(22):12280.
[28] LEVENSON JM, O’RIORDAN KJ, BROWN KD, et al. Regulation of histone acetylation during memory formation in the hippocampus. J Biol Chem. 2004;279(39):40545-40559.
[29] GANGULY A, HAN X, DAS U, et al. Hsc70 chaperone activity is required for the cytosolic slow axonal transport of synapsin. J Cell Biol. 2017; 216(7):2059-2074.
[30] PARRA LA, BAUST TB, SMITH AD, et al. The Molecular Chaperone Hsc70 Interacts with Tyrosine Hydroxylase to Regulate Enzyme Activity and Synaptic Vesicle Localization. J Biol Chem. 2016;291(34):17510-17522.
[31] DOMINGUES R, SANT’ANNA R, DA FONSECA ACC, et al. Extracellular alpha-synuclein: Sensors, receptors, and responses. Neurobiol Dis. 2022;168:105696.
[32] DINTER E, SARIDAKI T, NIPPOLD M, et al. Rab7 induces clearance of α-synuclein aggregates. Neurochem. 2016;138(5):758-774.
[33] SZEGÖ EM, VAN DEN HAUTE C, HÖFS L, et al. Rab7 reduces α-synuclein toxicity in rats and primary neurons. Exp Neurol. 2022;347:113900.
[34] VODICKA P, CHASE K, IULIANO M, et al. Autophagy Activation by Transcription Factor EB (TFEB) in Striatum of HDQ175/Q7 Mice. J Huntingtons Dis. 2016;5(3):249-260.
[35] HU K, GAIRE BP, SUBEDI L, et al. Interruption of Endolysosomal Trafficking After Focal Brain Ischemia. Front Mol Neurosci. 2021;14:719100.
[36] XI Z, DENG W, WANG L, et al. Association of Alpha-Soluble NSF Attachment Protein with Epileptic Seizure. J Mol Neurosci. 2015;57(3):417-425.
[37] CUPERTINO RB, KAPPEL DB, BANDEIRA CE, et al. SNARE complex in developmental psychiatry: neurotransmitter exocytosis and beyond. J Neural Transm (Vienna). 2016;123(8):867-883.
[38] MERINO P, DIAZ A, MANRIQUE LG, et al. Urokinase-type plasminogen activator (uPA) promotes ezrin-mediated reorganization of the synaptic cytoskeleton in the ischemic brain. J Biol Chem. 2018;293(24):9234-9247.
[39] SONG X, WANG W, WANG H, et al. Acetylation of ezrin regulates membrane-cytoskeleton interaction underlying CCL18-elicited cell migration. J Mol Cell Biol. 2020;12(6):424-437.
[40] YU Y, ZENG P, XIONG J, et al. Epigenetic drugs can stimulate metastasis through enhanced expression of the pro-metastatic Ezrin gene. PLoS One. 2010;5(9):e12710.
[41] WASIK AA, KOSKELAINEN S, HYVÖNEN ME, et al. Ezrin is down-regulated in diabetic kidney glomeruli and regulates actin reorganization and glucose uptake via GLUT1 in cultured podocytes. Am J Pathol. 2014;184(6):1727-1739.
[42] MEDINA C, DE LA FUENTE V, TOM DIECK S, et al. LIMK, Cofilin 1 and actin dynamics involvement in fear memory processing. Neurobiol Learn Mem. 2020;173:107275.
[43] SUNGUR A, STEMMLER L, WÖHR M, et al. Impaired Object Recognition but Normal Social Behavior and Ultrasonic Communication in Cofilin1 Mutant Mice. Front Behav Neurosci. 2018;12:25.
[44] TSUBOTA T, OKUBO-SUZUKI R, OHASHI Y, et al. Cofilin1 controls transcolumnar plasticity in dendritic spines in adult barrel cortex. PLoS Biol. 2015;13(2):e1002070. |