Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (14): 2265-2272.doi: 10.12307/2022.493
Previous Articles Next Articles
Microglia in spinal cord injury: M1/M2 phenotypic polarization and neurotoxic/neuroprotective effects
Li Chuanhong, Yu Xing, Yang Yongdong, Zhao He
- Department of Orthopedics, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China
-
Received:2021-07-03Revised:2021-07-05Accepted:2021-08-04Online:2022-05-18Published:2021-12-22 -
Contact:Yu Xing, MD, Professor, Doctoral supervisor, Chief physician, Department of Orthopedics, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China -
About author:Li Chuanhong, Doctoral candidate, Department of Orthopedics, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 81973882 (to YX); the National Natural Science Foundation of China for the Youth, No. 81804119 (to YYD); Dongzhimen Hospital 2020 Innovation of Science and Technology Project, No. DZMKJCX-2020-049 (to YX)
CLC Number:
Cite this article
Li Chuanhong, Yu Xing, Yang Yongdong, Zhao He. Microglia in spinal cord injury: M1/M2 phenotypic polarization and neurotoxic/neuroprotective effects[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2265-2272.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
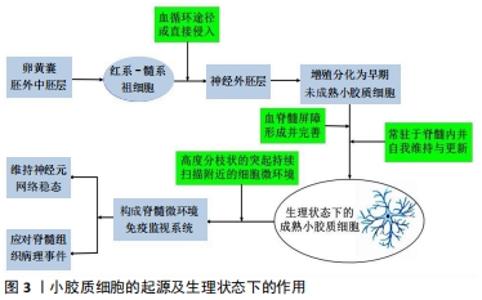
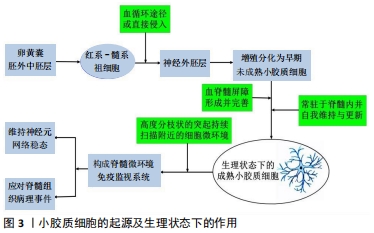
2.1 生理状态下的小胶质细胞 小胶质细胞源于胚胎原始造血系统卵黄囊产生的红系-髓系祖细胞(erythro-myeloid progenitors,EMPs),是中枢神经系统内重要的免疫细胞,本质是一种单核-吞噬细胞,在中枢神经系统各区域分布广泛,占中枢神经系统各类细胞总数的5%-15%[1]。生理条件下,小胶质细胞胞体外形小且不规则(细长、椭圆或杆状),由胞体经多级分化延伸出大量精细的侧枝(突起)。人们借助显微镜下延时摄影、双光子显微镜等发现,生理状态下的小胶质细胞胞体静止,而细胞表面高度分枝状的突起却活跃地进行着伸缩活动,每个细小的终末分枝负责“管理”周围30-50 μm范围内的神经区域,各分枝间互不重叠[2]。日本学者深入观察发现小胶质细胞突起在动态运动中以一定频率接触周围神经元的突触,该过程可增加突触及神经元的活性[3]。目前研究认为生理状态下的小胶质细胞具有活跃的免疫监测功能,利于维持神经元网络稳态[4],这些免疫细胞的失调可能导致中枢神经系统异常的免疫/炎症反应及神经元死亡等病理改变。这是一个里程碑式的发现,生理状态下的小胶质细胞并非处于早期定义的活动与功能的“静息状态”。与神经元、星形胶质细胞等同样呈高度分枝状的细胞所不同的是,小胶质细胞依靠其持续活动的精细分枝管理附近的细胞微环境[2],为中枢神经系统提供了一个高效的免疫监视系统,构成了应对中枢神经系统病理事件的第一道防线,见图3。 "
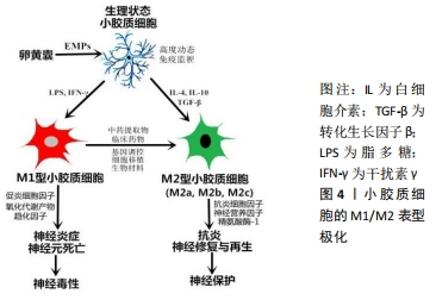
小胶质细胞研究的先驱RíO-HORTEGA早在一个世纪前即发表了多篇关于小胶质细胞激活的经典文献,当时已认识到小胶质细胞在病理状态下发生“阿米巴样”形态改变,吞噬能力增加[5]。目前认为,在中枢神经系统创伤、炎症、肿瘤、退变时,小胶质细胞表面丰富的膜受体[6]、膜离子通道等感知到微环境内的异常信号[7],二者发生相互作用经过复杂的分子机制转化导致小胶质细胞激活,此过程中涉及的具体分子生物学机制尚不十分清楚。激活后的小胶质细胞胞体增大、表面分枝状的突起回缩变短甚至消失,随后又伸出新的突起,细胞形态呈典型的阿米巴状。同时,其增殖、趋化迁移、吞噬、分泌细胞因子等功能明显增强,并具有较强的抗原呈递作用(表达MHCⅡ类分子),激活下游一系列免疫反应,参与介导中枢神经系统病变部位的继发损伤(炎性反应、凋亡、氧化应激等)与修复[8]。 小胶质细胞是可塑性很强的免疫细胞,对周围病理环境具有灵敏的感知与反应能力。早期研究认为激活态的小胶质细胞不存在异质性,近十余年研究证据表明,小胶质细胞可以被一定途径激活为功能状态、表面标志物迥异的两种表型,即M1表型(经典激活型)与M2表型(选择性激活型)[9],见图4。"
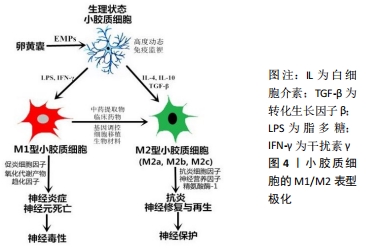
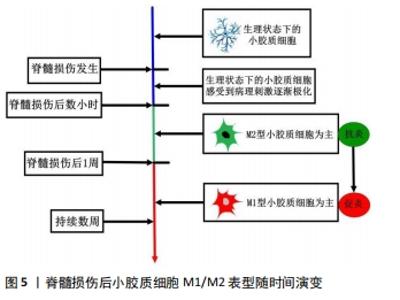
