中国组织工程研究 ›› 2022, Vol. 26 ›› Issue (16): 2576-2583.doi: 10.12307/2022.260
• 生物材料综述 biomaterial review • 上一篇 下一篇
钛植入物表面生物化学改性对骨整合的影响
车振家,朱正清,朱礼伟,李友斌,祝晨一,黄岚峰
- 吉林大学第二医院骨科,吉林省长春市 130041
-
收稿日期:2021-01-26修回日期:2021-01-28接受日期:2021-02-27出版日期:2022-06-08发布日期:2021-12-23 -
通讯作者:黄岚峰,博士,硕士生导师,主任医师,教授,吉林大学第二医院骨科,吉林省长春市 130041 -
作者简介:车振家,男,1996 年生,辽宁省营口市人,汉族,吉林大学第二医院在读硕士,主要从事骨组织工程细胞因子及骨与种植体整合的研究。 -
基金资助:吉林省财政厅卫生专项项目(201817294302),项目负责人:黄岚峰;吉林省科技发展计划资助项目(20200201327JC),项目负责人:黄岚峰
Effects of biochemical modification of titanium implant surface on the osseointegration
Che Zhenjia, Zhu Zhengqing, Zhu Liwei, Li Youbin, Zhu Chenyi, Huang Lanfeng
- Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China
-
Received:2021-01-26Revised:2021-01-28Accepted:2021-02-27Online:2022-06-08Published:2021-12-23 -
Contact:Huang Lanfeng, MD, Master’s supervisor, Chief physician, Professor, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Che Zhenjia, Master candidate, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
Supported by:the Health Special Project of Jilin Provincial Finance Department, No. 201817294302 (to HLF); the Science and Technology Development Program of Jilin Province, No. 20200201327JC (to HLF)
摘要:
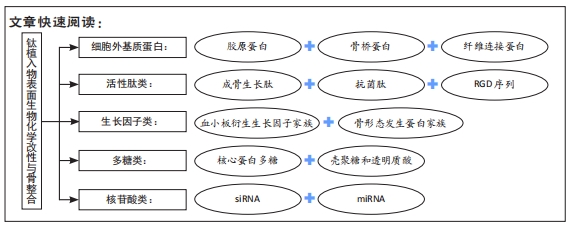
文题释义:
钛植入物骨整合:是指在人体活的骨组织与钛种植体之间发生的牢固、持久而直接的结合,即负载力量的种植体的表面与有活力的骨组织之间存在结构上和功能上直接的联系,种植体与骨组织之间没有任何结缔组织存在,不间隔任何组织。
生物化学改性:起初是作为化学改性的变异出现,是指通过将特定的蛋白、多肽、生长因子、多糖和核苷酸固定于种植体表面,这样可使生物活性分子直接在骨与植入体之间发挥作用,诱导成骨细胞增殖、分化和矿化,更为直接有效地促进骨整合。
背景:钛及钛合金因具有良好的机械性能和生物惰性等被广泛应用于骨科植入领域。
目的:综述不同的生物分子搭载到钛植入物表面对骨整合的影响。
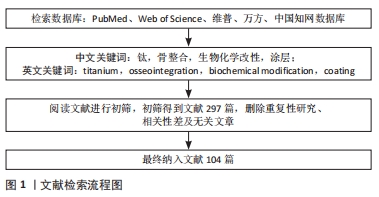
方法:由第一作者应用计算机以“titanium,osseointegration,biochemical modification,coating”为英文检索词,以“钛,骨整合,生物化学改性,涂层”为中文检索词,应用计算机检索PubMed、Web of Science、维普、万方、中国知网数据库中1991-2020年已发表的相关文献,并进行筛选、归纳与总结,最终纳入104篇相关文献进行综述。
结果与结论:钛植入体表面单一的物理和化学修饰方式主要是间接影响细胞行为,而生物分子涂层可以直接参与生物过程,这在诱导骨形成方面更有效,不仅显著提高了骨与植入体之间早期的骨整合,还降低了因炎症反应导致的手术失败及假体翻修发生率。虽然钛植入物的生物化学改性可以直接参与生物过程,但是目前还有许多问题尚待解决:生物活性物质在钛植入体表面的长期稳定性、释放的速率以及钛植入物与机体细胞和组织之间的作用机制问题还需进一步研究,从而得到骨与钛植入物之间的早期和长期的骨整合。
https://orcid.org/0000-0001-9029-4097 (车振家)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
中图分类号:
引用本文
车振家, 朱正清, 朱礼伟, 李友斌, 祝晨一, 黄岚峰. 钛植入物表面生物化学改性对骨整合的影响[J]. 中国组织工程研究, 2022, 26(16): 2576-2583.
Che Zhenjia, Zhu Zhengqing, Zhu Liwei, Li Youbin, Zhu Chenyi, Huang Lanfeng. Effects of biochemical modification of titanium implant surface on the osseointegration[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2576-2583.
2.1.1 胶原蛋白 Ⅰ型胶原蛋白已被证明是一种生物相容性高的有机生物材料,植入后容易被宿主组织吸收[25],可以有效改善种植体周围的骨重塑[26]。SCARANO等[27]先将钛植入物进行喷砂和酸蚀处理,再将Ⅰ型胶原蛋白共价结合到经过处理的钛植入物表面,将其植入到兔膝关节中,在第15,30,60天进行组织学分析和Micro-CT断层扫描,对涂覆和未涂覆胶原植入物的种植体骨结合率、骨区域内螺纹和骨区域外螺纹进行分析,结果显示钛种植体表面的Ⅰ型胶原蛋白提高了钛种植体表面生物活性,并且加速了早期成骨。然而,胶原蛋白存在着动物源性胶原的免疫反应和酶的快速降解等缺点,限制了其应用[28]。为了克服这些缺点,大多数胶原蛋白都是在交联后使用的,目前多采用化学试剂的方式进行交联,但这种方式所残留的化学试剂会对细胞产生一定的毒性作用,而伽马辐射交联的方式可以克服这一缺点[29]。
BAE等[30]通过伽马照射将胶原蛋白交联到植入物表面,由于交联过程无化学试剂的参与,所以体外细胞实验无毒性作用和化学试剂的不良反应,而且表现出较高的成骨分化和成骨基因的表达;将钛植入物植入到小鼠胫骨模型中,对该模型进行组织学分析和Micro-CT断层扫描,结果显示出良好的骨整合能力,并且也没有受到化学试剂的影响。
2.1.2 骨桥蛋白 在骨整合过程中,骨内种植体周围的骨桥蛋白在直接成骨过程中是必不可少的。MAKISHI等[31]将经过喷砂处理过的钛种植体分别放置于骨桥蛋白基因敲除和野生的小鼠的上颌内,植入3,5,7,28 d后,使用免疫组化、原位杂交和电子探针纤维分析仪对标本进行分析,结果表明,骨桥蛋白缺陷干扰了直接成骨,延迟了种植体的骨整合。FIORELLINN等[32]将骨桥蛋白涂覆在等离子喷涂钛的表面,并与未涂覆的等离子喷涂钛进行对比,犬模型的组织形态计量学分析显示,第4周时涂覆组植入物的骨-植入物接触和周围骨密度明显高于未涂覆组,这使植入手术产生更高的可预测性,并且降低了其风险。
2.1.3 纤维连接蛋白 在不同的细胞外基质蛋白中,纤连蛋白已被证明能诱导良好的细胞反应[33],它包含多个结构域,不仅与细胞相互作用,还与其他蛋白质相互作用,介导许多细胞过程,如细胞黏附、迁移、生长和分化[34]。不仅如此,纤连蛋白搭载在钛表面上时还能有效支持细胞生长。PRAMONO等[35]先用喷砂机将钛圆盘进行喷砂处理,再将人血浆纤连蛋白涂布在经过喷砂处理的钛圆盘上,与小鼠胚胎成骨细胞前体细胞(MC3T3-E1细胞)联合培养,发现表面涂有纤连蛋白的喷砂钛增强了细胞的黏附和增殖;除此之外,基质矿化和碱性磷酸酶活性也明显增强。
PIVERA-CHACON等[36]发现使用全长纤维连接蛋白可以使钛植入物功能化,从而显示出良好的骨传导能力。然而,全长纤维连接蛋白也存在一些不足,首先是其对蛋白水解、降解敏感,还可能引起免疫反应[37];其次是可以从人血浆中的纯化纤连蛋白的量很少,因此其临床应用受到了阻碍[38]。与此不同,GUILLEM-MARTI等[39]将纤维连接蛋白重组片段组成的细胞外基质样结构与聚乳酸共静电纺丝,并共价结合在抛光的钛盘上,为了克服聚乳酸的细胞黏附能力不足、提高细胞反应性,他们在纳米纤维中加入了纤维连接蛋白的细胞附着位点片段,结果显示其显著改善了人成骨肉瘤SaOS-2细胞的生物反应,提高了骨结合能力。在此基础上,GUILLEM-MARTI等[40]还将纤维连接蛋白肝素结合Ⅱ片段引入精氨酸-甘氨酸-天冬氨酸序列,使DNA突变生成了一个新的重组蛋白片段,再通过硅烷化法共价结合在钛盘上。一方面,这种新蛋白在钛上固定时可以提高细胞黏附性,同时保留了细胞分化能力;另一方面,它也表现出了良好的因子结合能力。所以,这种获得重组蛋白的新策略为设计再生医学活性分子打开了一个新的窗口。
2.2 活性肽类
2.2.1 成骨生长肽 成骨生长肽是一种可溶性、短而线性的生长因子肽片段,在体外可直接调节细胞增殖、成骨细胞分化和基质矿化,在体内可以促进骨形成和骨折愈合。由于成骨生长肽具有良好的骨诱导活性,所以当其固定在表面和支架上时可以促进成骨细胞分化和骨生长[41]。POLICASTRO等[42]证明,成骨生长肽功能化聚苯丙氨酸基显示了显著的组织与支架整合能力,并能促进其成骨。MOORE等[43]还发现,通过点击化学在合成底物上梯度固定成骨生长肽可以促进MC3T3-E1细胞增殖。然而,目前将成骨生长肽固定在钛种植体表面以改善骨结合的研究还尚少。
LAI等[41]在钛表面通过阳极氧化制备了一层直径约70 nm的定向二氧化钛纳米管,然后通过聚多巴胺中间层将成骨生长肽偶联到二氧化钛纳米管上,体外细胞培养实验表明,成骨生长肽功能化的二氧化钛纳米管能促进成骨细胞的扩散和分化。LIU等[44]将成骨生长肽与被证明可以抑制核因子κB激活和c-Fos基因表达的N-乙酰半胱氨酸结合[45],合成了多功能肽成骨生长肽-N-乙酰半胱氨酸,然后通过硅烷化法将多功能肽共价结合在钛表面,将其分别与RAW264.7细胞和成骨细胞共同培养,体外对破骨细胞分化因子诱导破骨细胞形成的研究表明,Ti-成骨生长肽-N-乙酰半胱氨酸表面可以抑制多核细胞的形成,抑制破骨细胞相关基因的表达;对成骨细胞的研究结果表明,Ti-成骨生长肽-N-乙酰半胱氨酸底物能促进成骨细胞的黏附和成骨。此研究为骨科领域制备生物功能化钛及钛合金植入物提供了新的思路和方法。
