Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (13): 2062-2068.doi: 10.12307/2022.334
Previous Articles Next Articles
Effect and mechanism of astrocytes in spinal cord injury
Li Shuai, Fan Yiming, Liu Fangyu, Zhang Hongyu, Wang Yansong
- Fifth Department of Orthopedics, First Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China
-
Received:2020-11-23Revised:2020-11-28Accepted:2020-12-31Online:2022-05-08Published:2021-12-20 -
Contact:Wang Yansong, MD, Chief physician, Fifth Department of Orthopedics, First Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China -
About author:Li Shuai, Master candidate, Fifth Department of Orthopedics, First Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China -
Supported by:National Natural Science Foundation of China, No. 81871781 (to WYS)
CLC Number:
Cite this article
Li Shuai, Fan Yiming, Liu Fangyu, Zhang Hongyu, Wang Yansong. Effect and mechanism of astrocytes in spinal cord injury[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2062-2068.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
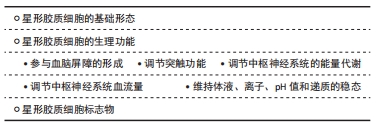
2.1.1 星形胶质细胞的基础形态 在大部分中枢神经系统区域中,星形胶质细胞的发育和产生通常滞后于神经元。由于细胞形态和解剖位置的不同,星形胶质细胞可分为2种亚型:原生质体星形胶质细胞和纤维状星形胶质细胞。原生质体星形胶质细胞存在于所有灰质中,形态呈多个茎分枝样突起。纤维状星形胶质细胞遍布于所有的白质中,形态呈长纤维样突起。神经解剖学研究表明2种亚型星形胶质细胞都与血管有广泛的接触,电子显微镜分析则表明2种亚型星形胶质细胞都以缝隙连接的方式与邻近星形胶质细胞的远端突起相连。在哺乳动物的中枢神经系统中,星形胶质细胞可通过同步神经元放电和协调突触网络实现调节神经元的功能。HALASSA等[4-5]进行了星形胶质细胞的三维结构重建,发现了星形胶质细胞以一种连续、有序、不重叠的方式平铺在整个中枢神经系统中,并通过单神经元染色方法估计出1个星形胶质细胞接触300-600个神经元树突,这一研究有助于进一步了解哺乳动物神经元与星形胶质细胞之间的相互作用。 2.1.2 星形胶质细胞的生理功能 (1)参与血脑屏障的形成:血脑屏障是指血浆和脑脊液之间的屏障,其可以根据极性和分子大小阻止某些物质流入脑实质,从而可保持脑组织内环境的基本稳定。