Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (7): 992-998.doi: 10.12307/2022.134
Previous Articles Next Articles
Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury
Hu Wei, Xie Xingqi, Tu Guanjun
- Department of Orthopedics, First Affiliated Hospital of China Medical University, Shenyang 110001, Liaoning Province, China
-
Received:2021-02-09Revised:2021-02-20Accepted:2021-03-24Online:2022-03-08Published:2021-10-29 -
Contact:Tu Guanjun, MD, Professor, Chief physician, Department of Orthopedics, First Affiliated Hospital of China Medical University, Shenyang 110001, Liaoning Province, China -
About author:Hu Wei, Master candidate, Department of Orthopedics, First Affiliated Hospital of China Medical University, Shenyang 110001, Liaoning Province, China -
Supported by:the Guidance Program of the Natural Science Foundation of Liaoning Province, No. 201602857 (to TGJ)
CLC Number:
Cite this article
Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
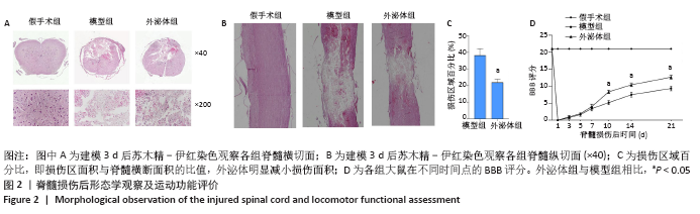
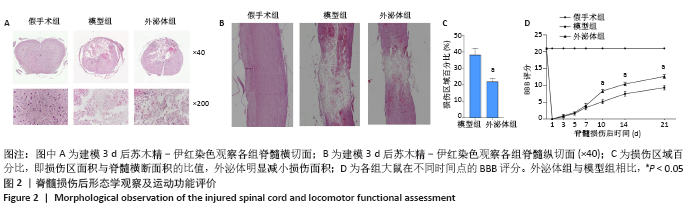
2.2 外泌体减轻脊髓病理损伤并促进运动功能恢复 脊髓损伤第3天,苏木精-伊红染色显示模型组损伤部位组织破裂、脊髓少量残留、红细胞及炎性细胞浸润、神经元丢失,可见损伤中心正常结构完全被破坏并伴有囊性腔及空洞。外泌体显著促进脊髓组织的修复,减少神经细胞丢失,见图2A。外泌体组和模型组的损伤百分比(%)分别为38.17±3.96,22.02±1.86 (P < 0.05),表明外泌体组表现出更好的完整性,见图2C。脊髓纵切面苏木精-伊红染色结果表明,模型组脊髓明显损伤,并且损伤沿脊髓长轴向外扩散;外泌体治疗后损伤面积明显缩小,并抑制损伤的扩散,见图2B。脊髓损伤后大鼠运动功能均受损,与模型组相比,外泌体组在损伤第10天后显著促进了运动功能恢复,术后第10天模型组与外泌体组的BBB评分分别为5.16±0.61,8.33±0.51(P < 0.05),见图2D。"
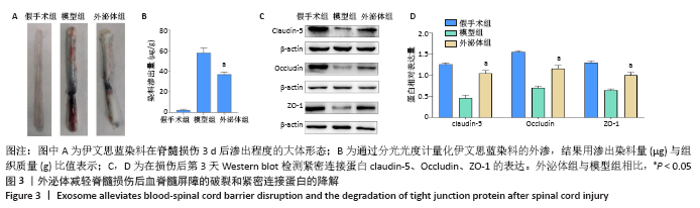
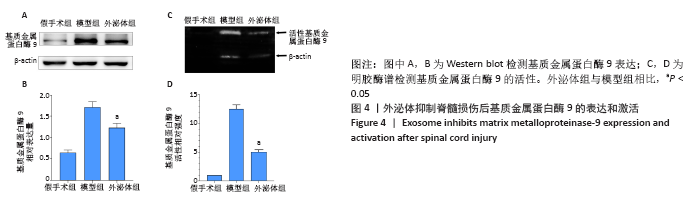
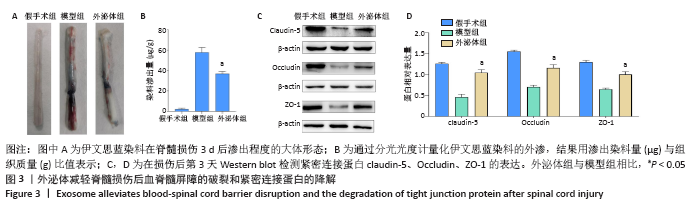
2.3 外泌体减少紧密连接蛋白的降解并减轻脊髓损伤后血脊髓屏障的破坏 在治疗后的第3天,使用伊文思蓝染料来评估血脊髓屏障的通透性。与假手术组相比,脊髓损伤导致伊文思蓝染料外渗量增加,见图3A,B,表明造模后血脊髓屏障被破坏。外泌体治疗可显著减少染料的渗出量,表明外泌体可减轻血脊髓屏障的破坏[模型组:(58.59±6.05) μg/g,外泌体组:(37.06±2.12) μg/g,P < 0.05]。血管内皮细胞中的紧密连接与血脊髓屏障的完整性密切相关,通过Western blot检测脊髓节段中紧密连接相关蛋白claudin-5、Occludin和ZO-1的表达。如图3C,D所示,相比于假手术组,脊髓损伤后紧密连接相关蛋白表达明显下降,外泌体治疗后紧密连接蛋白表达水平显著增加(P < 0.05)。结果表明,外泌体通过减少紧密连接蛋白的降解来保持血脊髓屏障的完整性。"
| [1] YILMAZ T, KAPTANOĞLU E. Current and future medical therapeutic strategies for the functional repair of spinal cord injury. World J Orthop. 2015;6(1):42-55. [2] PARK CS, LEE JY, CHOI HY, et al. Protocatechuic acid improves functional recovery after spinal cord injury by attenuating blood-spinal cord barrier disruption and hemorrhage in rats. Neurochem Int. 2019;124: 181-192. [3] BARTANUSZ V, JEZOVA D, ALAJAJIAN B, et al. The blood-spinal cord barrier: morphology and clinical implications. Ann Neurol. 2011;70(2): 194-206. [4] FAN B, WEI Z, YAO X, et al. Microenvironment Imbalance of Spinal Cord Injury. Cell Transplant. 2018;27(6):853-866. [5] FIGLEY SA, KHOSRAVI R, LEGASTO JM, et al. Characterization of vascular disruption and blood-spinal cord barrier permeability following traumatic spinal cord injury. J Neurotrauma. 2014;31(6):541-552. [6] ZLOKOVIC BV. The blood-brain barrier in health and chronic neurodegenerative disorders. Neuron. 