2.2.2 抗菌肽 目前对种植体和骨生物材料的研究主要集中在促进种植体骨整合和诱导成骨两个方面,而忽视了这一过程出现的免疫炎症反应。然而,种植体放置后的早期炎症反应和周围的慢性炎症反应对种植体骨整合的长期生存都是不利的[46],这往往导致体内和体外研究的冲突和分歧[47]。为了改善这一现状,近年来许多研究开始尝试将抗菌性能纳入成骨性能的评价体系[48],但是由于抗菌活性的有效性和生理环境协调性之间的差异,目前还没有获得最佳解决方案[49]。
植入物表面的抗菌涂层作为一种抗菌材料在临床研究中取得了一定的成功,该技术最大的优点是能使用的抗菌药物种类多,控释可行性强[50],例如:TAHERI等[51]制备纳米银颗粒涂层,显示出显著的抗菌效果;BADAR等[52]制备了负载环丙沙星的层状双氢氧化物涂层,以延长抗生素的释放。然而在转化应用方面,金属颗粒和抗生素涂层在持续使用方面都有其自身的局限性,特别是耐药细菌的产生远远超过了替代药物的发展[53]。而抗菌肽具有广谱抗菌活性,可以规避抗生素的耐药性问题[54],其中抗菌肽LL-37具有良好的抗菌特性和生物相容性,是一种理想的抗菌剂,它不仅可以杀灭细菌,还能促进血管生成和招募免疫细胞、干细胞等多种生理活动[8],已被证实在宿主防御和损伤愈合过程中具有多效性[55]。
HE等[56]先将钛种植体进行微弧氧化处理,再用聚多巴胺为中间层将抗菌肽LL-37连接到钛植入物表面,最后用磷脂在最外层覆以包衣形成复合涂层,将经过不同处理的钛表面与金黄色葡萄球菌和大肠杆菌联合培养,以验证复合涂层的抗菌活性和抗菌率,第6小时的抗菌活性结果显示,在裸钛上培养的大肠杆菌和金黄色葡萄球菌的形状是完整和饱满的,而接种在复合涂层上的金黄色葡萄球菌和大肠杆菌都表现出明显的形态变化,其中细菌膜变得皱缩和破碎,这表明LL-37的抗菌机制是破坏细菌的膜结构;第24小时的抗菌率结果显示,只经过微弧氧化的钛表面对金黄色葡萄球菌和大肠杆菌的抑菌率分别为25%和24.6%,而经过抗菌肽LL-37修饰过的钛表面对金黄色葡萄球菌和大肠杆菌的抑菌率达到了71.4%和66.7%,抗菌率的实验结果与早期的抗菌实验结果一致。在此基础上,他们还将不同处理的钛表面与骨髓间充质干细胞和成骨细胞共同培养来验证复合涂层的生物相容性,结果显示复合涂层组的骨髓间充质干细胞和成骨细胞的活性均未显示出负面影响,这为制备同时具有抗菌效果和细胞相容性的抗菌钛植入物提供了一种方法。
抗菌肽LL-37还可以显著促进骨髓间充质干细胞的迁移和旁分泌因子的表达[57]。HE等[8]经过微弧氧化和水热两步处理在种植体表面沉积羟基磷灰石,再通过聚多巴胺中间层将抗菌肽LL-37覆以种植体表面形成复合涂层,然后再将其与骨髓间充质干细胞进行联合培养,通过体外实验和体内实验得出:抗菌肽LL-37涂层可以诱导骨髓间充质干细胞迁移,刺激骨髓间充质干细胞发生营养反应,植入体钛表面的显微结构与羟基磷灰石沉积可促进间充质干细胞的成骨分化。这为通过骨髓间充质干细胞迁移来提高钛植入体的骨整合性能提供了一种可能。
除抗菌肽LL-37外,多肽GL13K是存在于人体中典型的阳离子抗菌肽之一。CHEN等[47]以硅烷为化学连接剂将抗菌肽GL13K固定在种植体钛表面,并与RAW264.7细胞联合培养,通过ELISA和qRT-PCR分别检测了不同极化的巨噬细胞炎性、抗炎细胞因子、炎症相关基因及抗炎基因的表达,研究表明抗菌肽GL13K固定的钛表面可以调节M1巨噬细胞细胞因子的释放,抑制巨噬细胞促炎因子的分泌,促进M2巨噬细胞抗炎因子的释放。巨噬细胞可以由促炎极化状态向抑炎极化状态转变,从而促进炎症过程向组织愈合过程转化,后期还可能影响骨整合。LI等[58]利用氧化处理在钛种植体表面覆以二氧化钛纳米管,通过浸泡技术把抗菌肽GL13K固定于表面形成复合涂层,并且在体外将其与MC3T3-E1细胞联合培养,结果显示抗菌肽GL13K早期可显著促进成骨细胞附着,中早期可预防种植体感染,后期可促进骨整合。
2.2.3 精氨酸-甘氨酸-天冬氨酸序列(RGD序列) RGD序列能促进成骨细胞在骨科植入物上的黏附和增殖,并且在钛植入物上固定有RGD序列的涂层已被证明具有良好的骨整合性[59],在体外不仅可以刺激成骨细胞的分化,还对内皮细胞的增殖和分化具有积极的作用[60];在体内可以增加骨与种植体界面的结合程度,从而缩短种植周期,提高种植成功率[61]。
PARFENOVA等[19]用等离子体电解氧化技术在钛表面形成无机多孔亚层,再通过双膦酸盐为化学连接剂,将线性RGD序列共价偶联到钛表面形成复合涂层,体外对成纤维细胞、间充质干细胞和成骨细胞样细胞的增殖和活性进行研究,结果表明经过双膦酸盐连接的钛表面会促进细胞的增殖,未修饰的钛表面对细胞增殖无影响。然而,对于功能化的钛植入物的体内研究还需进一步的实验。
此外,不同构象的RGD序列固定在钛植物表面可能对骨整合产生不同的效果。HELLER等[18]采取硅烷化法将环状RGD序列和线状RGD序列分别共价偶联在钛表面,比较两者在体外和体内对骨反应的影响,在体外将成骨细胞分别与不同钛表面联合培养,对其细胞黏附、增殖、分化及碱性磷酸酶和骨钙素进行评价;在体内将不同表面处理的钛植入兔胫骨近端,并对骨的生长参数进行分析,结果显示:在体外,环状RGD序列和线状RGD序列修饰钛组作用差异不大,但均强于未处理的钛组;体内研究表明,在植入的第3周和第6周,环状RGD序列修饰组的骨与种植体结合率均高于其他组,这些发现可以前瞻性地应用于植入材料,提高植入的成功率。
2.3 生长因子
2.3.1 血小板衍生生长因子家族 血小板衍生生长因子家族主要包括血小板衍生生长因子和血管内皮生长因子,其中血小板衍生生长因子是组织修复的一个关键调节因子,在组织损伤后由血小板释放,通过增加细胞的增殖和趋化性来加快愈合过程[62]。血小板衍生生长因子在钛表面的首次应用是在1991年的一项动物研究中[63]。在此基础上,ORTOLANI等[64]发现血小板衍生生长因子与胰岛素样生长因子1结合后,一方面增加了种植体周围间隙的骨填充,另一方面可有效改善兔股骨干骺端种植体周围骨整合的能力。KIM等[65]也发现,血小板衍生生长因子与骨形态发生蛋白2一同接种于肝素化的钛表面可以增强成骨细胞功能和骨整合。
MA等[66]通过阳极氧化和化学介导将血小板衍生生长因子以共价结合的方式固定在植入体钛表面,建立了一种新型的二氧化钛纳米管表面结构,并且在大鼠股骨远端模型中局部植入后,可以在骨髓间充质干细胞中见到骨钙素表达的爆发,反映了经过表面修饰的钛植入物周围骨形成的增强。
血管形成依赖于血管内皮生长因子,除了促进血管生成以外,血管内皮生长因子还可促进成骨[67-68]。此外,血管形成还可影响骨的生理性愈合[69]。IZQUIERDO-BARBA等[70]在Ti6Al4V支架上涂敷含硅的羟基磷灰石(SiHA),并在涂层表面搭载血管内皮生长因子,体外细胞培养实验证明,吸附血管内皮生长因子的支架可促进支架表面内皮细胞的增殖,含硅羟基磷灰石涂层的支架可促进成骨细胞的增殖;对绵羊模型的体内研究表明,在搭载血管内皮生长因子的含硅羟基磷灰石涂层支架上表现出协同作用,表现为更多的骨化和血管生成。
2.3.2 骨形态发生蛋白家族 骨形态发生蛋白家族从属于转化生长因子β超家族,在骨的创伤修复和诱导成骨细胞分化中起重要作用[71-72]。在已知的骨形态发生蛋白家族成员中,目前研究较多的主要是骨形态发生蛋白2,6,7和9,其中骨形态发生蛋白2 是目前应用最广泛的生长因子之一,其在骨再生过程中起着至关重要的作用。有研究表明,将合成支架与生长因子相结合后生长因子可以促进骨结合和血管的形成,能很好地愈合创伤造成的严重骨缺损[73],但由于骨形态发生蛋白2的半衰期短、扩散缓慢,存在短距离扩散和局部作用,其必须以可控和持续的方式携带和释放[74]。
TENG等[75]首先采用微弧氧化技术对多孔钛合金进行表面处理,然后通过电化学沉积将钙(Ca)和磷(P)结合到孔隙中,用骨形态发生蛋白2对Ca、P包覆钛进行功能化,体外实验表明,微弧氧化-CaP-骨形态发生蛋白2可促进骨细胞的增殖、分化和矿化,形成种植体中心;此外,他们还进行了新西兰大白兔颅骨植入实验,结果表明种植体的固定能力增强,具有明显促进骨愈合的潜力,同时沉积在Ca、P层的骨形态发生蛋白2释放时间可延长至35 d。WANG等[74]首先采用微弧氧化法在钛表面制备羟基磷灰石和二氧化钛复合涂层,然后采用浸涂法将骨形态发生蛋白2包裹的壳聚糖涂层负载在钛表面。这种复合涂层改变了钛植入体的生物性能,提高了其抗菌性,也改善了骨形态发生蛋白2的释放行为。
CHEN等[76]通过碱和热处理来制作多孔钛植入物与超亲水性和负电荷表面,然后通过物理吸附将带正电荷的鱼精蛋白、带负电荷的海藻酸盐、最外层带正电荷的鱼精蛋白在多孔钛植入物表面制备了“三明治样”涂层,然后利用生物功能化技术进一步固定骨形态形成蛋白2,从而形成多孔钛植入物-鱼精蛋白/海藻酸盐/鱼精蛋白-骨形态发生蛋白2生物涂层,体外生物学功能及体内成骨研究结果表明,多孔钛植入物-鱼精蛋白/海藻酸盐/鱼精蛋白-骨形态发生蛋白2显著改善了MC3T3-E1细胞的体外成骨分化,并且在体内可能通过激活整合素和骨形态发生蛋白/Smad信号通路来增强体内大鼠模型的骨整合。
除单因子外,有些研究通过双因子包覆在钛种植体表面来研究骨整合的可行性。与骨形态发生蛋白2相比,生长分化因子5作为促进骨形成的生长因子较少为人所知。YANG等[77]用肝素为中间涂层将骨形态发生蛋白2和生长分化因子5涂敷在种植体钛表面,结果表明,一方面可以延长涂层的控释时间至30 d;另一方面,两种生长因子包覆的钛增强了种植体与骨界面的骨形成和骨整合。
骨形态发生蛋白6在骨愈合过程中起着重要的作用[78]。BRIGAUD等[79]将骨形态发生蛋白6与纤连蛋白结合在钛-羟基磷灰石表面来测试其生物活性,结果显示纤连蛋白-骨形态发生蛋白6诱导骨整合,并与其他实验组相比产生了最小的不良反应。
除了对骨形态发生蛋白6单因子的研究,有实验将骨形态发生蛋白6与血小板衍生生长因子双因子涂敷在植入体表面进行研究。KECELI等[80]将钛植入物进行阳极氧化处理,用浸泡法在阳极氧化处理表面负载骨形态发生蛋白6,再通过静电纺丝将丝素蛋白和血小板生长因子涂敷在钛表面,形成阳极氧化处理-骨形态发生蛋白6-血小板生长因子-丝素蛋白复合涂层,在体外实验显示,由于骨形态发生蛋白6和血小板衍生生长因子可控释放的双重作用,该涂层细胞增殖、矿化和基因表达水平均增高,但是对骨整合的更进一步影响需要通过体内实验证明。
与成骨早期参与作用的骨形态发生蛋白2相比,骨形态发生蛋白7主要在骨形成的后期发挥作用[81]。此外,骨形态发生蛋白7还可诱导未分化的非成骨细胞向成骨细胞转化,并刺激已转化的成骨细胞成熟[82],在体内也显示出良好的骨再生能力[83]。