血脑屏障的主要细胞成分是大脑毛细血管内皮细胞,它们形成紧密的连接,并被基板、血管周细胞和星形胶质细胞末端所包围[6]。 ISRAELOV等[7]将毛细血管内皮细胞和星形胶质细胞共培养,发现了星形胶质细胞可以诱导内皮细胞屏障特性。此外,GOVIER-COLE等[8]使用基因敲除小鼠模型研究了BMPIA型受体(BMPRIA)介导的骨形态发生蛋白在神经发育中的作用,并证明星形胶质细胞可通过BMPRIA调节血管内皮生长因子mRNA的表达,从而在血脑屏障的形成中发挥重要作用。相反,一些研究证明了星形胶质细胞的发育要比血脑屏障的形成要晚,BAUER等[9]研究了小鼠血脑屏障在胚胎发育过程中的血管形成情况以及相关形态学特征,发现血脑屏障的形成先于星形胶质细胞的发育。在血脑屏障的形成过程中,针对星形胶质细胞的研究还需进一步深入,以确定可诱导脑内皮细胞血脑屏障特性的分子介质,这些分子介质也可能在发育过程中的不同时间点由不同类型的细胞提供。 (2)调节突触功能:大量研究表明,星形胶质细胞可通过改变细胞内Ca2+水平释放突触活性分子,如谷氨酸、ATP和γ-氨基丁酸等,这些分子在突触传递中直接发挥作用并可改变神经元的兴奋性[10],这些研究也促使“三方突触”假说的形成,该假说认为除了典型的神经元突触前和突触后之间信息传递外,星形胶质细胞也可与突触神经元元件进行信息传递,以对突触活动作出反应,发挥调节突触传递的功能,即脑功能实际上是由神经元和神经胶质细胞组成的细胞网络协同产生,这与传统公认的脑功能完全是由神经元活动产生的说法不同[11]。 (3)调节中枢神经系统的能量代谢:星形胶质细胞在中枢神经系统代谢中发挥着重要作用,星形胶质细胞的突起一方面接触血管,另一方面接触神经元核膜、轴突和突触。星形胶质细胞既可以从血管中吸收葡萄糖,又能够将能量代谢产物提供给灰质和白质。研究证明星形胶质细胞是中枢神经系统中糖原颗粒的主要储存部位,PHELPS[12]在1971年研究了巴比妥酸盐对脑超微结构的影响,并证明中枢神经系统的糖原颗粒主要储存在突触密度较高区域的星形胶质细胞胞浆中。在不同时期,星形胶质细胞中的糖原为中枢神经系统提供能量的能力不同,BROWN等[13]发现星形胶质细胞内的糖原主要在低血糖和神经元活动较高的时期为神经元的活动提供能量。目前星形胶质细胞对于中枢神经系统能量代谢的机制尚未完全清楚,因此相关研究应在未来进一步深入。 (4)调节中枢神经系统血流量:作为中枢神经系统局部血流量变化的主要介质,星形胶质细胞与血管接触广泛,并释放可调节中枢神经系统血流量的血管活性物质,如ATP、一氧化氮、前列腺素E2和环氧-二十碳三烯酸等,这些物质可以协同增加或减少中枢神经系统血管直径从而调节血流量,以对神经元活动的变化作出反应[14-17]。 (5)维持体液、离子、pH值和递质的稳态:突触间质液体、离子、pH值和递质的稳态对突触的传递功能至关重要。文献报道星形胶质细胞突起在水通道蛋白4和摄取K+的转运体中非常丰富并基本包裹着所有突触[18],其中与血管接触的星形胶质细胞突起可与水通道蛋白4聚集,起到调节健康中枢神经系统中液体内稳态的作用[19]。 2.1.3 星形胶质细胞标志物 LENHOSSéK在1895年使用金和氯化汞升华染色技术来标记胶质纤维酸性蛋白,从而区分了星形胶质细胞和放射状胶质细胞,胶质纤维酸性蛋白现已成为星形胶质细胞免疫组织化学鉴定中最典型、最常用的标志物。但胶质纤维酸性蛋白的使用也有局限性,许多成熟星形胶质细胞在健康中枢神经系统组织中并不表达高水平的胶质纤维酸性蛋白,并且星形胶质细胞的胶质纤维酸性蛋白表达由于受大量细胞内信号分子的动态调节从而表现出区域和局部的可变性。