Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (8): 1286-1292.doi: 10.12307/2022.237
Previous Articles Next Articles
Application of platelet-rich plasma in neuropathic pain
Kan Houming1, Fan Lijun1, Chen Xuetai1, Shen Wen2
- 1Jiangsu Provincial Key Laboratory of Anesthesiology, Xuzhou Medical University, Xuzhou 221004, Jiangsu Province, China; 2Department of Pain Management, Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, Jiangsu Province, China
-
Received:2021-03-08Revised:2021-03-09Accepted:2021-04-24Online:2022-03-18Published:2021-11-02 -
Contact:Shen Wen, Master, Professor, Chief physician, Department of Pain Management, Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, Jiangsu Province, China -
About author:Kan Houming, Master candidate, Physician, Jiangsu Provincial Key Laboratory of Anesthesiology, Xuzhou Medical University, Xuzhou 221004, Jiangsu Province, China Fan Lijun, Master candidate, Attending physician, Jiangsu Provincial Key Laboratory of Anesthesiology, Xuzhou Medical University, Xuzhou 221004, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81571066 (to SW)
CLC Number:
Cite this article
Kan Houming, Fan Lijun, Chen Xuetai, Shen Wen. Application of platelet-rich plasma in neuropathic pain[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1286-1292.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
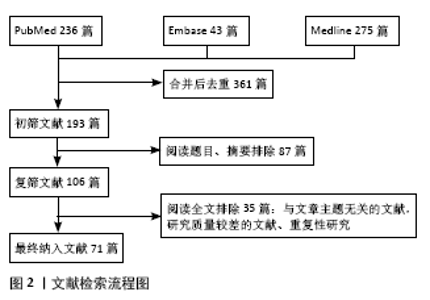
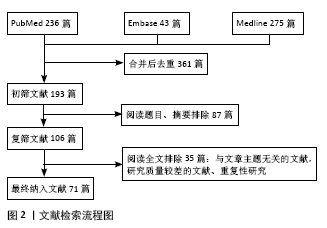
2.1 神经病理性疼痛的分类和发病机制 世界卫生组织 2018年发布国际疼痛分类第11次修订本,将神经病理性疼痛分为中枢神经病理性疼痛和周围神经病理性疼痛[13]。中枢性神经病理性疼痛是指中枢躯体感觉神经系统损伤或疾病引起的慢性疼痛[14],具体包含4个诊断类别:脊髓损伤相关神经病理性疼痛、脑损伤相关神经病理性疼痛、卒中后疼痛、多发硬化相关神经病理性疼痛[15]。周围神经病理性疼痛是指周围躯体感觉神经系统疾病或损伤引起的慢性疼痛[14],具体包含5个诊断类别:三叉神经痛、周围神经损伤后疼痛、痛性多发神经病变、带状疱疹后神经痛及痛性根性压迫病变[15]。周围神经病理性疼痛又可以根据病因分为缺血/代谢、感染/炎症、创伤、药物等不同类型[6],具体分型见图2。"

神经病理性疼痛发病机制分为周围机制和中枢机制两方面。处理疼痛的无髓鞘C纤维和细髓鞘Aδ纤维的周围终末受到刺激后(如代谢损伤、毒素、药物、细胞因子和其他炎症介质),导致神经纤维密度改变和神经元的过度兴奋,诱发神经病理性疼痛的发生[16]。异位放电信号可以沿着轴突传导,传至背根神经节或三叉神经节时[17],可导致神经节膜成分、突触性质和突触位置出现特异性变化[18]。在重复或强烈的刺激下,脊髓上传伤害性疼痛的通路对刺激敏感性增加,出现中枢敏化[19-20]。二级神经元的突触对刺激的敏感性增强,可能导致钙通透性增加、受体过度表达和突触位置的改变[18]。周围神经损伤导致许旺细胞活化,并释放促炎细胞因子、营养因子和趋化因子,将巨噬细胞、中性粒细胞和其他炎症细胞招募到损伤部位,促炎因子与抗炎因子共同促进轴突再生,如抗炎因子不足,损伤局部伤害性神经元神经生长因子酪氨酸激酶TrkA受体激活[21],小胶质细胞过度激活,会促进疼痛介质的过度释放,加剧慢性疼痛的状态[22]。 2.2 PRP的分类和制备方法 一般认为PRP中血小板浓度是全血的四五倍,不过一直缺少量化指标。2001年Marx[23]定义PRP每微升血浆中至少含有100万个血小板,这对PRP的标准做了一个量化指标。Dohan等[24]基于PRP中血小板、白细胞、纤维蛋白含量不同,将PRP分为纯PRP、富白细胞PRP、纯富血小板纤维蛋白、富含白细胞的血小板纤维蛋白4类。如无特别说明,目前常说的PRP一般指富白细胞PRP。PRP常为液态或凝胶状,2017年KIEB等[25]研制了一种粉剂的PRP衍生物,经检测其生长因子浓度与传统PRP产品类似,并且具有易保存、使用方便的优点。PRP还可以根据分离方法不同、是否额外进行体外激活、是否添加抗凝剂进行分类。 PRP依据分离方式不同,可分为手工分离法和全自动血浆分离法。全自动血浆离法采用医用血成分自动分离设备(如自体血小板分离仪),通过预设程序在一个封闭系统中离心得到PRP[26],该法优点是自动化程度高、操作方便、迅速、不易污染,提取的PRP纯度和浓度可控;缺点是需要的血量较大,需要建立静脉循环通道、设备价格昂贵。目前提取的PRP不易长时间保持生物活性,建议提取后尽快使用,限制了该法的使用。随着PRP制备工艺和保存方法的改进,这种可以大量提取高纯度PRP的方法具有明显优势。PRP的手工分离方法是依据血液中不同组分沉降系数不同,通过密度梯度离心法对浓缩血小板进行提取;依据离心次数不同,可以分为一次离心法、二次分离法、三次分离法,其中二次离心法血小板回收率和富集度均较好,是目前临床使用最广的方法[27]。1976年,SLITCHER等[28]创立了二次离心方法,第一次1 000×g离心9 min,然后3 000×g离心20 min。TAMIMI等[29]认为,过高的离心力可能导致了血小板超微结构的改变,他建议使用160×g离心10 min,二次离心采用400×g离心10 min。目前PRP的制备缺乏标准化的程序,离心时间和离心力存在差异,PRP的成分含量存在差异,阻碍了不同研究之间的疗效分析。CHAHLA等[30]对105项PRP相关研究进行系统分析,只有16%的研究提供了PRP成分的定量指标,仅10%提供了全面、可重复的PRP制备流程,因此呼吁建立PRP制备标准化流程和成分组成分析。 为避免血小板在注射前提前激活,一般会选择使用抗凝剂来抑制PRP活性。常用的抗凝剂有乙二胺四乙酸盐、柠檬酸葡萄糖A和柠檬酸钠。DO等[31]采用不同抗凝剂对PRP进行分析,研究发现乙二胺四乙酸盐样本中血小板浓度最高,不过血小板形态发生了改变,说明乙二胺四乙酸盐可能会损害血小板膜。KRAUS等[32]比较了柠檬酸葡萄糖A和柠檬酸钠对血小板浓度和形态的影响,研究发现使用柠檬酸葡萄糖A可以获得更高浓度的血小板。DU等[33]介绍了一种不需额外添加抗凝剂和激活剂的制备方法,即温控PRP,该法首先将全血在低温条件下(4 ℃)200×g离心10 min,随后将血浆转移到新的试管中,并在1 550×g离心10 min,然后在37 ℃下孵育活化15 min。温控PRP是一种新型制备和活化PRP的方法,不需要任何添加剂,与添加柠檬酸葡萄糖A的PRP相比,温控PRP更能反映血小板生理特性。 PRP使用前是否需要外源性激活尚无定论,常用的激活剂有凝血酶和钙剂,激活后PRP形成凝胶状态,影响基质发育、生长因子含量和伤口的愈合。目前有研究使用低温或金属钛激活,与外源性激活相比具有优势。NLü等[34]将PRP在-80 ℃下冷冻保存24 h,通过冷冻/解冻方式激活PRP,研究发现,PRP中生长因子水平与钙激活相对相当,但是该激活方式无需外加其他物质,最大限度保留了血小板的生理功能。TUNALI等[35]开发了一种使用钛管制备富血小板纤维蛋白的方法,该方法使用金属钛来诱导富血小板纤维蛋白的活性,与传统富含白细胞的血小板纤维蛋白相比,富血小板纤维蛋白中纤维蛋白网密度更大,这种差异可能与钛具有良好的组织相容性有关。 2.3 PRP治疗神经病理性疼痛的机制 机体在受到损伤之后,各种内外源性激活物质会促进血小板活化,血小板中的α-颗粒发生脱颗粒反应,释放大量生长因子、纤维蛋白原、组织蛋白酶和水解酶等[36-37]。释放的生长因子通过细胞膜上的跨膜受体,结合到靶细胞的细胞膜外表面,这些跨膜受体反过来诱导激活内源性信号蛋白,进一步激活细胞内的第二信使,后者诱导细胞增殖、基质形成及合成胶原蛋白等各种细胞内的基因表达[38]。有证据表明,血小板释放的细胞因子以及其他递质对减少/消除慢性神经病理性疼痛具有重要作用[39],具体机制可分为周围机制和中枢机制,见表1。 "


