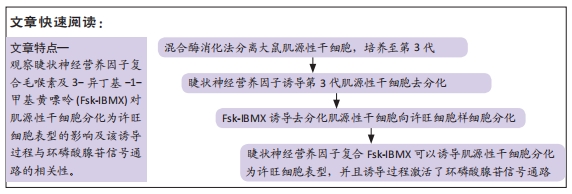
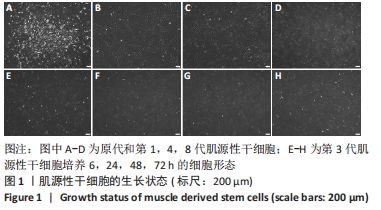
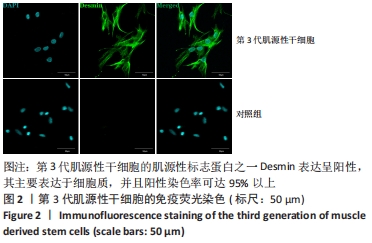
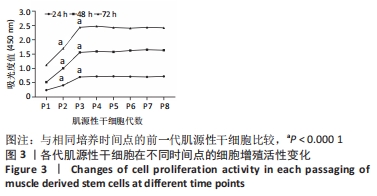
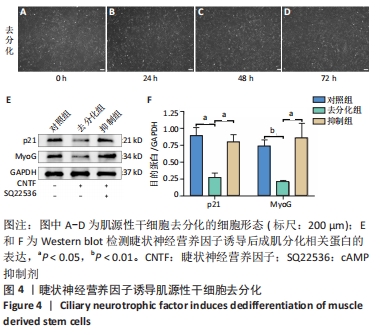
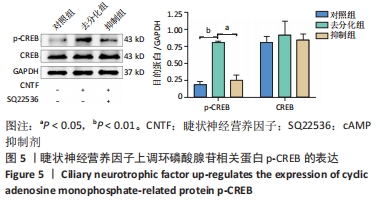
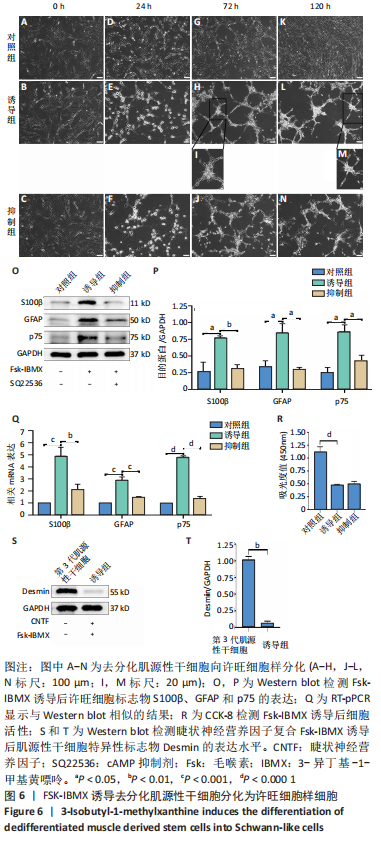
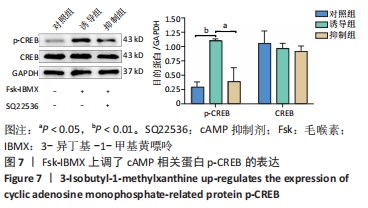
[1] YI S, ZHANG Y, GU X, et al. Application of stem cells in peripheral nerve regeneration. Burns Trauma. 2020;8:tkaa002.
[2] SULLIVAN R, DAILEY T, DUNCAN K, et al. Peripheral Nerve Injury: Stem Cell Therapy and Peripheral Nerve Transfer. Int J Mol Sci. 2016;17(12):2101.
[3] LI R, LIU Z, PAN Y, et al. Peripheral nerve injuries treatment: a systematic review. Cell Biochem Biophys. 2014;68(3):449-454.
[4] JIANG L, JONES S, JIA X. Stem Cell Transplantation for Peripheral Nerve Regeneration: Current Options and Opportunities. Int J Mol Sci. 2017;18(1):94.
[5] PANAGOPOULOS GN, MEGALOIKONOMOS PD, MAVROGENIS AF. The Present and Future for Peripheral Nerve Regeneration. Orthopedics. 2017;40(1):e141-e156.