2008;57(2):178-201. [7] LEE JY, CHOI HY, YUNE TY. Fluoxetine and vitamin C synergistically inhibits blood-spinal cord barrier disruption and improves functional recovery after spinal cord injury. Neuropharmacology. 2016;109:78-87. [8] MARTINS T, BAPTISTA S, GONÇALVES J, et al. Methamphetamine transiently increases the blood-brain barrier permeability in the hippocampus: role of tight junction proteins and matrix metalloproteinase-9. Brain Res. 2011;1411:28-40. [9] GONG Z, XIA K, XU A, et al. Stem Cell Transplantation: A Promising Therapy for Spinal Cord Injury. Curr Stem Cell Res Ther. 2020;15(4): 321-331. [10] ROMANELLI P, BIELER L, SCHARLER C, et al. Extracellular Vesicles Can Deliver Anti-inflammatory and Anti-scarring Activities of Mesenchymal Stromal Cells After Spinal Cord Injury. Front Neurol. 2019;10:1225. [11] JEONG JO, HAN JW, KIM JM, et al. Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ Res. 2011;108(11):1340-1347. [12] YUAN X, WU Q, WANG P, et al. Exosomes Derived From Pericytes Improve Microcirculation and Protect Blood-Spinal Cord Barrier After Spinal Cord Injury in Mice. Front Neurosci. 2019;13:319. [13] KYURKCHIEV D, BOCHEV I, IVANOVA-TODOROVA E, et al. Secretion of immunoregulatory cytokines by mesenchymal stem cells. World J Stem Cells. 2014;6(5):552-570. [14] MAROTE A, TEIXEIRA FG, MENDES-PINHEIRO B, et al. MSCs-Derived Exosomes: Cell-Secreted Nanovesicles with Regenerative Potential. Front Pharmacol. 2016;7:231. [15] WIKLANDER OPB, BRENNAN MÁ, LÖTVALL J, et al. Advances in therapeutic applications of extracellular vesicles. Sci Transl Med. 2019;11(492):eaav8521. [16] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. [17] SHAO J, ZARO J, SHEN Y. Advances in Exosome-Based Drug Delivery and Tumor Targeting: From Tissue Distribution to Intracellular Fate. Int J Nanomedicine. 2020;15:9355-9371. [18] GURUNATHAN S, KANG MH, JEYARAJ M, et al. Review of the Isolation, Characterization, Biological Function, and Multifarious Therapeutic Approaches of Exosomes. Cells. 2019;8(4):307. [19] KORNILOV R, PUHKA M, MANNERSTRÖM B, et al. Efficient ultrafiltration-based protocol to deplete extracellular vesicles from fetal bovine serum. J Extracell Vesicles. 2018;7(1):1422674. [20] RONG Y, LIU W, LV C, et al. Neural stem cell small extracellular vesicle-based delivery of 14-3-3t reduces apoptosis and neuroinflammation following traumatic spinal cord injury by enhancing autophagy by targeting Beclin-1. Aging (Albany NY). 2019;11(18):7723-7745. [21] WANG L, PEI S, HAN L, et al. Mesenchymal Stem Cell-Derived Exosomes Reduce A1 Astrocytes via Downregulation of Phosphorylated NFκB P65 Subunit in Spinal Cord Injury. Cell Physiol Biochem. 2018;50(4): 1535-1559. [22] BASSO DM, BEATTIE MS, BRESNAHAN JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12(1):1-21. [23] WU P, ZHANG B, OCANSEY DKW, et al. Extracellular vesicles: A bright star of nanomedicine. Biomaterials. 