2.3.1 周围机制 抗炎作用:外周致敏在神经损伤后神经病理性疼痛症状的发生中起重要作用。神经损伤部位浸润多种炎性细胞,如中性粒细胞、巨噬细胞和肥大细胞。炎性细胞过度积聚,构成了神经纤维过度兴奋和持续放电的基础[40]。炎症会释放大量的化学介质,如细胞因子、趋化因子和脂质介质,使伤害感受器变得敏感和兴奋,并引起局部化学环境的变化[41]。血小板具有很强的免疫抑制和抗炎作用,通过调节和分泌各种免疫调节因子、血管生成因子和营养因子,可以减少有害的免疫反应和炎症,并修复不同微环境中的不同组织损伤[42]。PRP可以通过多种机制来发挥抗炎作用,通过促进受损组织由促炎状态向抗炎状态转变,可以阻断施万细胞、巨噬细胞、中性粒细胞和肥大细胞释放促炎细胞因子,并抑制促炎因子受体的基因表达[43]。虽然血小板不释放白细胞介素10,但血小板通过诱导未成熟树突细胞产生大量白细胞介素10,减少γ-干扰素的产生,发挥抗炎作用[44]。血小板还可以通过触发核因子κB信号通路,参与抗炎活动[45]。 神经保护和促进轴突再生:神经损伤引起神经元兴奋性异常,诱发神经纤维变性,改变离子通道表达和组成,导致异位放电。神经末梢或轴突上的自发异位活动是自发疼痛的基础,并且是异常疼痛的驱动因素[46]。神经胶质源性神经营养因子是一种能够发挥神经保护作用的生长因子,许多研究将神经胶质源性神经营养因子及其受体作为开发新止痛药的靶点[47]。AL-MASSRI等[48]发现神经生长因子可以通过促进轴突生长和神经元维持及存活起到神经保护作用。ZHENG等[49]发现2.5%-20% PRP以剂量依赖性方式诱导神经生长因子和神经胶质源性神经营养因子合成,并且显著增加施万细胞的迁移,诱导神经增殖和轴突生长,增强髓鞘形成,并通过调节神经胶质源性神经营养因子来减轻疼痛。PRP中血小板衍生生长因子BB通过ERK1/2、PI3K/Akt和JNK信号通路促进人类脂肪衍生干细胞增殖,有利于起到神经再生作用[50]。此外,血管内皮生长因子是神经再生的重要调节因子,它可以通过结合血管生成、神经营养和神经保护来支持和促进再生神经纤维的生长,从而恢复神经功能[51]。血管内皮生长因子和血管生成素1可以促进血管生成,可能有利于神经病理性疼痛的恢复[52]。CASTRO等[53]证明在背根神经损伤模型大鼠中,PRP通过促进肿瘤坏死因子α、转化生长因子β、脑源性神经营养因子、神经胶质源性神经营养因子、血管内皮生长因子、神经生长因子、白细胞介素4、白细胞介素6、白细胞介素13在体内基因表达,部分恢复缩爪退缩反射,改善背根神经切断术后感觉运动的恢复。 免疫调节:免疫调节包含先天性免疫系统和适应性免疫系统两层概念。先天性免疫系统包含多种类型的受体,包括Toll样受体和RIG-1样受体[54]。血小板表面和细胞质中还表达几种特殊的免疫调节受体分子,如P-选择素、跨膜蛋白CD40配体(CD40L)、细胞因子(如白细胞介素1β、转化生长因子β)和血小板特异性Toll样受体[55]。中性粒细胞、单核细胞和树突状细胞是血液中最常见的先天免疫细胞。血小板上的Toll样受体 4可以调节中性粒细胞释放活性氧和髓过氧化物酶,调节白细胞氧化风暴,血小板可以通过诱导单核细胞核因子κB的活化来调节单核细胞功能,通过血小板-单核细胞CD40L-MAC-1直接介导髓过氧化物酶释放[56]。血小板通过免疫球蛋白JAM受体和中性粒细胞、单核细胞/巨噬细胞MAC-1整合素的相互作用招募树突状细胞[57]。血小板衍生生长因子可以促进树突状细胞分化,它是连接先天和适应性免疫系统的关键细胞[58]。在适应性免疫反应中,血小板与白细胞的相互作用有助于适应性免疫反应的激活[55]。血小板颗粒成分分泌的CD40L是调节适应性免疫反应的关键分 子[59],血小板通过CD40-CD40L增强了T细胞依赖B细胞的活化、分化及增殖。 镇痛作用:活化的血小板释放许多促炎和抗炎递质,这些递质可以诱发疼痛,但也能减轻炎症和疼痛。新制备的血小板在PRP中处于休眠状态,在直接或间接被激活后,血小板形态改变并促进血小板聚集,释放其胞内α-致密颗粒[60],致敏颗粒会刺激具有疼痛调控作用的5-羟色胺释 放[61]。目前,5-羟色胺受体多在外周神经中被检测到,5-羟色胺可以通过5-羟色胺1、5-羟色胺2、5-羟色胺3、5-羟色胺4和5-羟色胺7受体影响周围组织部位的伤害性传递[62]。 2.3.2 中枢机制 削弱和逆转中枢敏化:中枢敏化的特征是神经元兴奋性增加,中枢敏化被认为是导致神经病理性疼痛最重要的中枢机制。神经损伤后,脊髓背角兴奋性氨基酸(谷氨酸)释放增强,兴奋性N-甲基-d-天冬氨酸受体持续激活,维持传入神经向感觉脑的传递,在慢性神经损伤的长期刺激下,N-甲基-D-天冬氨酸受体被上调,从而建立一种中枢敏化状态[63]。动物模型表明,阻断N-甲基-D-天冬氨酸受体可以缓解神经病理性疼痛[64]。特异性的N-甲基-D-天冬氨酸受体在神经病理性疼痛中正在成为新兴的治疗靶标[65]。GUO等[66]发现骨髓间充质干细胞可以抑制N-甲基-D-天冬氨酸受体的表达,并保护它们免受谷氨酸兴奋毒性,减轻大鼠脊髓损伤后的机械性痛觉过敏,PRP-骨髓间充质干细胞复合物已被用于治疗脊髓损伤,具有巨大潜力。FRANCONI等[67]发现N-甲基-D-天冬氨酸是人类PRP中血小板聚集和血栓烷B2合成的有效抑制剂,说明N-甲基-D-天冬氨酸与血小板之间存在某种联系。但是目前尚缺乏PRP与N-甲基-D-天冬氨酸受体在神经病理性疼痛中的相关研究,该机制需进一步研究验证。 抑制胶质细胞活化:神经胶质细胞约占中枢神经系统细胞的70%,可分为3种类型:星形胶质细胞、少突胶质细胞和小胶质细胞[68]。神经损伤24 h内小胶质细胞被激活,星形胶质细胞在神经损伤后很快被激活,并且激活持续12周[69]。星形胶质细胞和小胶质细胞随后释放细胞因子,诱导一系列细胞反应,如糖皮质激素和谷氨酸受体上调,导致脊髓兴奋和神经可塑性改变,这与神经病理性疼痛的发生密切相关[70-71]。BORHANI-HAGHIGHI等[72]将PRP鞘内注射到自身免疫性脑脊髓炎小鼠模型中,研究发现PRP可以改善小鼠功能能力,促进髓鞘和少突胶质生成,此外,PRP治疗后,大量小胶质细胞、星形胶质细胞和浸润性炎性细胞以及促炎性细胞因子的表达被逆转。PRP中参与神经病理性疼痛的细胞因子,见表2。 2.4 PRP在中枢性神经病理性疼痛中的应用 中枢神经病理性疼痛是脊髓损伤后最常见的并发症之一,在脊髓损伤患者中发生率为65%-83%,约1/3患者会出现重度疼痛,严重影响患者的生活质量[74]。其发病机制并不十分明确,可能与S100钙结合β蛋白过度表达[75]、G蛋白偶联受体34上调[76]、连接蛋白表达上调[77]、哺乳动物雷帕霉素靶蛋白激活[78]、基质金属蛋白酶表达增加等因素有关[79],导致神经胶质细胞持续活化[80],神经元轴突再生受阻。SHEHADI等[81]将制备的PRP和浓缩骨髓混合,对慢性脊髓损伤患者进行鞘内注射,采用Oswestry功能障碍指数评估功能障碍,百分比越高表示残疾程度越高,脊髓损伤患者治疗前百分比为60%,治疗12个月后百分比降到20%。SALARINIA等[82]采用骨髓间充质干细胞和PRP联合治疗脊髓损伤的大鼠,结果提示,实验组Bcl-2和Caspase-3的表达升高,与对照组相比差异有显著性意义(P < 0.01);实验组大鼠轴突再生增加,细胞凋亡减少,运动功能得到改善。CHEN等[83]对脊髓损伤大鼠进行PRP鞘内注射,研究显示,PRP促进了血管生成和神经元的再生,促进了大鼠的运动能力的恢复。SALARINIA等[84]研究PRP对脊髓挫伤大鼠模型的影响,在造模后24 h鞘内注射PRP,大鼠出现功能性运动恢复和轴突再生。DE CASTRO等[85]对腰椎后根切断术大鼠模型注射PRP和人胚胎干细胞混合凝胶,结果表明,混合凝胶可以促进切断后的背根轴突再生,因此,PRP可以作为背根切断后恢复感觉-运动功能的一种治疗手段。脊髓损伤在细胞水平涉及复杂的变化,包括缺血、缺氧、坏死、病理性炎症因子的过度产生、兴奋性氨基酸的积累、钙离子的内流、氧自由基和一氧化氮释放,导致神经元和神经胶质细胞凋亡,神经功能紊乱[81]。脊髓损伤后出现神经病理性疼痛目前尚无最佳治疗方案,药物治疗仍为主要的治疗手段,神经电刺激治疗、手术治疗可以作为药物治疗无效时的补充治疗方案[86]。