AL-JARSHA等[84]用聚丙烯酸乙酯溶液在钛表面进行旋涂,通过物理相互作用在钛表面形成聚丙烯酸乙酯层,再将已经暴露生长因子和整合素结合域的纤维连接蛋白特异地结合在聚丙烯酸乙酯层表面,这种方法可以用极低浓度的骨形态发生蛋白7(25 μg/L)使这些底物功能化,从而创造一个微环境促进人骨髓间充质干细胞向成骨分化。他们还对人骨髓间充质干细胞进行体外实验,结果表明该复合涂层具有良好的生物相容性及晚期成骨分化的能力,这项技术开辟了局部使用极低剂量的骨形态发生蛋白7来使骨结合最大化的途径,从而提高了种植体的可行性。
在骨形态发生蛋白家族中,骨形态发生蛋白9被认为是最具骨诱导分化能力的骨形态发生蛋白之一[85-87],但是目前关于骨形态发生蛋白9对成骨细胞和钛之间相互作用的研究相对较少。SOUZA等[85]将MC3T3-E1细胞与钛共同培养,基于剂量反应实验选择了最适浓度的骨形态发生蛋白9(20 nmol/L)来评价骨形态发生蛋白9对生长在钛表面成骨细胞分化的影响,结果表明骨形态发生蛋白9诱导成骨细胞发育相关转录因子2、骨钙素和骨涎蛋白的高表达,并增加碱性磷酸酶活性和细胞外基质的矿化。然而,将骨形态发生蛋白9搭载到钛表面的研究还需要进一步探索。
2.4 多糖类
2.4.1 核心蛋白多糖 核心蛋白多糖是一种常见的细胞外基质分子,是一种小的富含亮氨酸的蛋白聚糖,由核心蛋白和糖胺聚糖链组成[88]。MOHAN等[89]研究发现,核心蛋白多糖可以通过诱导成纤维细胞凋亡和调节纤维结构抑制纤维包封。此外,核心蛋白多糖在体外也能促进成骨细胞矿化。
HE等[90]用聚多巴胺将核心蛋白多糖负载在钛的表面,观察纤维包被抑制和骨生长,体外培养成纤维细胞和成骨细胞实验结果表明,在核心蛋白多糖修饰的表面上,一方面成纤维细胞的增殖、迁移和胶原合成功能明显减弱;另一方面其还可以增强钙沉积和碱性磷酸酶活性。这种抑制了成纤维细胞的增殖、改善了成骨细胞功能的方法,对骨科种植体的强化具有潜在的应用价值。
2.4.2 壳聚糖和透明质酸 壳聚糖具有良好的生物相容性、降解性和抗菌性,是一种用于骨再生的生物活性物质,并且由于壳聚糖和细胞外基质结构之间的相似性,基于壳聚糖的复合材料可以促进细胞的黏附、增殖和分化[91]。
RASTEGARI等[92]对Ti6Al4V进行了碱处理和壳聚糖-二氧化硅纳米复合涂层处理,纳米复合涂层不仅提高了Ti6Al4V合金的生物性能,还增强了基底的表面湿润性、磷灰石形成能力、腐蚀保护、细胞活力和细胞黏附性。PALLA-RUBIO等[93]介绍了溶胶-凝胶法合成硅-壳聚糖杂化膜的方法,体外细胞培养分析表明,杂化膜对细胞无毒并能促进细胞增殖,其杂化膜中的壳聚糖不仅可以提高抗菌性,而且调节了硅的释放,对骨再生也起着重要的作用。
除壳聚糖外,透明质酸也是一种天然多糖,在生物反应启动中起着重要作用。VALVERDE等[94]通过逐层沉积将透明质酸和壳聚糖搭载到Ti6Al4V表面,对其进行了表面改性,实验结果表明,这种涂层不仅增加了钛植入物的抗菌性能,也提高了植入手术的成功率。
2.5 核苷酸类
2.5.1 siRNA 核糖核酸干扰技术是一种通过沉默特定基因来调节基因表达的理想方法,被广泛应用于靶向治疗或植入物表面修饰的研究[95]。越来越多的研究表明,功能性生物涂层如siRNA涂层、miRNA涂层,可以通过促进成骨细胞的增殖、分化和黏附来改善钛种植体的表面生物相容性[96]。此技术不仅可以提高植入成功率、增加植入体的稳定性、改善植入体周围软组织的整合,还可以通过其抗菌特性减少植入体周围的炎症[97]。
研究发现,抑制组织蛋白酶K可有效抑制破骨细胞分化,增加血小板源性生长因子水平,促进成骨的血管生成[98]。XING等[95]开发了一种新的分级纳米结构,用作临床使用的钛植入物表面涂层,将靶向调节组织蛋白酶K的siRNA通过明胶涂覆在金纳米粒子上,形成金纳米粒子-siRNA-抑制组织蛋白酶K,再将修饰过的金纳米粒子再一次通过明胶逐层沉积构建在钛种植体表面上,产生功能化纳米粒子的多层结构,植入体-骨界面上siRNA-抑制组织蛋白酶K修饰的金纳米粒子的受控释放使得基因沉默过程更加精确,不仅用于抑制破骨细胞分化,还用于刺激血管生成和成骨。
siRNA还能调节炎症反应,在炎症因子中最有效的是肿瘤坏死因子α,它在局部参与了骨关节炎中软骨的损伤,并且血清中肿瘤坏死因子α水平可用于提示骨关节炎的进展。WANG等[99]通过对钛表面进行阳极氧化处理和壳聚糖/三聚磷酸盐-透明质酸钠纳米颗粒将siRNA负载在植入体钛表面,形成壳聚糖/三聚磷酸盐-透明质酸钠-siRNA纳米颗粒,实验建立了小鼠间充质干细胞和小鼠单核细胞系RAW264.7细胞的间接共培养体系,将预先激活的RAW264.7细胞置于transwell小室的上室,将siRNA修饰的植入钛置于下室,以评估siRNA对肿瘤坏死因子α的调控以及对成骨分化的影响,研究证实,活化RAW264.7细胞分泌的肿瘤坏死因子α可显著影响间充质干细胞的成骨分化,通过siRNA修饰的钛表面肿瘤坏死因子α远距离下调,炎症因子解除后骨髓间充质干细胞的成骨分化得到改善。siRNA是不稳定的生物活性物质,其应用仍然有限。siRNA的应用需要有效的递送系统和载体,如用于递送siRNA的纳米颗粒等。通过负载siRNA进行表面修饰后,钛及其合金可以促进相关成骨细胞或干细胞的附着和成骨分化,并促进相关成骨标记物的表达[23, 100]。
2.5.2 miRNA miRNAs是内源性非编码单链RNA,长度约为22个核苷酸,在细胞发育、增殖、分化、凋亡和信号转导等多样的基础生物学过程中发挥重要作用。miRNAs模拟自然分化途径,并控制多个基因,构成一个更完善的干细胞分化刺激因子。近年来,关于miRNAs对成骨的调控作用的报道不断增多[101],其中miR-138被报道为间充质干细胞成骨分化的负调控因子,Anti- miR-138抑制内源性miR-138水平可增强体内的骨形成[102]。
WU等[16]将壳聚糖-AntimiR-138 配合物和透明质酸钠作为带正电荷和负电荷的聚电解质,采用分层方法制备聚电解质多层膜,再通过硅烷化法将聚电解质多层膜共价结合在经过微弧氧化的钛表面,体外转染结果显示壳聚糖-AntimiR-138纳米颗粒被细胞有效吸收,并导致miR-138显著下调,但未显示出显著的细胞毒性,壳聚糖-AntimiR-138/透明质酸钠聚电解质多层膜表面增强了间充质干细胞的成骨分化和碱性磷酸酶、胶原生成和细胞外基质矿化,在大鼠模型中观察到体内骨结合显著增强。这种新型miRNA功能化钛种植体可用于临床,以实现更有效、更稳定的骨整合。
此外,miR-21也已被证明与骨骼发育有关,参与多种细胞功能的调节[103]。GENG等[104]将钛种植体依次通过酸处理、锶磷灰石沉积和miR-21纳米胶囊固定进行改性,这种涂层不仅能促进血管生成因子CD31的表达,还能增强成骨细胞基因的表达,从而促进成血管和成骨。

| [1] EASTELL R, O’NEILL TW, HOFBAUER LC, et al. Postmenopausal osteoporosis. Nat Rev Dis Primers. 2016;2:16069. [2] HO-SHUI-LING A, BOLANDER J, RUSTOM LE, et al. Bone regeneration strategies: Engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials. 2018;180:143-162. [3] BALDWIN P, LI DJ, AUSTON DA, et al. Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery. J Orthop Trauma. 2019;33(4): 203-213. [4] GOODMAN SB, YAO Z, KEENEY M, et al. The future of biologic coatings for orthopaedic implants. Biomaterials. 2013;34(13):3174-3183. [5] AGARWAL R, GARCíA AJ. Biomaterial strategies for engineering implants for enhanced osseointegration and bone repair. Adv Drug Deliv Rev. 2015;94:53-62. [6] LARSSON C, THOMSEN P, ARONSSON BO, et al. Bone response to surface-modified titanium implants: studies on the early tissue response to machined and electropolished implants with different oxide thicknesses. Biomaterials. 1996;17(6):605-616. [7] CHEN L, BAI M, DU R, et al. The non-viral vectors and main methods of loading siRNA onto the titanium implants and their application. J Biomater Sci Polym Ed. 2020;31(16):2152-2168. [8] HE Y, MU C, SHEN X, et al. Peptide LL-37 coating on micro-structured titanium implants to facilitate bone formation in vivo via mesenchymal stem cell recruitment. Acta Biomater. 2018;80:412-424. [9] DARD M, SEWING A, MEYER J, et al. Tools for tissue engineering of mineralized oral structures. Clin Oral Investig. 2000;4(2):126-129. [10] IWATA N, NOZAKI K, HORIUCHI N, et al. Effects of controlled micro-/nanosurfaces on osteoblast proliferation. J Biomed Mater Res A. 2017; 105(9):2589-2596. [11] YU Y, JIN G, XUE Y, et al. Multifunctions of dual Zn/Mg ion co-implanted titanium on osteogenesis, angiogenesis and bacteria inhibition for dental implants. Acta Biomater. 