2.2.1 M1/M2表型命名来源 M1/M2表型命名最早用于描述巨噬细胞激活后的不同亚群,STEIN等[10]1992年提出巨噬细胞的选择性激活亚群,该亚群由Th2型细胞因子白细胞介素4所激活,特征为促炎因子分泌减少、内吞清除与抗原呈递能力增强,这与Th1型淋巴细胞产物干扰素γ诱导的经典促炎型巨噬细胞明显不同。2000年,MILLS等[11]将激活态巨噬细胞促炎和抗炎的功能特性分别定义为M1反应与M2反应,该命名来源于他们的实验发现:Th1小鼠品系(C57BL/6、B10D2)的巨噬细胞更易被脂多糖/干扰素γ诱导产生促炎的NO(M1反应),而Th2小鼠品系(BALB/c、DBA/2)来源的巨噬细胞激活后却表达抗炎的精氨酸酶(M2反应)。随后,人们发现主导M1与M2反应的巨噬细胞亚群具有不同的激活物、表面标志与生物学功能,遂将相应的亚群命名为M1(经典激活)和M2(选择性激活)表型[12],这与T淋巴细胞亚群Th1/Th2细胞的命名相似。小胶质细胞与巨噬细胞同属单核吞噬细胞,近20年来小胶质细胞的研究不断深入,M1/M2分型被逐渐用于描述极化的小胶质细胞,先用于探索不同极化状态小胶质细胞与大脑疾病的关系[13],后也用于脊髓损伤后小胶质细胞的分型。 2.2.2 M1/M2表型小胶质细胞的特征 小胶质细胞的极化指在特定的环境条件作用下,生理状态下的小胶质细胞激活并呈现出相对独特的、稳定的、功能性的表型的过程。脂多糖、干扰素γ等促炎分子诱导小胶质细胞极化为M1型,M1型小胶质细胞通过释放大量的炎症因子(肿瘤坏死因子α、白细胞介素1β、白细胞介素6、白细胞介素12等)、氧化代谢产物、趋化因子、基质金属蛋白酶及高表达诱导型一氧化氮合酶、Toll样受体、抗原呈递相关分子等增加对凋亡细胞的吞噬,提高中枢神经系统对伤害性刺激的防御清除能力[14],但同时导致神经炎症扩大、神经元死亡增加、神经组织损伤加重等神经毒性作用,所以M1型又被称为促炎/神经毒性型[15];白细胞介素4、白细胞介素10、转化生长因子β等抗炎递质驱动小胶质细胞极化为M2型,其特征是抗炎细胞因子(白细胞介素4、白细胞介素10、白细胞介素13、转化生长因子β等)、神经营养因子(胰岛素样生长因子1、脑源性神经营养因子、胶质细胞源性神经营养因子等)分泌增加,Arg-1、清道夫受体高表达,以及吞噬受损髓鞘的能力增强。M2型小胶质细胞发挥抗炎与神经支持作用[9],提高神经组织对外来伤害的耐受力,有助于炎症控制、血脊髓屏障重建、血管与神经再生、内环境稳态恢复等,故M2型又称抗炎/神经保护型[16]。M1与M2表型均具有吞噬功能,但各自吞噬的速度与质量并不相同[17]。M1型小胶质细胞吞噬坏死组织后形成酸性较弱成熟慢的吞噬小体,这一过程形成相应抗原并呈递给T细胞,触发下游免疫反应;而M2型小胶质细胞产生酸性强成熟快的吞噬小体,可高效清除有害坏死物质,利于脊髓损伤后的神经修复重建。 与巨噬细胞类似,小胶质细胞的M2表型又可细分为M2a、M2b、M2c 3种亚型,每种亚型有各自相对独特的触发因子和生物学功能[18-19]。M2a型是最典型的M2表型,由白细胞介素4和/或白细胞介素13诱导的选择性激活途径产生,分泌抗炎、促生长的细胞因子,吞噬清除细胞碎片,参与神经组织的修复与再生;M2b型(也称为Ⅱ型替代激活型)在病毒感染、白细胞介素1β+脂多糖、免疫复合物(IgA与Fcγ受体结合)的刺激下获得,该表型可同时产生促炎与抗炎的细胞因子,也具有较强的吞噬活性,被认为是介于M1与M2a之间的一种复合状态;M2c极化由白细胞介素10、转化生长因子β、糖皮质激素等触发,缺少传统意义上M2型小胶质细胞的部分功能,可释放较高水平的转化生长因子β,与神经结构重塑及免疫抑制相关,也称获得性失活表型。 2.2.3 脊髓损伤后小胶质细胞的M1/M2表型随时间轴演变 CHEN等[18]用高糖环境体外持续激活小胶质细胞,发现其表型转化遵循从M2a至M2b再至M1的演变过程,即从抗炎到促炎的功能转化过程,脊髓损伤后小胶质细胞的表型转化也有类似过程,见图5。"
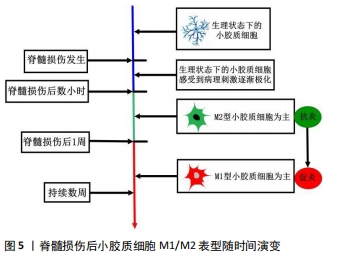
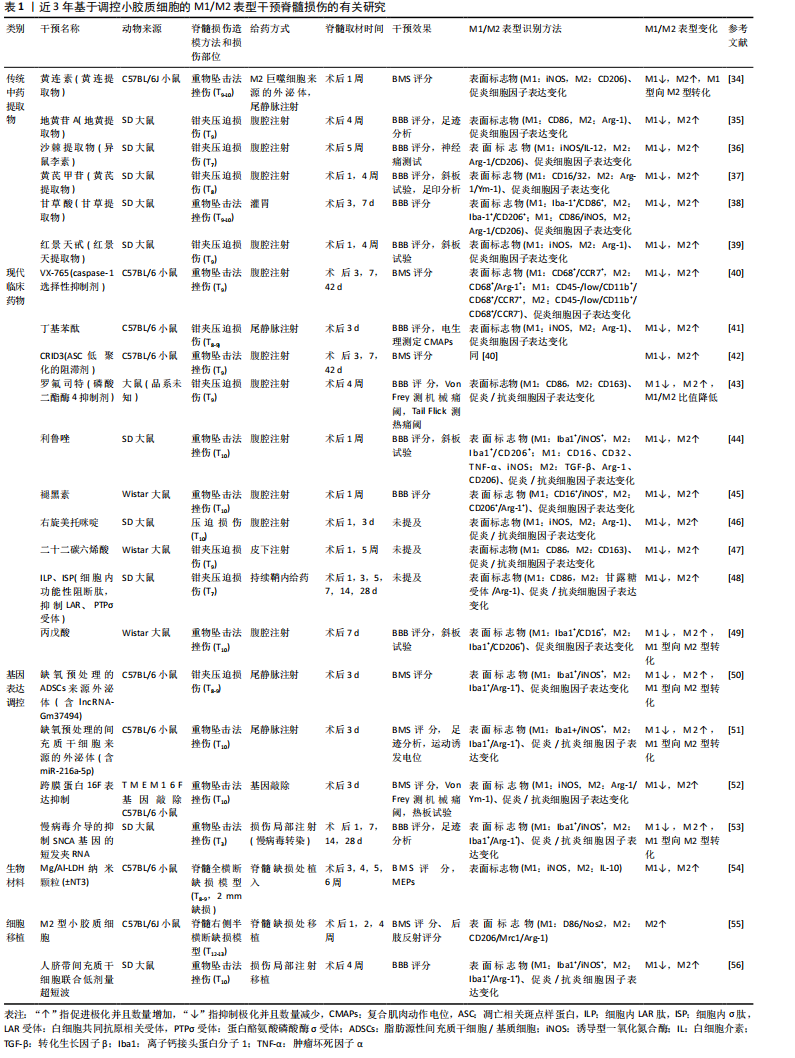
脊髓损伤后数小时内,损伤部位常驻及周围脊髓组织募集的小胶质细胞即在病理刺激下极化为M2表型[20],M2型小胶质细胞通过分泌抗炎细胞因子、神经营养因子等抑制过度的免疫/炎症反应,吞噬细胞碎片,保护周围正常组织。脊髓损伤后约1周,小胶质细胞急速增殖[21],在损伤脊髓内异常微环境的持续刺激下,M1表型的小胶质细胞逐渐占主导地位,极化达到峰值[9],M1型小胶质细胞发挥神经毒性作用,加重脊髓继发性损伤、阻碍神经再生与重塑,该过程可持续数周。脊髓损伤后小胶质细胞表型由M2型为主到M1型为主的快速转化是目前脊髓损伤后神经功能恢复不佳的重要原因之一。由此可见,M2型小胶质细胞只在脊髓损伤后的急性期短暂出现过数量优势,由于损伤部位新招募的M1型小胶质细胞不断增加,以及从M2到M1的表型转换,在关键的亚急性期M2表型不能产生足够的神经保护力量以抗衡M1表型的神经毒性作用。如果能维持脊髓损伤后短暂出现的M2极化,逆转M1表型的异常活化,恢复M1/M2表型间的平衡,将为脊髓损伤治疗带来新的曙光。 2.2.4 M1/M2表型分类的争议 M1/M2表型分类法的应用尽管便于理解,为较多学者所采用,但面对多样的病理刺激和微环境改变,小胶质细胞极化的过程呈高度多样化,M1/M2简单的抗炎/促炎二分模型不能完全涵盖小胶质细胞各种极化状态[22]。M1/M2表型可能只是一个连续的激活状态谱的两极,而两极之间可能还存在其他多种表型[23],已发现的M2a、M2b、M2c只是其中一部分。例如,首先在巨噬细胞中发现的Mox激活表型,在大麻二酚处理后的BV-2细胞(小鼠小胶质细胞)内同样存在[24];在衰老、慢性应激等病理刺激下,小胶质细胞出现一种新的表型——暗小胶质细胞(dark microglia),在突触的病理重塑中发挥作用[25]。随着高通量单细胞测序技术普及,更多的小胶质细胞功能亚群可能被发现[26]。 M1/M2表型的定义多根据体外细胞实验结果,而体外细胞实验是将分离的小胶质细胞暴露于某种纯化的体外刺激下,这与体内复杂多变的微环境相差甚远,体外确立的M1/M2表型特异性标志物同样不能完全预测体内小胶质细胞的表型(体内小胶质细胞标志物表达受多种因素调控)[27]。有学者发现,在体内微环境复杂信号的刺激下,小胶质细胞可能同时表达M1和M2表型标记物,显示出混合表型[28],用单纯的M1或M2表述并不恰当。