基础实验和临床试验证明PRP能促进脊髓神经轴突再生、感觉神经修复、运动功能恢复,可以与干细胞结合使用,在中枢神经病理性疼痛治疗中具有巨大潜力。 2.5 PRP在周围神经病理性疼痛中的应用 2.5.1 创伤后神经病理性疼痛 周围神经在受到切割、挤压、烧伤等创伤后,会产生周围神经损伤,每年有35万例患者受周围神经病理性疼痛的影响,每年的医疗费用为1 500亿美元[87]。PRP注射既可以促进周围神经功能恢复,也具有预防和改善神经病理性疼痛的作用,可以用于治疗周围神经病理性疼痛。LI等[88]在面神经挤压伤的大鼠模型中局部注射PRP,实验发现,PRP组触须运动、眼睑闭合和电生理功能恢复,许旺细胞和轴突显著再生,神经生长因子、脑源性神经营养因子、S-100蛋白表达水平更高。朱亚琼等[89]将PRP注射到挤压伤模型兔坐骨神经外膜周围,实验发现,PRP具有促进神经再生、抑制肌肉萎缩的作用,且多次注射疗效优于单次注射。ZHU等[90]采用PRP联合低剂量超短波治疗兔坐骨神经挤压伤,研究表明,PRP联合低剂量超短波可以加速轴突恢复,并减少靶肌肉萎缩。也有相关临床研究探讨PRP在周围神经病理性疼痛中的作用。IKUMI等[91]发现 PRP可以促进手指周围神经挤压伤患者的康复,患者在接受神经松解治疗后,在损伤近侧端神经周围注射0.5 mL PRP,术后即刻神经病理性疼痛减轻,术后2周手指活动改善,术后4周手指活动正常,术后9个月神经病理性疼痛消失。王兴平[92]采用PRP治疗后路颈椎减压后C5神经麻痹的患者,治疗后患者神经系统动作电位波幅提高,潜伏期缩短,神经感觉功能和运动功能改善,神经病理性疼痛减轻。也有研究探讨PRP治疗烧伤瘢痕导致的神经病理性疼痛的疗效,HUANG等[93]在大鼠烧伤瘢痕处局部注射PRP,注射治疗后神经病理性疼痛减轻,其机制可能与PRP降低神经细胞p-PTEN、p-mTOR、CCL2、p-p38MAPK、p-NFκB的表达有关。 2.5.2 神经卡压后神经病理性疼痛 脊柱退变性神经根痛和腕管综合征均可导致神经卡压后神经病理性疼痛。脊柱退变性神经根痛是由于脊柱退变侵袭神经根,导致受累神经支配区出现以疼痛为主要症状的疾病。脊柱椎间盘突出、小关节和/或钩椎关节增生、黄韧带肥厚等原因,常造成神经根受卡压,出现神经根性痛或坐骨神经痛[94]。将PRP经椎间孔或硬膜外途径注射至病变神经根周围,可以治疗脊柱退变性神经根痛。BHATIA等[95]将PRP注射至病变神经根周围的硬膜外腔,3个月后患者目测类比评分明显降低,患者神经根痛好转。BISE等[96]采用类似的方法治疗后外侧椎间盘突出,治疗后患者疼痛评分明显改善。RUIZ-LOPEZ等[97]分别采用糖皮质激素和PRP硬膜外注射治疗脊柱退变性神经根痛,研究发现,PRP的长期止痛效果优于糖皮质激素治疗。LAM等[98]采用超声引导下PRP注射治疗颈椎病,患者术前左肩部目测类比评分 6-7分,夜间加重时目测类比评分高达8-9分,治疗3周后,患者颈肩部疼痛明显改善(目测类比评分降至0-1分)。腕管综合征是上肢最常见的压迫性神经病,人群中腕管综合征的患病率为1%-5%[99],常采取休息、药物治疗、物理治疗、糖皮质激素注射治疗,疗效不佳时采用正中神经减压治疗。有相关研究表明,PRP也可作为腕管综合征保守治疗的一种方案。SENNA等[100]在超声引导下注射PRP治疗轻中度特发性腕管综合征,研究表明,PRP可有效治疗轻中度特发性腕管综合征,治疗后正中神经的疼痛减轻,神经功能和远端感觉潜伏期改善,疗效优于糖皮质激素。WU等[101]采取超声引导下PRP注射治疗腕管综合征,治疗后1,3个月时,两组患者的目测类比评分均较术前明显改善;治疗6个月时,PRP组的目测类比评分显著低于对照组。RAEISSADAT等[102]采取PRP联合腕部夹板治疗轻中度腕管综合征,治疗后8周发现,PRP联合腕部夹板的治疗效果并不优于单纯腕部夹板,作者认为腕关节内注射PRP并不能显著增加腕夹板保守治疗的效果。不过该研究设计也存在一定问题,首先是随访时间较短,仅对治疗后8周进行随访,无法说明PRP的中长期疗效,另外,该研究未在影像下引导操作,这可能影响注射的精确度。 2.5.3 糖尿病周围神经病理性疼痛 糖尿病周围神经病理性疼痛是由糖尿病引起的周围神经病理性疼痛,常表现为肢体末端灼痛、刺痛或感觉迟钝,伴有痛觉过敏和痛觉超敏,体格检查可伴有手套、袜套样感觉受损。PRP治疗糖尿病并发症的研究多集中于糖尿病足神经性溃疡,有关PRP治疗糖尿病周围神经病理性疼痛的研究较少。HASSANIEN等[103]在超声引导下对糖尿病周围神经病理性疼痛患者采取神经周围PRP注射治疗,在治疗后1,3,6个月时,患者疼痛和麻木症状均较对照组明显改善,因此,神经周围注射PRP是减轻糖尿病神经病变疼痛和麻木、增强周围神经功能的有效疗法。 2.5.4 感染/炎症性神经病理性疼痛 水痘-带状疱疹病毒、麻风分支杆菌、梅毒、艾滋病病毒等感染人体后,会造成特异的神经病理性疼痛。PRP治疗感染/炎症性神经病理性疼痛的相关文献很少,仅有数篇临床研究性报道,缺乏基础实验验证。PRP是否可以在此类疾病中应用,尚需进一步研究。麻风病(Hansen病)是由麻风分枝杆菌引起的慢性肉芽肿性感染,周围神经病变为皮肤和神经系统表现。2014年ANJAYANI等[104]报道了一项随机双盲对照试验研究,证明 1 mL的PRP神经周围注射2周后,可以改善麻风病患者的目测类比疼痛评分,不过PRP治疗麻风病周围神经病仅有此1篇文献。2018年RAVINDRAN等[105]对ANJAYANI等[104]发表的这篇文献进行评论,认为这项研究的设计存在问题,因为患者纳入标准中没有提到麻风病的谱系,而且随访时间较短,不能确定PRP的远期效果如何。黄立荣等[106]采用MRI引导下背根神经节脉冲射频联合PRP注射治疗带状疱疹后神经痛,治疗后患者术后目测类比评分均较术前明显降低,不过该研究为回顾性分析,缺乏对照组,且治疗为药物+背根神经节脉冲射频+PRP的联合方案,随访时间较短,不能确定PRP单独治疗带状疱疹后神经痛的临床疗效。 2.5.5 莫顿氏(Morton)神经瘤 莫顿氏神经瘤是跖痛症的一种形式,跖骨横韧带远端的指间神经膨出导致趾底神经卡压,通常位于第3个跖骨间区域,表现为足底剧烈的灼痛和麻木。DE ANGELIS等[107]对5例莫顿氏神经瘤手术后伤口不愈合患者采用PRP联合透明质酸治疗,术后30 d所有患者的伤口都完全愈合,足部疼痛麻木明显缓解,作者推测PRP也可以在非手术情况下治疗莫顿氏神经瘤,但并未见该作者的进一步报道。PRP治疗莫顿氏神经瘤仅此1例报道,而且该报道是在手术切除后局部注射PRP,目前尚无单纯PRP注射治疗莫顿氏神经瘤的相关研究。因此,PRP是否可应用于莫顿氏神经瘤的临床治疗,尚需进一步验证。 "

| [1] SCHOLZ J, FINNERUP NB, ATTAL N, et al. The IASP classification of chronic pain for ICD-11: chronic neuropathic pain. Pain. 2019;160(1): 53-59. [2] KUFFLER DP. Mechanisms for Reducing Neuropathic Pain. Mol Neurobiol. 2020;57(1):67-87. [3] BOUHASSIRA D. Neuropathic pain: Definition, assessment and epidemiology. Rev Neurol (Paris). 2019;175(1-2):16-25. [4] FINNERUP NB, SINDRUP SH, JENSEN TS. The evidence for pharmacological treatment of neuropathic pain. Pain. 2010;150(3): 573-581. [5] LOVAGLIO AC, SOCOLOVSKY M, DI MASI G, et al. Treatment of neuropathic pain after peripheral nerve and brachial plexus traumatic injury. Neurol India. 2019;67(Supplement):S32-S37. [6] 朱谦, 樊碧发, 张达颖, 等. 周围神经病理性疼痛诊疗中国专家共识[J]. 中国疼痛医学杂志,2020,26(5):321-328. [7] KINGSLEY CS. Blood coagulation; evidence of an antagonist to factor VI in platelet-rich human plasma. Nature. 