[6] MUSAVI L, BRANDACHER G, HOKE A, et al. Muscle-derived stem cells: important players in peripheral nerve repair. Expert Opin Ther Targets. 2018;22(12):1009-1016.
[7] KUBIAK CA, GROCHMAL J, KUNG TA, et al. Stem-cell-based therapies to enhance peripheral nerve regeneration. Muscle Nerve. 2020;61(4):449-459.
[8] LAVASANI M, THOMPSON SD, POLLETT JB, et al. Human muscle-derived stem/progenitor cells promote functional murine peripheral nerve regeneration. J Clin Invest. 2014;124(4):1745-1756.
[9] ZHOU J, CUI H, LU H, et al. Muscle-derived stem cells in peripheral nerve regeneration: reality or illusion? Regen Med. 2017;12(4):459-472.
[10] SAYAD FATHI S, ZAMINY A. Stem cell therapy for nerve injury. World J Stem Cells. 2017;9(9):144-151.
[11] TAMAKI T. Biomedical applications of muscle-derived stem cells: from bench to bedside. Expert Opin Biol Ther. 2020;20(11):1361-1371.
[12] CHEN ZX, LU HB, JIN XL, et al. Skeletal muscle-derived cells repair peripheral nerve defects in mice. Neural Regen Res. 2020;15(1):152-161.
[13] LV P, WANG W, CAO Z, et al. Fsk and IBMX inhibit proliferation and proapoptotic of glioma stem cells via activation of cAMP signaling pathway. J Cell Biochem. 2019;120(1):321-331.
[14] SHAHBAZI A, SAFA M, ALIKARAMI F, et al. Rapid Induction of Neural Differentiation in Human Umbilical Cord Matrix Mesenchymal Stem Cells by cAMP-elevating Agents. Int J Mol Cell Med. 2016;5(3):167-177.
[15] ZHANG P, CHEN XP. The regulation of myoblast plasticity and its mechanism. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2012;28(6):524-531.
[16] ZHANG L, SEITZ LC, ABRAMCZYK AM, et al. cAMP initiates early phase neuron-like morphology changes and late phase neural differentiation in mesenchymal stem cells. Cell Mol Life Sci. 2011;68(5):863-876.
[17] THOMPSON R, CASALI C, CHAN C. Forskolin and IBMX Induce Neural Transdifferentiation of MSCs Through Downregulation of the NRSF. Sci Rep. 2019; 9(1):2969.
[18] DE LA ROSA MB, KOZIK EM, SAKAGUCHI DS. Adult Stem Cell-Based Strategies for Peripheral Nerve Regeneration. Adv Exp Med Biol. 2018;1119:41-71.
[19] CHING RC, WIBERG M, KINGHAM PJ. Schwann cell-like differentiated adipose stem cells promote neurite outgrowth via secreted exosomes and RNA transfer. Stem Cell Res Ther. 2018;9(1):266.
[20] ROTSHENKER S. Wallerian degeneration: the innate-immune response to traumatic nerve injury. J Neuroinflammation. 2011;8:109.
[21] SEDDON HJ, MEDAWAR PB, SMITH H. Rate of regeneration of peripheral nerves in man. J Physiol. 1943;102(2):191-215.
[22] WANG D, LIU XL, ZHU JK, et al. Bridging small-gap peripheral nerve defects using acellular nerve allograft implanted with autologous bone marrow stromal cells in primates. Brain Res. 2008;1188:44-53.
[23] LU L, CHEN X, ZHANG CW, et al. Morphological and functional characterization of predifferentiation of myelinating glia-like cells from human bone marrow stromal cells through activation of F3/Notch signaling in mouse retina. Stem Cells. 2008;26(2):580-590.
[24] MAURO A. Satellite cell of skeletal muscle fibers. J Biophys Biochem Cytol. 1961; 9(2):493-495.
[25] TAMAKI T. Therapeutic capacities of human and mouse skeletal muscle-derived stem cells for a long gap peripheral nerve injury. Neural Regen Res. 2017;12(11): 1811-1813.
[26] TAMAKI T. Bridging long gap peripheral nerve injury using skeletal muscle-derived multipotent stem cells. Neural Regen Res. 2014;9(14):1333-1336.
[27] TAMAKI T, HIRATA M, NAKAJIMA N, et al. A Long-Gap Peripheral Nerve Injury Therapy Using Human Skeletal Muscle-Derived Stem Cells (Sk-SCs): An Achievement of Significant Morphological, Numerical and Functional Recovery. PLoS One. 2016;11(11):e0166639.