HERRMANN等[20]使用Cre/loxP重组酶系统从星形胶质细胞中删除信号转导子及转录激活因子3(signal transduction and activator of transcription 3,STAT3),发现了胶质纤维酸性蛋白在反应性星形胶质细胞和胶质瘢痕形成过程中明显表达,但在健康的中枢神经系统中大多数星形胶质细胞表达胶质纤维酸性蛋白并不是必需的。除此之外,胶质纤维酸性蛋白在某些哺乳动物并不表达,如幼虫[21]。胶质纤维酸性蛋白可能不是星形胶质细胞的最准确标志物,但它提供了一种方便的检测方法,随着技术的进步和对星形胶质细胞认识的深入,脂质运载蛋白2(Lipocalin-2,LCN2)和丝氨酸肽酶抑制剂进化A成员3N(SERPINA3N)等已被证实为新的反应性星形胶质细胞标志物,Lcn2的表达短暂,其可能是反应性星形胶质细胞最早的标志物;而Serpina3n的表达至少在1周内保持相对高水平,因此其可能是更持久的反应性星形胶质细胞的标志物[22-23] 。 2.2 反应性星形胶质细胞 "
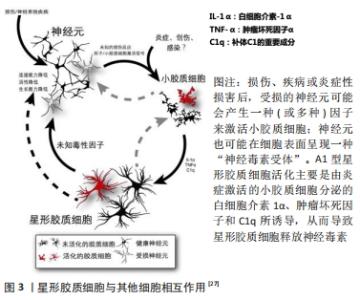
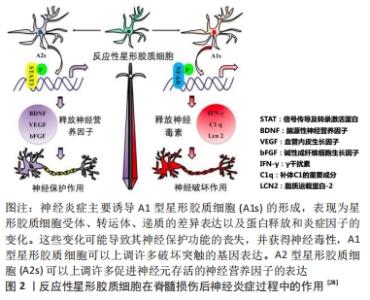
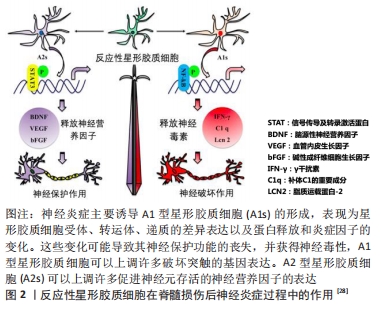
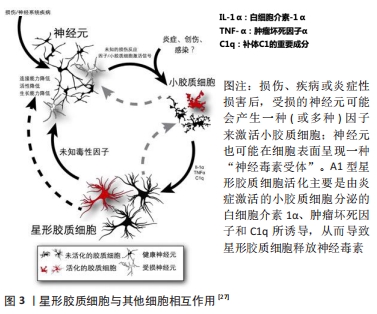
2012年,ZAMANIAN等[23]分别从脂多糖诱导的神经炎症模型和大脑中动脉结扎法建立的缺血模型中获取反应性星形胶质细胞,并对所获取的反应性星形胶质细胞的基因表达进行了分析,他们发现2种损伤模型产生了不同类型的反应性星形胶质细胞,这2种反应性星形胶质细胞都有一组核心基因上调,但至少有50%的基因表达为不同损伤类型特有,这一发现证明了反应性星形胶质细胞的极化类型很大程度上依赖于损伤类型。2017年,LIDDELOW等[26-27]提出反应性星形胶质细胞可根据其表型不同分为 A1型星形胶质细胞(A1s)和 A2 型星形胶质细胞(A2s),这与巨噬细胞/小胶质细胞的“M1/M2”分型相似。反应性星形胶质细胞有着高度的异质性,可以对不同的损伤做出反应,神经炎症发生时反应性星形胶质细胞向促炎的A1型星形胶质细胞极化,缺血缺氧时反应性星形胶质细胞向抑炎的A2 型星形胶质细胞极化,见图2[28]。目前认为A1型星形胶质细胞是一种有害的类型,其活化由炎症激活的小胶质细胞分泌的白细胞介素1α、肿瘤坏死因子和C1q三种因子所诱导。实验证明,敲除以上1个或2个因子时小胶质细胞不能诱导A1型星形胶质细胞的活化,因此这3种细胞因子的共同作用是A1型星形胶质细胞极化的必要条件。A1型星形胶质细胞失去了促神经元存活、突触生长、突触形成以及吞噬的能力,获得了诱导神经元、少突胶质细胞死亡的有害能力,对中枢神经系统有不利的促炎作用,其高度上调可破坏突触的基因表达(如补体级联基因),导致突触形成数量减少、功能减退。A1型星形胶质细胞还可能分泌一种可溶性毒素,导致中枢神经系统中神经元和成熟少突胶质细胞的死亡,见图3。补体成分C3在A1型星形胶质细胞中特异性升高,而在A2型星形胶质细胞中不表达,因此补体成分C3可作为A1型星形胶质细胞的表面标记物。"
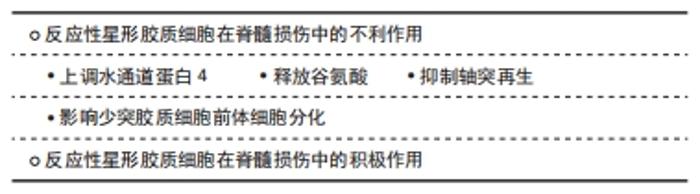
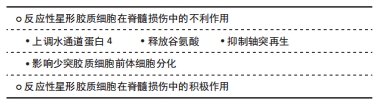