2017;49:590-603. [12] LEE S, CHANG YY, LEE J, et al. Surface engineering of titanium alloy using metal-polyphenol network coating with magnesium ions for improved osseointegration. Biomater Sci. 2020;8(12):3404-3417. [13] KILIAN KA, BUGARIJA B, LAHN BT, et al. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc Natl Acad Sci U S A. 2010;107(11):4872-4877. [14] LUTZ R, SROUR S, NONHOFF J, et al. Biofunctionalization of titanium implants with a biomimetic active peptide (P-15) promotes early osseointegration. Clin Oral Implants Res. 2010;21(7):726-734. [15] CHANG YC, FENG SW, HUANG HM, et al. Surface analysis of titanium biological modification with glow discharge. Clin Implant Dent Relat Res. 2015;17(3):469-475. [16] WU K, LIU M, LI N, et al. Chitosan-miRNA functionalized microporous titanium oxide surfaces via a layer-by-layer approach with a sustained release profile for enhanced osteogenic activity. J Nanobiotechnology. 2020;18(1):127. [17] JURCZAK P, WITKOWSKA J, RODZIEWICZ-MOTOWIDłO S, et al. Proteins, peptides and peptidomimetics as active agents in implant surface functionalization. Adv Colloid Interface Sci. 2020;276:102083. [18] HELLER M, KUMAR VV, PABST A, et al. Osseous response on linear and cyclic RGD-peptides immobilized on titanium surfaces in vitro and in vivo. J Biomed Mater Res A. 2018;106(2):419-427. [19] PARFENOVA LV, LUKINA ES, GALIMSHINA ZR, et al. Biocompatible Organic Coatings Based on Bisphosphonic Acid RGD-Derivatives for PEO-Modified Titanium Implants. Molecules. 2020;25(1):229. [20] CROES M, AKHAVAN B, SHARIFAHMADIAN O, et al. A multifaceted biomimetic interface to improve the longevity of orthopedic implants. Acta Biomater. 2020;110:266-279. [21] KäMMERER PW, PABST AM, DAU M, et al. Immobilization of BMP-2, BMP-7 and alendronic acid on titanium surfaces: Adhesion, proliferation and differentiation of bone marrow-derived stem cells. J Biomed Mater Res A. 2020;108(2):212-220. [22] GURZAWSKA K, SVAVA R, JøRGENSEN NR, et al. Nanocoating of titanium implant surfaces with organic molecules. Polysaccharides including glycosaminoglycans. J Biomed Nanotechnol. 2012;8(6):1012-1024. [23] SAKURAI T, YOSHINARI M, TOYAMA T, et al. Effects of a multilayered DNA/protamine coating on titanium implants on bone responses. J Biomed Mater Res A. 2016;104(6):1500-1509. [24] SONG W, YANG C, SVEND LE DQ, et al. Calcium-MicroRNA Complex-Functionalized Nanotubular Implant Surface for Highly Efficient Transfection and Enhanced Osteogenesis of Mesenchymal Stem Cells. ACS Appl Mater Interfaces. 2018;10(9):7756-7764. [25] SONG E, YEON KIM S, CHUN T, et al. Collagen scaffolds derived from a marine source and their biocompatibility. Biomaterials. 2006;27(15):2951-2961. [26] SVERZUT AT, CRIPPA GE, MORRA M, et al. Effects of type I collagen coating on titanium osseointegration: histomorphometric, cellular and molecular analyses. Biomed Mater. 2012;7(3):035007. [27] SCARANO A, LORUSSO F, ORSINI T, et al. Biomimetic Surfaces Coated with Covalently Immobilized Collagen Type I: An X-Ray Photoelectron Spectroscopy, Atomic Force Microscopy, Micro-CT and Histomorphometrical Study in Rabbits. Int J Mol Sci. 2019;20(3):724. [28] ZHANG X, XU L, HUANG X, et al. Structural study and preliminary biological evaluation on the collagen hydrogel crosslinked by γ-irradiation. J Biomed Mater Res A. 2012;100(11):2960-2969. [29] LUO X, GUO Z, HE P, et al. Study on structure, mechanical property and cell cytocompatibility of electrospun collagen nanofibers crosslinked by common agents. Int J Biol Macromol. 2018;113:476-486. [30] BAE EB, YOO JH, JEONG SI, et al. Effect of Titanium Implants Coated with Radiation-Crosslinked Collagen on Stability and Osseointegration in Rat Tibia. Materials (Basel). 2018;11(12):2520. [31] MAKISHI S, SAITO K, OHSHIMA H. Osteopontin-deficiency disturbs direct osteogenesis in the process of achieving osseointegration following immediate placement of endosseous implants. Clin Implant Dent Relat Res. 2017;19(3):496-504. [32] FIORELLINI JP, GLINDMANN S, SALCEDO J, et al. The Effect of Osteopontin and an Osteopontin-Derived Synthetic Peptide Coating on Osseointegration of Implants in a Canine Model. Int J Periodontics Restorative Dent. 2016;36(6):e88-88e94. [33] RAPUANO BE, HACKSHAW KM, SCHNIEPP HC, et al. Effects of coating a titanium alloy with fibronectin on the expression of osteoblast gene markers in the MC3T3 osteoprogenitor cell line. Int J Oral Maxillofac Implants. 