在未完全清楚小胶质细胞表型随体内微环境信号转化的规律时,作者设想根据微环境的差异建立小胶质细胞表型的分类框架可能更有实际意义,如“神经发育状态”“生理状态”“急性炎症状态”“慢性炎症状态”“神经修复状态”等,这需要结合小胶质细胞不同状态下的标志物(膜蛋白、胞内效应蛋白、胞外分泌蛋白等)加以验证及细化分类[29],有赖于未来在分子细胞生物学、生物信息学等领域的深入研究。 激活态小胶质细胞相关的术语也在改变,“激活”是从巨噬细胞延续过来的概念,表示小胶质细胞从不活跃的静息状态转变为功能状态的过程,但目前研究发现生理状态下的小胶质细胞具有活跃的免疫监视功能,“静息状态”表述可能不正确,相应的“激活”也就不恰当了[17]。部分学者认为小胶质细胞的生理表型与脊髓损伤后的M1/M2表型都属于功能表型[30],代表小胶质细胞在生理病理条件下不同的功能状态,这3种表型可以根据细胞所处的微环境发生互相转换,而不存在所谓的“活化”与“休眠”的过程。为了更加贴切的描述生理状态下小胶质细胞潜在的活力,有学者建议使用“surveying”“monitoring”等术语替代原有的“resting”[30-31]。 2.3 以调控小胶质细胞 M1/M2表型为靶点的脊髓损伤治疗 小胶质细胞的异质性既是一种挑战,更是一种机遇:小胶质细胞存在复杂的功能亚群,同时对外界环境具有出色的感应能力,这表明小胶质细胞面对不同的治疗方法可能表现出相应的表型改变,进而产生功能的调整。一旦了解了这种异质性,针对特定小胶质细胞亚群的靶向治疗可能会使表型调控更加精确,治疗更加集中,不良反应更少。由此可见,以小胶质细胞为靶点治疗脊髓损伤具有诱人的前景[32]。 2.3.1 治疗理念的演变 早期观点将小胶质细胞分为“静息”与“激活”两大类,后者囊括了小胶质细胞病理状态下所有的表型,过度简化了对小胶质细胞功能的理解,认为激活的小胶质细胞促进免疫/炎症反应,加重脊髓损伤,小胶质细胞的激活具有很大的负面意义,治疗侧重于抑制小胶质细胞的激活,以减轻对损伤部位及其周围组织的神经毒性作用[33]。但中枢神经系统这样一个高度脆弱且组织再生困难的重要器官,很难相信其内部广泛分布的小胶质细胞(基本细胞)仅仅是作为一个危险因素而被进化得来。随着M1/M2分型的提出,M1型小胶质细胞继承了传统“激活”小胶质细胞的神经毒性功能,同为激活状态的M2型小胶质细胞则具有神经保护功能,针对小胶质细胞的脊髓损伤治疗理念由此出现了转折,小胶质细胞表型调控的目标转变为:抑制M1型极化促进M2型极化,或诱导M1型转化为M2型(增加M2型细胞数量,降低M1型细胞数量,提高M2/M1的比率)。近年来,基于抑制M1/促进M2的理论基础,各国学者从中药提取物、具有明确分子作用机制的现代临床药物、基因表达调控、细胞移植、生物材料等方面探索脊髓损伤的治疗,通过一系列分子作用机制调控啮齿动物脊髓损伤模型内小胶质细胞的极化表型,在降低M1型小胶质细胞数量减少神经毒性的同时,提高M2型小胶质细胞数量增强神经保护功能,神经行为学评价、电生理检查和组织形态学观察等明确了各项干预措施的有效性,见表1。"

2.3.2 基于M1/M2表型调控的思考 关于小胶质细胞的M1/M2分型在脊髓损伤干预中的应用,人们从未停止自己的思考。在脊髓损伤后,将M1型小胶质细胞完全消除,仅保留M2型,能否达到神经保护作用的最大化呢?目前研究认为,完全没有M1型小胶质细胞,脊髓损伤转归可能更不理想。动物研究中,抑制脊髓损伤后小胶质细胞的激活(未完全抑制)可以改善脊髓功能[57],若在脊髓损伤前清除小胶质细胞,神经功能恢复情况反而比无干预的空白对照组更差[21,58]。这提示,将小胶质细胞的M1表型极化控制在一个合理的范围,保持适度的炎性环境,有益于脊髓损伤后神经的修复[59],适量的M1型小胶质细胞对坏死组织的吞噬与清除、抗原提呈、招募有益的免疫细胞进入损伤部位等功能,也可为神经修复创造条件。理想情况下应该避免M1型的过度极化,促进M2型的极化,将脊髓损伤后的炎症反应限定在一个有利于损伤脊髓修复的程度上。脊髓损伤后如何能将M1/M2型小胶质细胞的数量及比例调控到最佳平衡状态?人们未来的脊髓损伤研究任重而道远。 随着对小胶质细胞未知功能表型的深入探索,加之精准调控各类表型的手段日益丰富,“抑制M1型促进M2型”这句经典的口号迟早会被抛弃,代之以根据脊髓损伤患者年龄、性别、遗传背景、身体功能、疾病情况等个体化因素而“量身定制”并动态调整的小胶质细胞功能表型,将个性化切换小胶质细胞功能作为脊髓损伤治疗的突破口。"

| [1] THION MS, GINHOUX F, GAREL S. Microglia and early brain development: An intimate journey. Science. 2018;362(6411):185-189. [2] RAIVICH G. Like cops on the beat: the active role of resting microglia. Trends Neurosci. 2005;28(11):571-573. [3] AKIYOSHI R, WAKE H, KATO D, et al. Microglia Enhance Synapse Activity to Promote Local Network Synchronization. eNeuro. 2018;5(5): ENEURO.0088-18.2018. [4] UMPIERRE AD, WU LJ. How microglia sense and regulate neuronal activity. Glia. 2021;69(7):1637-1653. [5] SIERRA A, DE CASTRO F, DEL RJ, et al. The “Big-Bang” for modern glial biology: Translation and comments on Pio del Rio-Hortega 1919 series of papers on microglia. Glia. 2016;64(11):1801-1840. [6] RODRIGUEZ-GOMEZ JA, KAVANAGH E, ENGSKOG-VLACHOS P, et al. Microglia: Agents of the CNS Pro-Inflammatory Response. Cells. 