1954;173(4407):723-724. [8] CHAMATA ES, BARTLETT EL, WEIR D, et al. Platelet-Rich Plasma: Evolving Role in Plastic Surgery. Plast Reconstr Surg, 2021;147(1):219-230. [9] JUSTICZ N, DERAKHSHAN A, CHEN JX, et al. Platelet-Rich Plasma for Hair Restoration. Facial Plast Surg Clin North Am, 2020;28(2):181-187. [10] TANG R, WANG S, YANG J, et al. Application of platelet-rich plasma in traumatic bone infections. Expert Rev Anti Infect Ther. 2020:1-9. [11] SHAO S, PAN R, CHEN Y. Autologous Platelet-Rich Plasma for Diabetic Foot Ulcer. Trends Endocrinol Metab. 2020;31(12):885-890. [12] EVERTS P, ONISHI K, JAYARAM P, et al. Platelet-Rich Plasma: New Performance Understandings and Therapeutic Considerations in 2020. Int J Mol Sci. 2020;21(20):7794-7830. [13] ORGANIZATION WHO. WHO releases new International Classification of Diseases (ICD 11).https://www.who.int/news-room/detail/18-06-2018-who-releases-new-international-classification-ofdiseases-(icd-11). [14] 吕岩, 程建国, 樊碧发, 等. ICD-11慢性疼痛分类中文编译版[J]. 中国疼痛医学杂志,2018,24(11):801-805. [15] 陈军, 王江林. 国际疼痛学会对世界卫生组织ICD-11慢性疼痛分类的修订与系统化分类[J]. 中国疼痛医学杂志,2019,25(5):323-330. [16] WHITE FA, JUNG H, MILLER RJ. Chemokines and the pathophysiology of neuropathic pain. Proc Natl Acad Sci USA. 2007;104(51):20151-20158. [17] MCLACHLAN EM, JANIG W, DEVOR M, et al. Peripheral nerve injury triggers noradrenergic sprouting within dorsal root ganglia. Nature. 1993;363(6429):543-546. [18] WOOLF CJ, SHORTLAND P, COGGESHALL RE. Peripheral nerve injury triggers central sprouting of myelinated afferents. Nature. 1992; 355(6355):75-78. [19] WOOLF CJ. Evidence for a central component of post-injury pain hypersensitivity. Nature. 1983;306(5944):686-688. [20] FINNERUP NB, KUNER R, JENSEN TS. Neuropathic Pain: From Mechanisms to Treatment. Physiol Rev. 2021;101(1):259-301. [21] SEVCIK MA, GHILARDI JR, PETERS CM, et al. Anti-NGF therapy profoundly reduces bone cancer pain and the accompanying increase in markers of peripheral and central sensitization. Pain. 2005;115(1-2): 128-141. [22] JI RR, BERTA T, NEDERGAARD M. Glia and pain: is chronic pain a gliopathy? Pain. 2013;154 (1):S10-S28. [23] MARX RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10(4):225-228. [24] DOHAN ED, RASMUSSON L, ALBREKTSSON T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009;27(3): 158-167. [25] KIEB M, SANDER F, PRINZ C, et al. Platelet-Rich Plasma Powder: A New Preparation Method for the Standardization of Growth Factor Concentrations. Am J Sports Med. 2017;45(4):954-960. [26] CASTILLO TN, POULIOT MA, KIM HJ, et al. Comparison of growth factor and platelet concentration from commercial platelet-rich plasma separation systems. Am J Sports Med. 2011;39(2):266-271. [27] 卫愉轩, 张昭远, 范峥莹, 等. 中国富血小板血浆临床制备方法的研究进展[J]. 中华关节外科杂志(电子版),2020,14(2):196-200. [28] SLICHTER SJ, HARKER LA. Preparation and storage of platelet concentrates. Transfusion. 1976;16(1):8-12. [29] TAMIMI FM, MONTALVO S, TRESGUERRES I, et al. A comparative study of 2 methods for obtaining platelet-rich plasma. J Oral Maxillofac Surg. 2007;65(6):1084-1093. [30] CHAHLA J, CINQUE ME, PIUZZI NS, et al. A Call for Standardization in Platelet-Rich Plasma Preparation Protocols and Composition Reporting: A Systematic Review of the Clinical Orthopaedic Literature. J Bone Joint Surg Am. 2017;99(20):1769-1779. [31] DO AR, DA SN, HADDAD NF, et al. Platelet-Rich Plasma Obtained with Different Anticoagulants and Their Effect on Platelet Numbers and Mesenchymal Stromal Cells Behavior In Vitro. Stem Cells Int. 2016; 2016(3):1-11. [32] KRAUS M, NEEB H, STRASSER E. ACD vs. sodium-citrate as an anticoagulant for platelet rich plasma (PRP) preparation influencestheextent of platelet shape change during spreading-quantitative morphometric data from standardized robotic darkfield microscopy. Hamostaseologie. 