2012;27(5):1081-1090. [34] PANKOV R, YAMADA KM. Fibronectin at a glance. J Cell Sci. 2002; 115(Pt 20):3861-3863. [35] PRAMONO S, PUGDEE K, SUWANPRATEEP J, et al. Sandblasting and fibronectin-derived peptide immobilization on titanium surface increase adhesion and differentiation of osteoblast-like cells (MC3T3-E1). J Dent Sci. 2016;11(4):427-436. [36] RIVERA-CHACON DM, ALVARADO-VELEZ M, ACEVEDO-MORANTES CY, et al. Fibronectin and vitronectin promote human fetal osteoblast cell attachment and proliferation on nanoporous titanium surfaces. J Biomed Nanotechnol. 2013;9(6):1092-1097. [37] FRANZ S, RAMMELT S, SCHARNWEBER D, et al. Immune responses to implants - a review of the implications for the design of immunomodulatory biomaterials. Biomaterials. 2011;32(28):6692-6709. [38] SPEZIALE P, VISAI L, RINDI S, et al. Purification of human plasma fibronectin using immobilized gelatin and Arg affinity chromatography. Nat Protoc. 2008;3(3):525-533. [39] GUILLEM-MARTI J, BOIX-LEMONCHE G, GUGUTKOV D, et al. Recombinant fibronectin fragment III8-10/polylactic acid hybrid nanofibers enhance the bioactivity of titanium surface. Nanomedicine (Lond). 2018;13(8):899-912. [40] GUILLEM-MARTI J, GELABERT M, HERAS-PARETS A, et al. RGD Mutation of the Heparin Binding II Fragment of Fibronectin for Guiding Mesenchymal Stem Cell Behavior on Titanium Surfaces. ACS Appl Mater Interfaces. 2019;11(4):3666-3678. [41] LAI M, JIN Z, SU Z. Surface modification of TiO(2) nanotubes with osteogenic growth peptide to enhance osteoblast differentiation. Mater Sci Eng C Mater Biol Appl. 2017;73:490-497. [42] POLICASTRO GM, LIN F, SMITH CALLAHAN LA, et al. OGP functionalized phenylalanine-based poly(ester urea) for enhancing osteoinductive potential of human mesenchymal stem cells. Biomacromolecules. 2015;16(4):1358-1371. [43] MOORE NM, LIN NJ, GALLANT ND, et al. The use of immobilized osteogenic growth peptide on gradient substrates synthesized via click chemistry to enhance MC3T3-E1 osteoblast proliferation. Biomaterials. 2010;31(7):1604-1611. [44] LIU J, TANG Y, YANG W, et al. Functionalization of titanium substrate with multifunctional peptide OGP-NAC for the regulation of osteoimmunology. Biomater Sci. 2019;7(4):1463-1476. [45] WATANABE J, YAMADA M, NIIBE K, et al. Preconditioning of bone marrow-derived mesenchymal stem cells with N-acetyl-L-cysteine enhances bone regeneration via reinforced resistance to oxidative stress. Biomaterials. 2018;185:25-38. [46] MOUNTZIARIS PM, SPICER PP, KASPER FK, et al. Harnessing and modulating inflammation in strategies for bone regeneration. Tissue Eng Part B Rev. 2011;17(6):393-402. [47] CHEN X, ZHOU L, WU D, et al. The Effects of Titanium Surfaces Modified with an Antimicrobial Peptide GL13K by Silanization on Polarization, Anti-Inflammatory, and Proinflammatory Properties of Macrophages. Biomed Res Int. 2020;2020:2327034. [48] ZHANG W, ZHAO F, HUANG D, et al. Strontium-Substituted Submicrometer Bioactive Glasses Modulate Macrophage Responses for Improved Bone Regeneration. ACS Appl Mater Interfaces. 2016; 8(45):30747-30758. [49] LIU P, ZHAO Y, YUAN Z, et al. Construction of Zn-incorporated multilayer films to promote osteoblasts growth and reduce bacterial adhesion. Mater Sci Eng C Mater Biol Appl. 2017;75: 998-1005. [50] LIU Z, ZHU Y, LIU X, et al. Construction of poly (vinyl alcohol)/poly (lactide-glycolide acid)/vancomycin nanoparticles on titanium for enhancing the surface self-antibacterial activity and cytocompatibility. Colloids Surf B Biointerfaces. 2017;151:165-177. [51] TAHERI S, CAVALLARO A, CHRISTO SN, et al. Substrate independent silver nanoparticle based antibacterial coatings. Biomaterials. 2014; 35(16):4601-4609. [52] BADAR M, RAHIM MI, KIEKE M, et al. Controlled drug release from antibiotic-loaded layered double hydroxide coatings on porous titanium implants in a mouse model. J Biomed Mater Res A. 2015;103(6):2141-2149. [53] CAMPOCCIA D, MONTANARO L, SPEZIALE P, et al. Antibiotic-loaded biomaterials and the risks for the spread of antibiotic resistance following their prophylactic and therapeutic clinical use. Biomaterials. 2010;31(25):6363-6377. [54] CRUZ J, ORTIZ C, GUZMáN F, et al. Antimicrobial peptides: promising compounds against pathogenic microorganisms. Curr Med Chem. 2014;21(20):2299-2321. [55] XHINDOLI D, PACOR S, BENINCASA M, et al. The human cathelicidin LL-37--A pore-forming antibacterial peptide and host-cell modulator. Biochim Biophys Acta. 2016;1858(3):546-566. [56] HE Y, ZHANG Y, SHEN X, et al. The fabrication and in vitro properties of antibacterial polydopamine-LL-37-POPC coatings on micro-arc oxidized titanium. Colloids Surf B Biointerfaces. 