2020; 9(7):1717. [7] LUO L, SONG S, EZENWUKWA C C, et al. Ion channels and transporters in microglial function in physiology and brain diseases. Neurochem Int. 2021;142:104925. [8] VENKATESH K, GHOSH S K, MULLICK M, et al. Spinal cord injury: pathophysiology, treatment strategies, associated challenges, and future implications. Cell Tissue Res. 2019;377(2):125-151. [9] PETRALLA S, DE CHIRICO F, MITI A, et al. Epigenetics and Communication Mechanisms in Microglia Activation with a View on Technological Approaches. Biomolecules. 2021;11(2):306. [10] STEIN M, KESHAV S, HARRIS N, et al. Interleukin 4 potently enhances murine macrophage mannose receptor activity: a marker of alternative immunologic macrophage activation. J Exp Med. 1992; 176(1):287-292. [11] MILLS CD, KINCAID K, ALT JM, et al. M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol. 2000;164(12):6166-6173. [12] GORDON S. Alternative activation of macrophages. Nat Rev Immunol. 2003;3(1):23-35. [13] LEE E, EO JC, LEE C, et al. Distinct Features of Brain-Resident Macrophages: Microglia and Non-Parenchymal Brain Macrophages. Mol Cells. 2021;44(5): 281-291. [14] ZHOU X, ZHANG J, LI Y, et al. Astaxanthin inhibits microglia M1 activation against inflammatory injury triggered by lipopolysaccharide through down-regulating miR-31-5p. Life Sci. 2021;267:118943. [15] GOMES-LEAL W. Why microglia kill neurons after neural disorders? The friendly fire hypothesis. Neural Regen Res. 2019;14(9):1499-1502. [16] YU T, YU H, ZHANG B, et al. Promising Neuroprotective Function for M2 Microglia in Kainic Acid-Induced Neurotoxicity Via the Down-Regulation of NF-kappaB and Caspase 3 Signaling Pathways. Neuroscience. 2019; 406: 86-96. [17] CHERRY JD, OLSCHOWKA JA, O’BANION MK. Are “resting” microglia more “m2”? Front Immunol. 2014;5:594. [18] CHEN C, WU S, HONG Z, et al. Chronic hyperglycemia regulates microglia polarization through ERK5. Aging (Albany NY). 2019;11(2): 697-706. [19] BELL-TEMIN H, CULVER-COCHRAN AE, CHAPUT D, et al. Novel Molecular Insights into Classical and Alternative Activation States of Microglia as Revealed by Stable Isotope Labeling by Amino Acids in Cell Culture (SILAC)-based Proteomics. Mol Cell Proteomics. 2015;14(12): 3173-3184. [20] HU X, LEAK R K, SHI Y, et al. Microglial and macrophage polarization-new prospects for brain repair. Nat Rev Neurol. 2015;11(1):56-64. [21] BELLVER-LANDETE V, BRETHEAU F, MAILHOT B, et al. Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat Commun. 