2018;38:A66-A67. [33] DU L, MIAO Y, LI X, et al. A Novel and Convenient Method for the Preparation and Activation of PRP without Any Additives: Temperature Controlled PRP. Biomed Res Int. 2018;2018:1761865. [34] NLÜ A, YILMAZ S, EKER B, et al. A Cycle of Freezing and Thawing as a Modified Method for Activating Platelets in Platelet-rich Plasma to Use in Regenerative Medicine. Panam J Trauma Crit Care Emerg Surg. 2020;9(2):101-104. [35] TUNALI M, OZDEMIR H, KUCUKODACI Z, et al. A novel platelet concentrate: titanium-prepared platelet-rich fibrin. Biomed Res Int. 2014;2014:209548. [36] Bordon Y. Innate immunity: Platelets on the prowl. Nat Rev Immunol. 2018;18(1):3. [37] PARK HB, YANG JH, CHUNG KH. Characterization of the cytokine profile of platelet rich plasma (PRP) and PRP-induced cell proliferation and migration: Upregulation of matrix metalloproteinase-1 and -9 in HaCaT cells. Korean J Hematol. 2011;46(4):265-273. [38] MITANI N, YUJIRI T, TANAKA Y, et al. Hematopoietic progenitor cell count, but not immature platelet fraction value, predicts successful harvest of autologous peripheral blood stem cells. J Clin Apher. 2011;26(3):105-110. [39] KUFFLER DP. Platelet-rich plasma and the elimination of neuropathic pain. Mol Neurobiol. 2013;48(2):315-332. [40] MEACHAM K, SHEPHERD A, MOHAPATRA DP, et al. Neuropathic Pain: Central vs. Peripheral Mechanisms. Curr Pain Headache Rep. 2017; 21(6):28. [41] ELLIS A, BENNETT DL. Neuroinflammation and the generation of neuropathic pain. Br J Anaesth. 2013;111(1):26-37. [42] HERTER JM, ROSSAINT J, ZARBOCK A. Platelets in inflammation and immunity. J Thromb Haemost. 2015;12(11):1764-1775. [43] EL-SHARKAWY H, KANTARCI A, DEADY J, et al. Platelet-rich plasma: growth factors and pro- and anti-inflammatory properties. J Periodontol. 2007;78(4):661-669. [44] HAGIHARA M, HIGUCHI A, TAMURA N, et al. Platelets, after exposure to a high shear stress, induce IL-10-producing, mature dendritic cells in vitro. J Immunol. 2004;172(9):5297-5303. [45] PARK JS, LEE EJ, LEE JC, et al. Anti-inflammatory effects of short chain fatty acids in IFN-gamma-stimulated RAW 264.7 murine macrophage cells: involvement of NF-kappaB and ERK signaling pathways. Int Immunopharmacol. 2007;7(1):70-77. [46] SANCHEZ M, ANITUA E, DELGADO D, et al. Platelet-rich plasma, a source of autologous growth factors and biomimetic scaffold for peripheral nerve regeneration. Expert Opin Biol Ther. 2017;17(2):197-212. [47] MERIGHI A. Targeting the glial-derived neurotrophic factor and related molecules for controlling normal and pathologic pain. Expert Opin Ther Targets. 2016;20(2):193-208. [48] AL-MASSRI KF, AHMED LA, EL-ABHAR HS. Mesenchymal stem cells therapy enhances the efficacy of pregabalin and prevents its motor impairment in paclitaxel-induced neuropathy in rats: Role of Notch1 receptor and JAK/STAT signaling pathway. Behav Brain Res. 2019;360: 303-311. [49] ZHENG C, ZHU Q, LIU X, et al. Effect of platelet-rich plasma (PRP) concentration on proliferation, neurotrophic function and migration of Schwann cells in vitro. J Tissue Eng Regen Med. 2016;10(5):428-436. [50] LAI F, KAKUDO N, MORIMOTO N, et al. Platelet-rich plasma enhances the proliferation of human adipose stem cells through multiple signaling pathways. Stem Cell Res Ther. 2018;9(1):107. [51] ZOR F, DEVECI M, KILIC A, et al. Effect of VEGF gene therapy and hyaluronic acid film sheath on peripheral nerve regeneration. Microsurgery. 2014;34(3):209-216. [52] MAMMOTO T, JIANG A, JIANG E, et al. Platelet rich plasma extract promotes angiogenesis through the angiopoietin1-Tie2 pathway. Microvasc Res. 2013;89:15-24. [53] CASTRO MV, SILVA M, CHIAROTTO GB, et al. Reflex arc recovery after spinal cord dorsal root repair with platelet rich plasma (PRP). Brain Res Bull. 2019;152:212-224. [54] NEWTON K, DIXIT VM. Signaling in innate immunity and inflammation. Cold Spring Harb Perspect Biol. 2012;4(3):829-841. [55] COGNASSE F, LARADI S, BERTHELOT P, et al. Platelet Inflammatory Response to Stress. Front Immunol. 