[28] TAMAKI T, HIRATA M, SOEDA S, et al. Preferential and comprehensive reconstitution of severely damaged sciatic nerve using murine skeletal muscle-derived multipotent stem cells. PLoS One. 2014;9(3):e91257.
[29] DEUMENS R, BOZKURT A, MEEK MF, et al. Repairing injured peripheral nerves: Bridging the gap. Prog Neurobiol. 2010;92(3):245-276.
[30] XU Z, CHEN Z, FENG W, et al. Grafted muscle-derived stem cells promote the therapeutic efficiency of epimysium conduits in mice with peripheral nerve gap injury. Artif Organs. 2020;44(5):E214-E225.
[31] DEASY BM, GHARAIBEH BM, POLLETT JB, et al. Long-term self-renewal of postnatal muscle-derived stem cells. Mol Biol Cell. 2005;16(7):3323-3333.
[32] ARSIC N, MAMAEVA D, LAMB NJ, et al. Muscle-derived stem cells isolated as non-adherent population give rise to cardiac, skeletal muscle and neural lineages. Exp Cell Res. 2008;314(6):1266-1280.
[33] ALESSANDRI G, PAGANO S, BEZ A, et al. Isolation and culture of human muscle-derived stem cells able to differentiate into myogenic and neurogenic cell lineages. Lancet. 2004;364(9448):1872-1883.
[34] KWON JS, KIM GH, KIM DY, et al. Chitosan-based hydrogels to induce neuronal differentiation of rat muscle-derived stem cells. Int J Biol Macromol. 2012;51(5): 974-979.
[35] KIM ES, KIM GH, KANG ML, et al. Potential induction of rat muscle-derived stem cells to neural-like cells by retinoic acid. J Tissue Eng Regen Med. 2011;5(5): 410-414.
[36] TANG Y, HE H, CHENG N, et al. PDGF, NT-3 and IGF-2 in combination induced transdifferentiation of muscle-derived stem cells into Schwann cell-like cells. PLoS One. 2014;9(1):e73402.
[37] KINJO T, WATABE T, KOBACHI K, et al. Single-Cell Activation of the cAMP-Signaling Pathway in 3D Tissues with FRET-Assisted Two-Photon Activation of bPAC. ACS Chem Biol. 2020;15(11):2848-2853.
[38] 江声伟,刘德武,孙宝丽,等.cAMP信号通路对卵泡发育调控机制的研究进展[J].黑龙江畜牧兽医,2020(23):44-47.
[39] ROONEY GE, HOWARD L, O’BRIEN T, et al. Elevation of cAMP in mesenchymal stem cells transiently upregulates neural markers rather than inducing neural differentiation. Stem Cells Dev. 2009;18(3):387-398.
[40] 张东云,靳伟东,刘宏亮,等.CREB信号通路在神经系统疾病中的调控作用及机制[J].中华全科医学,2019,17(4):644-648.
[41] WANG H, XU J, LAZAROVICI P, et al. cAMP Response Element-Binding Protein (CREB): A Possible Signaling Molecule Link in the Pathophysiology of Schizophrenia. Front Mol Neurosci. 2018;11:255.
[42] DEZAWA M, KANNO H, HOSHINO M, et al. Specific induction of neuronal cells from bone marrow stromal cells and application for autologous transplantation. J Clin Invest. 2004;113(12):1701-1710.
[43] 徐如祥,戴宜武,姜晓丹,等.成年骨髓间质干细胞体外诱导分化成神经细胞研究[J].中华神经外科疾病研究杂志,2002,1(1):63-67.
[44] HERMANN A, MAISEL M, STORCH A. Epigenetic conversion of human adult bone mesodermal stromal cells into neuroectodermal cell types for replacement therapy of neurodegenerative disorders. Expert Opin Biol Ther. 2006;6(7):653-670.
[45] EL-DEEB OS, GHANEM HB, EL-ESAWY RO, et al. The modulatory effects of luteolin on cyclic AMP/Ciliary neurotrophic factor signaling pathway in experimentally induced autoimmune encephalomyelitis. IUBMB Life. 2019;71(9):1401-1408.
[46] HELLSTRÖM M, HARVEY AR. Cyclic AMP and the regeneration of retinal ganglion cell axons. Int J Biochem Cell Biol. 2014;56:66-73.
|