2019;10(1):518. [22] DEVANNEY NA, STEWART AN, GENSEL JC. Microglia and macrophage metabolism in CNS injury and disease: The role of immunometabolism in neurodegeneration and neurotrauma. Exp Neurol. 2020;329:113310. [23] CHERRY JD, OLSCHOWKA JA, O’BANION MK. Neuroinflammation and M2 microglia: the good, the bad, and the inflamed. J Neuroinflammation. 2014;11:98. [24] JUKNAT A, GAO F, COPPOLA G, et al. miRNA expression profiles and molecular networks in resting and LPS-activated BV-2 microglia-Effect of cannabinoids. PLoS One. 2019;14(2):e212039. [25] BISHT K, SHARMA K P, LECOURS C, et al. Dark microglia: A new phenotype predominantly associated with pathological states. Glia. 2016;64(5): 826-839. [26] HAMMOND TR, DUFORT C, DISSING-OLESEN L, et al. Single-Cell RNA Sequencing of Microglia throughout the Mouse Lifespan and in the Injured Brain Reveals Complex Cell-State Changes. Immunity. 2019; 50(1):253-271. [27] RANSOHOFF RM. A polarizing question: do M1 and M2 microglia exist? Nat Neurosci. 2016;19(8):987-991. [28] MORGANTI JM, RIPARIP LK, ROSI S. Call Off the Dog(ma): M1/M2 Polarization Is Concurrent following Traumatic Brain Injury. PLoS One. 2016;11(1): e148001. [29] JURGA AM, PALECZNA M, KUTER KZ. Overview of General and Discriminating Markers of Differential Microglia Phenotypes. Front Cell Neurosci. 2020;14:198. [30] HANISCH UK, KETTENMANN H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci. 2007; 10(11):1387-1394. [31] HOWE ML, BARRES BA. A novel role for microglia in minimizing excitotoxicity. BMC Biol. 2012;10:7. [32] PRINZ M, JUNG S, PRILLER J. Microglia Biology: One Century of Evolving Concepts. Cell. 2019;179(2):292-311. [33] BANATI RB, GEHRMANN J, SCHUBERT P, et al. Cytotoxicity of microglia. Glia. 1993;7(1):111-118. [34] GAO ZS, ZHANG CJ, XIA N, et al. Berberine-loaded M2 macrophage-derived exosomes for spinal cord injury therapy. Acta Biomater. 2021; 126:211-223. [35] XIAO S, WANG C, YANG Q, et al. Rea regulates microglial polarization and attenuates neuronal apoptosis via inhibition of the NF-kappaB and MAPK signalings for spinal cord injury repair. J Cell Mol Med. 2021;25(3):1371-1382. [36] CHEN F, HU M, SHEN Y, et al. Isorhamnetin promotes functional recovery in rats with spinal cord injury by abating oxidative stress and modulating M2 macrophages/microglia polarization. Eur J Pharmacol. 