2019;10:1478. [56] GROS A, SYVANNARATH V, LAMRANI L, et al. Single platelets seal neutrophil-induced vascular breaches via GPVI during immune-complex-mediated inflammation in mice. Blood. 2015;126(8):1017-1026. [57] GILS JMV, ZWAGINGA JJ, HORDIJK PL. Molecular and functional interactions among monocytes, platelets, and endothelial cells and their relevance for cardiovascular diseases. J Leukoc Biol. 2008;85(2): 195-204. [58] ACOSTA-RODRIGUEZ EV, NAPOLITANI G, LANZAVECCHIA A, et al. Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells. Nat Immunol. 2007;8(9):942-949. [59] HENN V, SLUPSKY JR, GRAFE M, et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature. 1998; 391(6667):591-594. [60] WANG S, YANG J, ZHAO G, et al. Current applications of platelet gels in wound healing-A review. Wound Repair Regen. 2021;18(1):27-34. [61] ODEM MA, BAVENCOFFE AG, CASSIDY RM, et al. Isolated nociceptors reveal multiple specializations for generating irregular ongoing activity associated with ongoing pain. Pain. 2018;159(11):2347-2362. [62] NICHOLSON R, SMALL J, DIXON AK, et al. Serotonin receptor mRNA expression in rat dorsal root ganglion neurons. Neurosci Lett. 2003; 337(3):119-122. [63] KAMP J, VAN VELZEN M, OLOFSEN E, et al. Pharmacokinetic and pharmacodynamic considerations for NMDA-receptor antagonist ketamine in the treatment of chronic neuropathic pain: an update of the most recent literature. Expert Opin Drug Metab Toxicol. 2019; 15(12):1033-1041. [64] AIYER R, MEHTA N, GUNGOR S, et al. A Systematic Review of NMDA Receptor Antagonists for Treatment of Neuropathic Pain in Clinical Practice. Clin J Pain. 2018;34(5):450-467. [65] WONG HH, RANNIO S, JONES V, et al. NMDA receptors in axons: there’s no coincidence. J Physiol. 2021;599(2):367-387. [66] GUO W, CHU YX, IMAI S, et al. Further observations on the behavioral and neural effects of bone marrow stromal cells in rodent pain models. Mol Pain. 2016;12:1744806916658043. [67] FRANCONI F, MICELI M, ALBERTI L, et al. Further insights into the anti-aggregating activity of NMDA in human platelets. Br J Pharmacol. 1998; 124(1):35-40. [68] TSUDA M. Modulation of Pain and Itch by Spinal Glia. Neurosci Bull. 2018;34(1):178-185. [69] YANG PP, CHUEH SH, SHIE HL, et al. Effects of Hericium erinaceus Mycelium Extracts on the Functional Activity of Purinoceptors and Neuropathic Pain in Mice with L5 Spinal Nerve Ligation. Evid Based Complement Alternat Med. 2020;2020(3):1-12. [70] COHEN SP, MAO J. Neuropathic pain: mechanisms and their clinical implications. BMJ. 2014;348:f7656. [71] MIKA J, ZYCHOWSKA M, POPIOLEK-BARCZYK K, et al. Importance of glial activation in neuropathic pain. Eur J Pharmacol. 2013;716(1-3): 106-119. [72] BORHANI-HAGHIGHI M, MOHAMADI Y. The therapeutic effect of platelet-rich plasma on the experimental autoimmune encephalomyelitis mice. J Neuroimmunol. 2019;333:476958. [73] GIUSTI I, D’ASCENZO S, MACCHIARELLI G, et al. In vitro evidence supporting applications of platelet derivatives in regenerative medicine. Blood Transfus, 2020;18(2):117-129. [74] SIDDALL PJ, MCCLELLAND JM, RUTKOWSKI SB, et al. A longitudinal study of the prevalence and characteristics of pain in the first 5 years following spinal cord injury. Pain. 2003;103(3):249-257. [75] MICHETTI F, D’AMBROSI N, TOESCA A, et al. The S100B story: from biomarker to active factor in neural injury. J Neurochem. 2019;148(2): 168-187. [76] SAYO A, KONISHI H, KOBAYASHI M, et al. GPR34 in spinal microglia exacerbates neuropathic pain in mice. J Neuroinflammation. 2019; 16(1):82. [77] WANG A, XU C. The role of connexin43 in neuropathic pain induced by spinal cord injury. Acta Biochim Biophys Sin (Shanghai). 2019;51(6): 555-561. [78] UTTAM S, WONG C, AMORIM IS, et al. Translational profiling of dorsal root ganglia and spinal cord in a mouse model of neuropathic pain. Neurobiol Pain. 2018;4:35-44. [79] MIRANPURI GS, NGUYEN J, MORENO N, et al. Folic Acid Modulates Matrix Metalloproteinase-9 Expression Following Spinal Cord Injury. Ann Neurosci. 