2021;269:120467. [24] MENG W, HE C, HAO Y, et al. Prospects and challenges of extracellular vesicle-based drug delivery system: considering cell source. Drug Deliv. 2020; 27(1):585-598. [25] KJELL J, OLSON L. Rat models of spinal cord injury: from pathology to potential therapies. Dis Model Mech. 2016;9(10):1125-1137. [26] YU D, SUN R, SHEN D, et al. Nuclear heme oxygenase-1 improved the hypoxia-mediated dysfunction of blood-spinal cord barrier via the miR-181c-5p/SOX5 signaling pathway. Neuroreport. 2021;32(2):112-120. [27] REINHOLD AK, RITTNER HL. Barrier function in the peripheral and central nervous system-a review. Pflugers Arch. 2017;469(1):123-134. [28] KUMAR H, ROPPER AE, LEE SH, et al. Propitious Therapeutic Modulators to Prevent Blood-Spinal Cord Barrier Disruption in Spinal Cord Injury. Mol Neurobiol. 2017;54(5):3578-3590. [29] KUMAR H, JO MJ, CHOI H, et al. Matrix Metalloproteinase-8 Inhibition Prevents Disruption of Blood-Spinal Cord Barrier and Attenuates Inflammation in Rat Model of Spinal Cord Injury. Mol Neurobiol. 2018; 55(3):2577-2590. [30] SHENDE P, SUBEDI M. Pathophysiology, mechanisms and applications of mesenchymal stem cells for the treatment of spinal cord injury. Biomed Pharmacother. 2017;91:693-706. [31] MUKHAMEDSHINA YO, GRACHEVA OA, MUKHUTDINOVA DM, et al. Mesenchymal stem cells and the neuronal microenvironment in the area of spinal cord injury. Neural Regen Res. 2019;14(2):227-237. [32] HA XQ, YANG B, HOU HJ, et al. Protective effect of rhodioloside and bone marrow mesenchymal stem cells infected with HIF-1-expressing adenovirus on acute spinal cord injury. Neural Regen Res. 2020;15(4):690-696. [33] ZHOU Z, CHEN Y, ZHANG H, et al. Comparison of mesenchymal stromal cells from human bone marrow and adipose tissue for the treatment of spinal cord injury. Cytotherapy. 2013;15(4):434-448. [34] YANG Y, YE Y, SU X, et al. MSCs-Derived Exosomes and Neuroinflammation, Neurogenesis and Therapy of Traumatic Brain Injury. Front Cell Neurosci. 2017;11:55. [35] XU G, AO R, ZHI Z, et al. miR-21 and miR-19b delivered by hMSC-derived EVs regulate the apoptosis and differentiation of neurons in patients with spinal cord injury. J Cell Physiol. 2019;234(7):10205-10217. [36] HUANG JH, YIN XM, XU Y, et al. Systemic Administration of Exosomes Released from Mesenchymal Stromal Cells Attenuates Apoptosis, Inflammation, and Promotes Angiogenesis after Spinal Cord Injury in Rats. J Neurotrauma. 2017;34(24):3388-3396. [37] JIANG D, GONG F, GE X, et al. Neuron-derived exosomes-transmitted miR-124-3p protect traumatically injured spinal cord by suppressing the activation of neurotoxic microglia and astrocytes. J Nanobiotechnology. 2020;18(1):105. [38] RUPPERT KA, NGUYEN TT, PRABHAKARA KS, et al. Human Mesenchymal Stromal Cell-Derived Extracellular Vesicles Modify Microglial Response and Improve Clinical Outcomes in Experimental Spinal Cord Injury. Sci Rep. 2018;8(1):480. [39] HUANG JH, XU Y, YIN XM, et al. Exosomes Derived from miR-126-modified MSCs Promote Angiogenesis and Neurogenesis and Attenuate Apoptosis after Spinal Cord Injury in Rats. Neuroscience. 