2021;895:173878. [37] LIN J, PAN X, HUANG C, et al. Dual regulation of microglia and neurons by Astragaloside IV-mediated mTORC1 suppression promotes functional recovery after acute spinal cord injury. J Cell Mol Med. 2020;24(1): 671-685. [38] SU XQ, WANG XY, GONG FT, et al. Oral treatment with glycyrrhizin inhibits NLRP3 inflammasome activation and promotes microglial M2 polarization after traumatic spinal cord injury. Brain Res Bull. 2020;158:1-8. [39] WANG C, WANG Q, LOU Y, et al. Salidroside attenuates neuroinflammation and improves functional recovery after spinal cord injury through microglia polarization regulation. J Cell Mol Med. 2018;22(2):1148-1166. [40] CHEN J, CHEN YQ, SHI YJ, et al. VX-765 reduces neuroinflammation after spinal cord injury in mice. Neural Regen Res. 2021;16(9):1836-1847. [41] WANG L, WU J P, HE X J. Butylphthalide has an Anti-Inflammatory Role in Spinal Cord Injury by Promoting Macrophage/Microglia M2 Polarization via p38 Phosphorylation. Spine (Phila Pa 1976). 2020; 45(17):E1066-E1076. [42] CHEN YQ, WANG SN, SHI YJ, et al. CRID3, a blocker of apoptosis associated speck like protein containing a card, ameliorates murine spinal cord injury by improving local immune microenvironment. J Neuroinflammation. 2020; 17(1):255. [43] MORADI K, GOLBAKHSH M, HAGHIGHI F, et al. Inhibition of phosphodiesterase IV enzyme improves locomotor and sensory complications of spinal cord injury via altering microglial activity: Introduction of Roflumilast as an alternative therapy. Int Immunopharmacol. 2020;86:106743. [44] WU Q, ZHANG Y, ZHANG Y, et al. Riluzole improves functional recovery after acute spinal cord injury in rats and may be associated with changes in spinal microglia/macrophages polarization. Neurosci Lett. 2020;723:134829. [45] ZHANG Y, LIU Z, ZHANG W, et al. Melatonin improves functional recovery in female rats after acute spinal cord injury by modulating polarization of spinal microglial/macrophages. J Neurosci Res. 2019; 97(7):733-743. [46] HE H, ZHOU Y, ZHOU Y, et al. Dexmedetomidine Mitigates Microglia-Mediated Neuroinflammation through Upregulation of Programmed Cell Death Protein 1 in a Rat Spinal Cord Injury Model. J Neurotrauma. 2018; 35(21):2591-2603. [47] MANZHULO O, TYRTYSHNAIA A, KIPRYUSHINA Y, et al. Docosahexaenoic acid induces changes in microglia/macrophage polarization after spinal cord injury in rats. Acta Histochem. 2018;120(8):741-747. [48] DYCK S, KATARIA H, ALIZADEH A, et al. Perturbing chondroitin sulfate proteoglycan signaling through LAR and PTPsigma receptors promotes a beneficial inflammatory response following spinal cord injury. J Neuroinflammation. 2018;15(1):90. [49] CHEN S, YE J, CHEN X, et al. Valproic acid attenuates traumatic spinal cord injury-induced inflammation via STAT1 and NF-kappaB pathway dependent of HDAC3. J Neuroinflammation. 2018;15(1):150. [50] SHAO M, JIN M, XU S, et al. Exosomes from Long Noncoding RNA-Gm37494-ADSCs Repair Spinal Cord Injury via Shifting Microglial M1/M2 Polarization. Inflammation. 