2018;170:54-63. [57] COFFELT SB, MARINI FC, WATSON K, et al. The pro-inflammatory peptide LL-37 promotes ovarian tumor progression through recruitment of multipotent mesenchymal stromal cells. Proc Natl Acad Sci U S A. 2009;106(10):3806-3811. [58] LI T, WANG N, CHEN S, et al. Antibacterial activity and cytocompatibility of an implant coating consisting of TiO(2) nanotubes combined with a GL13K antimicrobial peptide. Int J Nanomedicine. 2017;12:2995-3007. [59] ZHANG G, ZHANG X, YANG Y, et al. Dual light-induced in situ antibacterial activities of biocompatibleTiO(2)/MoS(2)/PDA/RGD nanorod arrays on titanium. Biomater Sci. 2020;8(1):391-404. [60] XU D, YANG W, HU Y, et al. Surface functionalization of titanium substrates with cecropin B to improve their cytocompatibility and reduce inflammation responses. Colloids Surf B Biointerfaces. 2013;110:225-235. [61] HASAN J, CRAWFORD RJ, IVANOVA EP. Antibacterial surfaces: the quest for a new generation of biomaterials. Trends Biotechnol. 2013; 31(5):295-304. [62] WANG F, HOU K, CHEN W, et al. Transgenic PDGF-BB/sericin hydrogel supports for cell proliferation and osteogenic differentiation. Biomater Sci. 2020;8(2):657-672. [63] LYNCH SE, BUSER D, HERNANDEZ RA, et al. Effects of the platelet-derived growth factor/insulin-like growth factor-I combination on bone regeneration around titanium dental implants. Results of a pilot study in beagle dogs. J Periodontol. 1991;62(11):710-716. [64] ORTOLANI E, GUERRIERO M, COLI A, et al. Effect of PDGF, IGF-1 and PRP on the implant osseointegration. An histological and immunohistochemical study in rabbits. Ann Stomatol (Roma). 2014; 5(2):66-68. [65] KIM SE, YUN YP, LEE JY, et al. Co-delivery of platelet-derived growth factor (PDGF-BB) and bone morphogenic protein (BMP-2) coated onto heparinized titanium for improving osteoblast function and osteointegration. J Tissue Eng Regen Med. 2015;9(12):E219-228. [66] MA Q, JIANG N, LIANG S, et al. Functionalization of a clustered TiO(2) nanotubular surface with platelet derived growth factor-BB covalent modification enhances osteogenic differentiation of bone marrow mesenchymal stem cells. Biomaterials. 2020;230:119650. [67] RUMNEY R, LANHAM SA, KANCZLER JM, et al. In vivo delivery of VEGF RNA and protein to increase osteogenesis and intraosseous angiogenesis. Sci Rep. 2019;9(1):17745. [68] DIOMEDE F, MARCONI GD, CAVALCANTI M, et al. VEGF/VEGF-R/RUNX2 Upregulation in Human Periodontal Ligament Stem Cells Seeded on Dual Acid Etched Titanium Disk. Materials (Basel). 2020;13(3):706. [69] GROSSO A, BURGER MG, LUNGER A, et al. It Takes Two to Tango: Coupling of Angiogenesis and Osteogenesis for Bone Regeneration. Front Bioeng Biotechnol. 2017;5:68. [70] IZQUIERDO-BARBA I, SANTOS-RUIZ L, BECERRA J, et al. Synergistic effect of Si-hydroxyapatite coating and VEGF adsorption on Ti6Al4V-ELI scaffolds for bone regeneration in an osteoporotic bone environment. Acta Biomater. 2019;83:456-466. [71] FUJIOKA-KOBAYASHI M, SCHALLER B, SAULACIC N, et al. Absorbable collagen sponges loaded with recombinant bone morphogenetic protein 9 induces greater osteoblast differentiation when compared to bone morphogenetic protein 2. Clin Exp Dent Res. 2017;3(1):32-40. [72] FUJIOKA-KOBAYASHI M, ABD EL RAOUF M, SAULACIC N, et al. Superior bone-inducing potential of rhBMP9 compared to rhBMP2. J Biomed Mater Res A. 2018;106(6):1561-1574. [73] FISHERO BA, KOHLI N, DAS A, et al. Current concepts of bone tissue engineering for craniofacial bone defect repair. Craniomaxillofac Trauma Reconstr. 2015;8(1):23-30. [74] WANG X, LI B, ZHANG C. Preparation of BMP-2/chitosan/hydroxyapatite antibacterial bio-composite coatings on titanium surfaces for bone tissue engineering. Biomed Microdevices. 2019;21(4):89. [75] TENG FY, TAI IC, HO ML, et al. Controlled release of BMP-2 from titanium with electrodeposition modification enhancing critical size bone formation. Mater Sci Eng C Mater Biol Appl. 2019;105:109879. [76] CHEN T, WANG S, HE F, et al. Promotion of Osseointegration Using Protamine/Alginate/Bone Morphogenic Protein 2 Biofunctionalized Composite Coating on Nanopolymorphic Titanium Surfaces. J Biomed Nanotechnol. 2018;14(5):933-945. [77] YANG DH, MOON SW, LEE DW. Surface Modification of Titanium with BMP-2/GDF-5 by a Heparin Linker and Its Efficacy as a Dental Implant. Int J Mol Sci. 2017;18(1):229. [78] CHIARI C, GRGUREVIC L, BORDUKALO-NIKSIC T, et al. Recombinant Human BMP6 Applied Within Autologous Blood Coagulum Accelerates Bone Healing: Randomized Controlled Trial in High Tibial Osteotomy Patients. J Bone Miner Res. 2020;35(10):1893-1903. [79] BRIGAUD I, AGNIEL R, LEROY-DUDAL J, et al. Synergistic effects of BMP-2, BMP-6 or BMP-7 with human plasma fibronectin onto hydroxyapatite coatings: A comparative study. Acta Biomater. 