2019;26(2):60-65. [80] GWAK YS, HULSEBOSCH CE, LEEM JW. Neuronal-Glial Interactions Maintain Chronic Neuropathic Pain after Spinal Cord Injury. Neural Plast. 2017;2017:2480689. [81] SHEHADI JA, ELZEIN SM, BEERY P, et al. Combined administration of platelet rich plasma and autologous bone marrow aspirate concentrate for spinal cord injury: a descriptive case series. Neural Regen Res. 2021; 16(2):362-366. [82] SALARINIA R, HOSSEINI M, MOHAMADI Y, et al. Combined use of platelet-rich plasma and adipose tissue-derived mesenchymal stem cells shows a synergistic effect in experimental spinal cord injury. J Chem Neuroanat. 2020;110:101870. [83] CHEN NF, SUNG CS, WEN ZH, et al. Therapeutic Effect of Platelet-Rich Plasma in Rat Spinal Cord Injuries. Front Neurosci. 2018;12:252-264. [84] SALARINIA R, SADEGHNIA HR, ALAMDARI DH, et al. Platelet rich plasma: Effective treatment for repairing of spinal cord injury in rat. Acta Orthop Traumatol Turc. 2017;51(3):254-257. [85] DE CASTRO MV, DA SM, CHIAROTTO GB, et al. Spinal Reflex Recovery after Dorsal Rhizotomy and Repair with Platelet-Rich Plasma (PRP) Gel Combined with Bioengineered Human Embryonic Stem Cells (hESCs). Stem Cells Int. 2020;2020:8834360. [86] 冯振奋, 周宾宾, 魏卫兵, 等. 脊髓损伤后疼痛治疗的研究进展[J]. 中国康复理论与实践,2019,25(6):652-656. [87] GRIFFIN JW, HOGAN MV, CHHABRA AB, et al. Peripheral nerve repair and reconstruction. J Bone Joint Surg Am. 2013;95(23):2144-2151. [88] LI L, CAI J, YUAN Y, et al. Platelet-rich plasma can release nutrient factors to promote facial nerve crush injury recovery in rats. Saudi Med J. 2019;40(12):1209-1217. [89] 朱亚琼, 金壮, 汪靖, 等. 超声引导注射富血小板血浆修复坐骨神经挤压伤[J]. 中国组织工程研究,2020,24(20):3196-3201. [90] ZHU Y, JIN Z, FANG J, et al. Platelet-Rich Plasma Combined with Low-Dose Ultrashort Wave Therapy Accelerates Peripheral Nerve Regeneration. Tissue Eng Part A. 2020;26(3-4):178-192. [91] IKUMI A, HARA Y, OKANO E, et al. Intraoperative Local Administration of Platelet-Rich Plasma (PRP) during Neurolysis Surgery for the Treatment of Digital Nerve Crush Injury. Case Rep Orthop. 2018;2018:1275713. [92] 王兴平.富血小板血浆在后路颈椎减压后C5神经麻痹治疗中的价值[J].中国组织工程研究,2016,20(46):6930-6936. [93] HUANG SH, WU SH, LEE SS, et al. Platelet-Rich Plasma Injection in Burn Scar Areas Alleviates Neuropathic Scar Pain. Int J Med Sci. 2018;15(3): 238-247. [94] 中华医学会疼痛学分会. 脊柱退变性神经根疼痛治疗专家共识[J]. 中华医学杂志,2019,99(15):1133-1137. [95] BHATIA R, CHOPRA G. Efficacy of Platelet Rich Plasma via Lumbar Epidural Route in Chronic Prolapsed Intervertebral Disc Patients-A Pilot Study. J Clin Diagn Res. 2016;10(9):C5-C7. [96] BISE S, DALLAUDIERE B, PESQUER L, et al. Comparison of interlaminar CT-guided epidural platelet-rich plasma versus steroid injection in patients with lumbar radicular pain. Eur Radiol. 2020;30(6):3152-3160. [97] RUIZ-LOPEZ R, TSAI YC. A Randomized Double-Blind Controlled Pilot Study Comparing Leucocyte-Rich Platelet-Rich Plasma and Corticosteroid in Caudal Epidural Injection for Complex Chronic Degenerative Spinal Pain. Pain Pract. 2020;20(6):639-646. [98] LAM K, HUNG CY, WU TJ. Ultrasound-Guided Cervical Intradiscal Injection with Platelet-Rich Plasma with Fluoroscopic Validation for the Treatment of Cervical Discogenic Pain: A Case Presentation and Technical Illustration. J Pain Res. 2020;13:2125-2129. [99] SHIRI R, MIRANDA H, HELIOVAARA M, et al. Physical work load factors and carpal tunnel syndrome: a population-based study. Occup Environ Med. 2009;66(6):368-373. [100] SENNA MK, SHAAT RM, ALI A. Platelet-rich plasma in treatment of patients with idiopathic carpal tunnel syndrome. Clin Rheumatol. 2019; 38(12):3643-3654. [101] WU YT, HO TY, CHOU YC, et al. Six-month efficacy of platelet-rich plasma for carpal tunnel syndrome: A prospective randomized, single-blind controlled trial. Sci Rep. 2017;7(1):94. [102] RAEISSADAT SA, KARIMZADEH A, HASHEMI M, et al. Safety and efficacy of platelet-rich plasma in treatment of carpal tunnel syndrome; a randomized controlled trial. BMC Musculoskelet Disord. 