2020;43(4):1536-1547. [51] LIU W, RONG Y, WANG J, et al. Exosome-shuttled miR-216a-5p from hypoxic preconditioned mesenchymal stem cells repair traumatic spinal cord injury by shifting microglial M1/M2 polarization. J Neuroinflammation. 2020;17(1):47. [52] ZHAO J, GAO QY. TMEM16F inhibition limits pain-associated behavior and improves motor function by promoting microglia M2 polarization in mice. Biochem Biophys Res Commun. 2019;517(4):603-610. [53] ZENG H, LIU N, YANG YY, et al. Lentivirus-mediated downregulation of alpha-synuclein reduces neuroinflammation and promotes functional recovery in rats with spinal cord injury. J Neuroinflammation. 2019;16(1):283. [54] ZHU R, ZHU X, ZHU Y, et al. Immunomodulatory Layered Double Hydroxide Nanoparticles Enable Neurogenesis by Targeting Transforming Growth Factor-beta Receptor 2. ACS Nano. 2021;15(2):2812-2830. [55] KOBASHI S, TERASHIMA T, KATAGI M, et al. Transplantation of M2-Deviated Microglia Promotes Recovery of Motor Function after Spinal Cord Injury in Mice. Mol Ther. 2020;28(1):254-265. [56] NA L, WANG S, LIU T, et al. Ultrashort Wave Combined with Human Umbilical Cord Mesenchymal Stem Cell (HUC-MSC) Transplantation Inhibits NLRP3 Inflammasome and Improves Spinal Cord Injury via MK2/TTP Signalling Pathway. Biomed Res Int. 2020;2020:3021750. [57] ZHANG Y, ZHANG L, SHEN J, et al. Two-photon-excited fluorescence microscopy as a tool to investigate the efficacy of methylprednisolone in a mouse spinal cord injury model. Spine (Phila Pa 1976). 2014;39(8): E493-E499. [58] FU H, ZHAO Y, HU D, et al. Depletion of microglia exacerbates injury and impairs function recovery after spinal cord injury in mice. Cell Death Dis. 2020;11(7):528. [59] XIONG XY, LIU L, YANG QW. Functions and mechanisms of microglia/macrophages in neuroinflammation and neurogenesis after stroke. Prog Neurobiol. 2016;142:23-44. [60] FUGER P, HEFENDEHL JK, VEERARAGHAVALU K, et al. Microglia turnover with aging and in an Alzheimer’s model via long-term in vivo single-cell imaging. Nat Neurosci. 2017;20(10):1371-1376. [61] EVANS TA, BARKAUSKAS DS, MYERS JT, et al. Intravital imaging of axonal interactions with microglia and macrophages in a mouse dorsal column crush injury. J Vis Exp. 2014(93):e52228. [62] GALATRO TF, HOLTMAN IR, LERARIO AM, et al. Transcriptomic analysis of purified human cortical microglia reveals age-associated changes. Nat Neurosci. 2017;20(8):1162-1171. [63] GOSSELIN D, SKOLA D, COUFAL NG, et al. An environment-dependent transcriptional network specifies human microglia identity. Science. 2017;356(6344):eaal3222. [64] ORMEL PR, VIEIRA DSR, van BODEGRAVEN EJ, et al. Microglia innately develop within cerebral organoids. Nat Commun. 