2017;55:481-492. [80] KECELI HG, BAYRAM C, CELIK E, et al. Dual delivery of platelet-derived growth factor and bone morphogenetic factor-6 on titanium surface to enhance the early period of implant osseointegration. J Periodontal Res. 2020;55(5):694-704. [81] CHEN R, YU J, GONG HL, et al. An easy long-acting BMP7 release system based on biopolymer nanoparticles for inducing osteogenic differentiation of adipose mesenchymal stem cells. J Tissue Eng Regen Med. 2020;14(7):964-972. [82] ZHANG F, REN LF, LIN HS, et al. The optimal dose of recombinant human osteogenic protein-1 enhances differentiation of mouse osteoblast-like cells: an in vitro study. Arch Oral Biol. 2012;57(5):460-468. [83] MATHAVAN N, RAINA DB, TäGIL M, et al. Longitudinal in vivo monitoring of callus remodeling in BMP-7- and Zoledronate-treated fractures. J Orthop Res. 2020;38(9):1905-1913. [84] AL-JARSHA M, MOULISOVá V, LEAL-EGAñA A, et al. Engineered Coatings for Titanium Implants To Present Ultralow Doses of BMP-7. ACS Biomater Sci Eng. 2018;4(5):1812-1819. [85] SOUZA A, BEZERRA B, OLIVEIRA FS, et al. Effect of bone morphogenetic protein 9 on osteoblast differentiation of cells grown on titanium with nanotopography. J Cell Biochem. 2018;119(10):8441-8449. [86] HUANG X, WANG F, ZHAO C, et al. Dentinogenesis and Tooth-Alveolar Bone Complex Defects in BMP9/GDF2 Knockout Mice. Stem Cells Dev. 2019;28(10):683-694. [87] VHORA I, LALANI R, BHATT P, et al. Lipid-nucleic acid nanoparticles of novel ionizable lipids for systemic BMP-9 gene delivery to bone-marrow mesenchymal stem cells for osteoinduction. Int J Pharm. 2019;563:324-336. [88] NEILL T, SCHAEFER L, IOZZO RV. Decorin: a guardian from the matrix. Am J Pathol. 2012;181(2):380-387. [89] MOHAN RR, GUPTA R, MEHAN MK, et al. Decorin transfection suppresses profibrogenic genes and myofibroblast formation in human corneal fibroblasts. Exp Eye Res. 2010;91(2):238-245. [90] HE R, LU Y, REN J, et al. Decreased fibrous encapsulation and enhanced osseointegration in vitro by decorin-modified titanium surface. Colloids Surf B Biointerfaces. 2017;155:17-24. [91] KARIMI S, SALAHINEJAD E, SHARIFI E, et al. Bioperformance of chitosan/fluoride-doped diopside nanocomposite coatings deposited on medical stainless steel. Carbohydr Polym. 2018;202:600-610. [92] RASTEGARI S, SALAHINEJAD E. Surface modification of Ti-6Al-4V alloy for osseointegration by alkaline treatment and chitosan-matrix glass-reinforced nanocomposite coating. Carbohydr Polym. 2019;205:302-311. [93] PALLA-RUBIO B, ARAúJO-GOMES N, FERNáNDEZ-GUTIéRREZ M, et al. Synthesis and characterization of silica-chitosan hybrid materials as antibacterial coatings for titanium implants. Carbohydr Polym. 2019;203:331-341. [94] VALVERDE A, PéREZ-ÁLVAREZ L, RUIZ-RUBIO L, et al. Antibacterial hyaluronic acid/chitosan multilayers onto smooth and micropatterned titanium surfaces. Carbohydr Polym. 2019;207:824-833. [95] XING H, WANG X, XIAO G, et al. Hierarchical assembly of nanostructured coating for siRNA-based dual therapy of bone regeneration and revascularization. Biomaterials. 2020;235:119784. [96] JENNY G, JAUERNIK J, BIERBAUM S, et al. A systematic review and meta-analysis on the influence of biological implant surface coatings on periimplant bone formation. J Biomed Mater Res A. 2016;104(11): 2898-2910. [97] HäGI TT, ENGGIST L, MICHEL D, et al. Mechanical insertion properties of calcium-phosphate implant coatings. Clin Oral Implants Res. 2010; 21(11):1214-1222. [98] KUSUMBE AP, ADAMS RH. Osteoclast progenitors promote bone vascularization and osteogenesis. Nat Med. 2014;20(11):1238-1240. [99] WANG Z, HU Z, ZHANG D, et al. Silencing tumor necrosis factor-alpha in vitro from small interfering RNA-decorated titanium nanotube array can facilitate osteogenic differentiation of mesenchymal stem cells. Int J Nanomedicine. 2016;11:3205-3214. [100] HUANG Y, ZHENG Y, XU Y, et al. Titanium Surfaces Functionalized with siMIR31HG Promote Osteogenic Differentiation of Bone Marrow Mesenchymal Stem Cells. ACS Biomater Sci Eng. 2018;4(8):2986-2993. [101] XU T, LUO Y, WANG J, et al. Exosomal miRNA-128-3p from mesenchymal stem cells of aged rats regulates osteogenesis and bone fracture healing by targeting Smad5. J Nanobiotechnology. 2020;18(1):47. [102] ESKILDSEN T, TAIPALEENMäKI H, STENVANG J, et al. MicroRNA-138 regulates osteogenic differentiation of human stromal (mesenchymal) stem cells in vivo. Proc Natl Acad Sci U S A. 2011;108(15):6139-6144. [103] SMIESZEK A, MARCINKOWSKA K, PIELOK A, et al. The Role of miR-21 in Osteoblasts-Osteoclasts Coupling In Vitro. Cells. 2020;9(2):479. [104] GENG Z, WANG X, ZHAO J, et al. The synergistic effect of strontium-substituted hydroxyapatite and microRNA-21 on improving bone remodeling and osseointegration. Biomater Sci. 2018;6(10):2694-2703. |