2020;424: 133-145. [40] LU Y, ZHOU Y, ZHANG R, et al. Bone Mesenchymal Stem Cell-Derived Extracellular Vesicles Promote Recovery Following Spinal Cord Injury via Improvement of the Integrity of the Blood-Spinal Cord Barrier. Front Neurosci. 2019;13:209. [41] STERNLICHT MD, WERB Z. How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol. 2001;17:463-516. [42] LEE JY, CHOI HY, PARK CS, et al. Mithramycin A Improves Functional Recovery by Inhibiting BSCB Disruption and Hemorrhage after Spinal Cord Injury. J Neurotrauma. 2018;35(3):508-520. [43] NOBLE LJ, DONOVAN F, IGARASHI T, et al. Matrix metalloproteinases limit functional recovery after spinal cord injury by modulation of early vascular events. J Neurosci. 2002;22(17):7526-7535. [44] LEE JY, CHOI HY, NA WH, et al. Ghrelin inhibits BSCB disruption/hemorrhage by attenuating MMP-9 and SUR1/TrpM4 expression and activation after spinal cord injury. Biochim Biophys Acta. 2014;1842 (12 Pt A):2403-2412. [45] BECK KD, NGUYEN HX, GALVAN MD, et al. Quantitative analysis of cellular inflammation after traumatic spinal cord injury: evidence for a multiphasic inflammatory response in the acute to chronic environment. Brain. 2010;133(Pt 2):433-447. [46] YAO Y, XU J, YU T, et al. Flufenamic acid inhibits secondary hemorrhage and BSCB disruption after spinal cord injury. Theranostics. 2018;8(15): 4181-4198. [47] SAIWAI H, OHKAWA Y, YAMADA H, et al. The LTB4-BLT1 axis mediates neutrophil infiltration and secondary injury in experimental spinal cord injury. Am J Pathol. 2010;176(5):2352-2366. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1316-1322. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1323-1329. |
| [3] | Kan Houming, Fan Lijun, Chen Xuetai, Shen Wen. Application of platelet-rich plasma in neuropathic pain [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1345-1353. |
| [4] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [5] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [6] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [7] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [8] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [9] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [10] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [11] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1032-1037. |
| [12] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [13] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [14] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1051-1056. |
| [15] | Xiong Tinglin, Ying Menghui, Zhang Lisha, Zhang Xiaogang, Yang Yan. Electrophysiological characteristics of cardiomyocytes differentiated from induced pluripotent stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1063-1067. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||