2018;9(1):4167. [65] HASSELMANN J, BLURTON-JONES M. Human iPSC-derived microglia: A growing toolset to study the brain’s innate immune cells. Glia. 2020; 68(4):721-739. [66] DUBBELAAR ML, KRACHT L, EGGEN B, et al. The Kaleidoscope of Microglial Phenotypes. Front Immunol. 2018;9:1753. [67] XU R, LI X, BORELAND AJ, et al. Human iPSC-derived mature microglia retain their identity and functionally integrate in the chimeric mouse brain. Nat Commun. 2020;11(1):1577. [68] JIANG P, TURKALJ L, XU R. High-Fidelity Modeling of Human Microglia with Pluripotent Stem Cells. Cell Stem Cell. 2020;26(5):629-631. [69] POSFAI B, CSEREP C, ORSOLITS B, et al. New Insights into Microglia-Neuron Interactions: A Neuron’s Perspective. Neuroscience. 2019; 405:103-117. [70] GAUDET AD, FONKEN LK. Glial Cells Shape Pathology and Repair After Spinal Cord Injury. Neurotherapeutics. 2018;15(3):554-577. [71] REYNOLDS JL, MAHATO RI. Nanomedicines for the Treatment of CNS Diseases. J Neuroimmune Pharmacol. 2017;12(1):1-5. [72] JIN K, LUO Z, ZHANG B, et al. Biomimetic nanoparticles for inflammation targeting. Acta Pharm Sin B. 2018;8(1):23-33. [73] ALLAVENA P, PALMIOLI A, AVIGNI R, et al. PLGA Based Nanoparticles for the Monocyte-Mediated Anti-Tumor Drug Delivery System. J Biomed Nanotechnol. 2020;16(2):212-223. [74] SHIN WH, LEE DY, PARK KW, et al. Microglia expressing interleukin-13 undergo cell death and contribute to neuronal survival in vivo. Glia. 2004;46(2):142-152. [75] DECZKOWSKA A, AMIT I, SCHWARTZ M. Microglial immune checkpoint mechanisms. Nat Neurosci. 2018;21(6):779-786. [76] BORGGREWE M, KOOISTRA S M, NOELLE R J, et al. Exploring the VISTA of microglia: immune checkpoints in CNS inflammation. J Mol Med (Berl). 2020;98(10):1415-1430. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [5] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [6] | Tang Wenjing, Wu Siyuan, Yang Chen, Tao Xi. Inflammatory responses in post-stroke depression [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1278-1285. |
| [7] | Kan Houming, Fan Lijun, Chen Xuetai, Shen Wen. Application of platelet-rich plasma in neuropathic pain [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1286-1292. |
| [8] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [9] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [10] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [11] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [12] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [13] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [14] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [15] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