2018;19(1): 49. [103] HASSANIEN M, ELAWAMY A, KAMEL EZ, et al. Perineural Platelet-Rich Plasma for Diabetic Neuropathic Pain, Could It Make a Difference? Pain Med. 2020;21(4):757-765. [104] ANJAYANI S, WIROHADIDJOJO YW, ADAM AM, et al. Sensory improvement of leprosy peripheral neuropathy in patients treated with perineural injection of platelet-rich plasma. Int J Dermatol. 2014;53(1): 109-113. [105] RAVINDRAN S, CRITON S. Sensory improvement of leprosy peripheral neuropathy in patients treated with perineural injection of platelet-rich plasma. Int J Dermatol. 2018;57(4):491-492. [106] 黄立荣, 郭佳妮, 张慧芝, 等. MRI导航下背根神经节脉冲射频联合富血小板血浆治疗带状疱疹后神经痛[J]. 中国疼痛医学杂志, 2020,26(6):459-462. [107] DE ANGELIS B, LUCARINI L, ORLANDI F, et al. Regenerative surgery of the complications with Morton’s neuroma surgery: use of platelet rich plasma and hyaluronic acid. Int Wound J. 2013;10(4):372-376. |
| [1] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [2] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [3] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [4] | Wang Kang, Zhi Xiaodong, Wang Wei. Effect and mechanism of human amniotic epithelial cells on nerve injury repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4046-4051. |
| [5] | Shi Yao, Han Shufeng, Yuan Yitong, Du Ruochen, Jing Zhijie, Zhao Bichun, Zhang Ruxin, Zhang Yujuan, Wang Chunfang. Efficacy and safety of human umbilical cord mesenchymal stem cells in the treatment of spinal cord injury: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4093-4100. |
| [6] | Chen Pingbo, Wang Jing, Sun Yong, Xu Xiaofeng, Chen Qian, Zhang Zhijian. Genipin crosslinked sonic hedgehog composite fibrin scaffolds for the repair of spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3345-3350. |
| [7] | Li Jianfeng, Zhang Shulian, Yan Jinyu, Li jiayi, Jin Zhao, Deng Qi. Vimentin silenced by small interfering RNA inhibits glial scar formation in a rat model of acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3190-3195. |
| [8] | Sun Xinzheng, Chen Xiaoke, Wang Chenghao, He Hui. Exercise improves pain induced by sciatic nerve injury in animal models: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 321-328. |
| [9] | Li Chuanhong, Yu Xing, Yang Yongdong, Zhao He. Microglia in spinal cord injury: M1/M2 phenotypic polarization and neurotoxic/neuroprotective effects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2265-2272. |
| [10] | Zheng Youfei, Shi Wentao, Liu Xiaogu, Zhuang Qin, Lü Demin, Zhang Jiayin, Yuan Yiwen, Zhang Zhijian, Xu Xiaofeng. Biocompatibility of plant-derived nerve conduits to nasal ectomesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2040-2044. |
| [11] | Li Shuai, Fan Yiming, Liu Fangyu, Zhang Hongyu, Wang Yansong. Effect and mechanism of astrocytes in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2062-2068. |
| [12] | Sun Jianwei, Yang Xinming, An Xiaogang. Effects of various methods on improving bone marrow mesenchymal stem cell transplantation for spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2075-2080. |
| [13] | Gong Chao, Zhang Yuqiang, Wang Wei. Role and mechanism of cell therapy in repair of peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2114-2119. |
| [14] | Duan Zhaoyuan, Wu Mingli, Luo Meng, Gao Jing, Li Ruiqing, Feng Xiaodong. Glutathione peroxidase 4 regulates neuronal ferroptosis after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(12): 1956-1962. |
| [15] | Jiang Shengyuan, Deng Bowen, Xu Lin, Liu Gang, He Feng, Zhao Yi, Ren Jingpei, Mu Xiaohong. Role and mechanism of tetramethylpyrazine in spinal cord injury repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1799-1804. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||