| [1] | 薛亚东, 周新社, 裴立家, 孟繁宇, 李 键, 王金子. 自体髂骨块联合钛板重建Paprosky Ⅲ型髋臼骨缺损为假体提供坚强的初始固定#br#[J]. 中国组织工程研究, 2022, 26(9): 1424-1428. |
| [2] | 梁学振, 杨 曦, 李嘉程, 骆 帝, 许 波, 李 刚. 补肾活血胶囊介导Hedgehog信号通路调控大鼠骨髓间充质干细胞成骨成脂分化[J]. 中国组织工程研究, 2022, 26(7): 1020-1026. |
| [3] | 袁 景, 孙效虎, 陈 慧, 乔永杰, 王立新. 成人继发性膝外翻畸形股骨远端的数字化测量与分析[J]. 中国组织工程研究, 2022, 26(6): 881-885. |
| [4] | 李 键, 鲍正齐, 周平辉, 朱瑞直, 李志想, 王金子. 颈椎后路单开门椎板成形与颈椎体次全切除植骨融合治疗多节段颈椎病对颈椎矢状位平衡参数的影响[J]. 中国组织工程研究, 2022, 26(6): 949-953. |
| [5] | 彭 坤. 骨质疏松性骨折治疗效果的改善:研究现状及策略分析[J]. 中国组织工程研究, 2022, 26(6): 980-984. |
| [6] | 胡维帆, 郑 力, 李大地, 孙 阳, 赵凤朝. 过表达miR-25通过 NFATc1信号通路调控钛颗粒诱导的破骨细胞分化[J]. 中国组织工程研究, 2022, 26(5): 682-687. |
| [7] | 邱 鹏, 傅琪淋, 刘 敏, 兰玉燕, 王 频. 聚醚醚酮、二氧化锆和纯钛基台材料表面口腔微生物黏附的对比[J]. 中国组织工程研究, 2022, 26(4): 540-545. |
| [8] | 陈 硕, 肖东琴, 李兴平, 冉 斌, 匙 峰, 张成栋, 邓 丽, 黄南翔, 刘 康, 冯 刚, 段 可. 钛植入体表面钽功能涂层制备及性能表征[J]. 中国组织工程研究, 2022, 26(4): 546-552. |
| [9] | 杨 峰, 赵 骞, 张世轩, 赵铁男, 冯 博. 雷帕霉素联合CD133抗体支架预防血管再狭窄的有效性和安全性[J]. 中国组织工程研究, 2022, 26(4): 579-584. |
| [10] | 康坤龙, 王新涛. 生物支架材料促进骨髓间充质干细胞成骨分化的研究热点[J]. 中国组织工程研究, 2022, 26(4): 597-603. |
| [11] | 李 辉, 陈良龙. 植骨材料在脊柱结核治疗中的应用与特征[J]. 中国组织工程研究, 2022, 26(4): 626-630. |
| [12] | 李 路, 唐 文. 下颈椎后路椎弓根钉置钉:人工智能发展将如何提高治疗效果[J]. 中国组织工程研究, 2022, 26(3): 474-479. |
| [13] | 李 凯, 刘振东, 李小磊, 王静成. 后外侧入路全髋关节置换后假体反复性脱位的危险因素[J]. 中国组织工程研究, 2022, 26(3): 354-358. |
| [14] | 曹 炜, 冒符荣, 胡小华, 杨晓红. N-6甲基腺嘌呤RNA甲基化调控骨髓间充质干细胞的成骨及成脂分化[J]. 中国组织工程研究, 2022, 26(2): 266-270. |
| [15] | 张红梅, 孙溪饶, 王程越. 聚甲基丙烯酸甲酯/矿化胶原/Mg-Ca合金复合材料与小鼠前成骨细胞的成骨分化[J]. 中国组织工程研究, 2022, 26(16): 2498-2503. |
据研究报道,良好的骨整合是钛种植体与骨界面长期结合的关键[7],但钛种植体周围的骨形成不足是制约着它们应用的原因[8]。为了实现更好的骨整合,各种研究都试图对钛种植体表面进行改良[9]。改变表面性能(如微、亚微和纳米尺度的形貌和湿润性[10])和在支架表面搭载无机物等传统的植入修饰技术,对提高成骨活性较为有限[11-13],这种基于物理和化学的表面修饰方式主要是间接影响细胞行为,而将生物分子固定在钛种植体表面的生物化学改性可以直接参与生物过程,这在诱导骨形成方面更有效[14-15], 尤其是在骨状况不佳的情况下[16]。
目前常用的生物分子有细胞外基质蛋白[17]、多肽[18-19]、生长因子[20-21]、多糖和核苷酸等[22-24]。因此,许多研究采用种植体表面生物化学涂层的方法来促进种植体周围骨再生,以改善骨质结构并提高种植体骨结合率。文章针对钛种植体表面的5种生物化学改性对种植体周围骨结合影响的研究现状与进展进行综述。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
1.2 入选与排除标准
纳入标准:①与钛植入物表面生物化学改性密切相关的文章;②与生物活性物质对种植体骨整合产生积极作用的文章;③同一领域的相关研究选择近年发表的文献。
排除标准:重复性研究;相关性差及无关文章。
1.3 资料提取 根据检索词共检索到297篇相关文献,阅读标题和摘要,按照纳入及排除标准筛选后最终共纳入104篇文献进行分析和整理。文献检索流程图,见图1。
钛或钛合金植入物经过微弧氧化、喷砂、酸蚀、水热法、等离子喷涂、电化学沉积等物理或化学改性的方法在植入物表面制备氧化膜或者粗糙表面,这种理化改性可以改变钛或钛合金植入物表面的湿润度、亲水性、电荷、形貌等,也为生物活性分子的搭载提供了条件。同时,在此基础上将二氧化钛纳米管、钙磷涂层、羟基磷灰石、明胶、壳聚糖、肝素或聚多巴胺等作为载体,通过静电纺丝、简单涂敷、物理吸附、硅烷化法或层层自组装技术等不同方法将生物活性分子搭载到钛植入物表面,使其在骨与钛植入物之间形成一层具有一定生物活性的涂层。通过这些方法将细胞外基质蛋白、活性肽、生长因子、多糖和核苷酸搭载到钛植入物表面,直接参与生物过程的生物化学改性主要有3个方面的优势:①首先,比单一的物理和化学改性具有更高的仿生性和更确切的成骨效果;②其次,在体外细胞学评价中生物活性分子可以直接调节成骨细胞的黏附、增殖、迁移和分化,并增加碱性磷酸酶活性和细胞外基质的矿化,且进一步的分子和基因水平显示,活化的钛植入物可以诱导细胞发育相关转录因子2、骨钙素和骨涎蛋白等成骨相关基因的表达;纤维连接蛋白还展现出了良好的因子结合能力,成骨生长肽除了促进成骨细胞的黏附和成骨外,还可以抑制破骨细胞的表达;核心蛋白多糖可以诱导成纤维细胞凋亡和调节纤维结构,从而抑制纤维包封;③最后,体内实验的组织学及Micro-CT等结果显示,经过生物化学改性的钛植入物可以显著提高骨与植入物的结合率,并且加速早期成骨。此外,抗菌肽和核苷酸等的修饰还可以改善骨整合过程中另一重要的炎症反应,经过抗菌修饰的钛植入物可以抑制植入手术的早期炎症反应及种植体周围的慢性炎症,从而降低因炎症反应引起的手术失败和假体翻修概率,增加了手术的可预测性,降低了其风险。
目前要将这种钛植入物表面的修饰技术大量应用于临床,还有4个问题需要解决:首先,生物活性分子在钛植入物表面的结合强度较低时,会导致涂层的松动或者在植入体内过程中存在涂层因摩擦而大量磨损消耗的问题;其次,生物活性分子搭载后的完整性、微观结构和天然构象是否发生了改变,以及经过修饰后的钛植入物如何在表面干燥的情况下长期保存;再者,生长因子的搭载量、释放量、释放率和释放时间都难以控制,都需要进一步优化而达到适量、均一、缓慢、长期的释放效果;最后,在钛植入物表面搭载多种物质而制备的复合涂层中的某一种物质会存在降解问题,比如含有壳聚糖的复合涂层在壳聚糖完全降解时其抗菌性能就会消失,这样难以维持复合涂层的长期稳定性。此外,在复合涂层的多种物质中存在着协同或者拮抗的作用,这难以控制该复合涂层中的每一种物质都达到最适的搭载量。除了这些问题,有些实验的结果显示出体内和体外研究结果的冲突和分歧,这都亟需深入探讨。
随着植入体和生物材料的不断发展,在今后会发现更加理想的搭载方法以及生物活性物质,从而解决目前存在的问题。首先,将物理、化学和生物化学改性更加有机地结合在一起,从而使生物活性物质产生更强的附着力,降低在植入过程中因摩擦而产生的损耗;其次,在已经用生物活性分子修饰过的钛植入物表面再覆以磷脂或其他成分的外层包衣,这不但可以维持复合涂层的完整性,还可以延长其保存时间;再者,将生物活性物质作为3D打印的材料,使其可以长期、稳定、均匀地释放,从而达到植入物的长期稳定性;最后,理想的生物活性物质或涂层不但可以促进骨整合,而且还应该具有抗炎等作用,所以将具有不同作用的生物活性分子共同搭载到同一钛植入体表面,或者通过基因水平设计出同时满足既抗菌又可以促进成血管成骨的复合涂层。此外,还可以通过基因设计出产生靶向作用的物质,从而使整个骨整合过程更加可预测。这些理想的搭载方法以及生物活性物质,一方面可以增强种植体植入的早期和长期稳定性,另一方面可以防止种植体周围感染,并且满足临床应用的可行性。然而,对于骨与植入体的生物相容性在分子甚至基因水平的研究,以及涂层对骨整合的具体调控机制等问题还需要不断探索,早日实现种植体与骨组织之间的牢固、持久而直接的结合。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程

文题释义:#br# 钛植入物骨整合:是指在人体活的骨组织与钛种植体之间发生的牢固、持久而直接的结合,即负载力量的种植体的表面与有活力的骨组织之间存在结构上和功能上直接的联系,种植体与骨组织之间没有任何结缔组织存在,不间隔任何组织。#br# 生物化学改性:起初是作为化学改性的变异出现,是指通过将特定的蛋白、多肽、生长因子、多糖和核苷酸固定于种植体表面,这样可使生物活性分子直接在骨与植入体之间发挥作用,诱导成骨细胞增殖、分化和矿化,更为直接有效地促进骨整合。
该文系统综述了钛植入体表面分别搭载五种生物活性物质的生物化学改性对骨整合的影响,相较于单一的物理或化学改性,生物化学改性可以使钛植入物直接参与生物过程,所以首先介绍了各种生物活性分子搭载到钛植入物表面的方法,并通过体内外实验来证明经过修饰后的钛植入物对骨整合的影响;其次对各种生物化学改性的优势、不足以及可能的优化方法进行了归纳总结;最后对骨组织与植入体间牢固、持久而直接的结合进行了展望。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程#br#
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||