相反,A2 型星形胶质细胞被认为是一种保护型细胞,其可分泌多种抗炎因子和神经营养因子,发挥着对中枢神经系统有利的作用。S100A10为S100蛋白家族的成员,在细胞增殖、膜修复和抑制凋亡等多个方面发挥作用,现其已被证明是A2 型星形胶质细胞的表面标志物 [29]。近期,SONN等[30]还发现了由白细胞介素4诱导的M2型巨噬细胞可通过Wnt/β-catenin途径来刺激反应性星形胶质细胞的趋化,随着Wnt/β-catenin途径被抑制,反应性星形胶质细胞拉长的百分比显著减少,Wnt/β-catenin途径激动剂则可增加反应性星形胶质细胞平均长度,这一发现证实白细胞介素4诱导的M2型巨噬细胞是反应性星形胶质细胞极化的诱导剂,而单纯巨噬细胞和M1型巨噬细胞则无诱导作用。目前对反应性星形胶质细胞的了解还不全面,仍有大量问题需要解决,如反应性星形胶质细胞是否还有其他活化类型,A2 型星形胶质细胞的极化机制等。关于反应性星形胶质细胞在脊髓损伤中的研究需要进一步开展,以更好地了解脊髓损伤病理过程,明确反应性星形胶质细胞的活化机制,并为脊髓损伤治疗提供新的方向。 2.2.1 反应性星形胶质细胞在脊髓损伤中的不利作用 (1)上调水通道蛋白4:水通道蛋白4是中枢神经系统中最常见的水通道蛋白,为血管源性水肿的限速因子,其参与了星形胶质细胞的细胞毒性水肿形成。2019年HUANG等[31]证明了反应性星形胶质细胞可上调水通道蛋白4的表达,生物信息学分析表明水通道蛋白4与细胞色素氧化酶5(cytochrome coxidase 5,COX5A)之间存在一定的相关性,即抑制水通道蛋白4可导致星形胶质细胞中的COX5A表达显著上调,减轻细胞毒性水肿,这可能与COX5A影响能量代谢有关。 (2)释放谷氨酸:谷氨酸是介导兴奋性突触传递的主要形式,过度释放可导致神经毒性。OH等[32]证明,在蛋白酶激活受体1激活后,星形胶质细胞内钙离子浓度升高使Best1通道开放,从而导致谷氨酸释放,这揭示了一种新的由通道介导的谷氨酸释放机制,有助于进一步了解星形胶质细胞在脊髓损伤中的作用。 (3)抑制轴突再生:脊髓损伤后轴突再生一直是神经功能恢复的关键,反应性星形胶质细胞主要从2个方面影响轴突再生:形成阻碍轴突生长的瘢痕以及分泌抑制性蛋白。大量实验证明,在脊髓损伤的慢性期,星形胶质细胞与周细胞、血管周围成纤维细胞、室管膜细胞和外周免疫细胞等在病变周围形成可以隔离整个受损区域的胶质瘢痕,这是神经细胞再生的主要机械屏障[33]。在分泌抑制性蛋白方面,瘢痕组织中的反应性星形胶质细胞大量表达硫酸软骨素蛋白多糖,早在1991年,MCKEON等[34]证明在脊髓损伤后的24 h内,细胞外基质中沉积大量硫酸软骨素蛋白多糖,并在数个月内一直存在于损伤区域附近阻碍轴突再生。此外,硫酸软骨素蛋白多糖也可通过激活表皮生长因子受体对轴突生长产生抑制[35]。 (4)影响少突胶质细胞前体细胞分化:少突胶质细胞前体细胞是中枢神经系统中增殖最多、分布最广的前体细胞,在脊髓损伤后少突胶质细胞前体细胞可及时补充大量丢失的少突胶质细胞,并发挥极强的再髓鞘化能力,这对脊髓损伤后的神经功能恢复至关重要。2011年WANG等[36]发现来自大鼠损伤脊髓的反应性星形胶质细胞可抑制少突胶质细胞前体细胞的分化,且骨形态发生蛋白在反应性星形胶质细胞中的表达显著增加,使用骨形态发生蛋白受体拮抗剂noggin可逆转反应性星形胶质细胞对少突胶质细胞前体细胞分化的抑制作用,这表明调控骨形态发生蛋白信号可能是一种有效的脊髓损伤治疗策略。骨形态发生蛋白是一组具有类似结构的高度保守的功能蛋白,是体内诱导骨和软骨形成的主要因子,骨形态发生蛋白信号因其可促进骨骼的胚胎发育和再生修复以及抑制少突胶质细胞前体细胞向少突胶质细胞分化而广为人知。此外,脊髓损伤后沉积的硫酸软骨素蛋白多糖也可抑制少突胶质细胞前体细胞的生长和分化,这一过程可被硫酸软骨素酶ABC完全逆转[37]。2014年HAMMOND等[38]还发现了内皮素1在反应性星形胶质细胞中高表达,其可通过激活Notch通路抑制少突胶质细胞前体细胞的分化从而影响脊髓损伤再髓鞘化。反应性星形胶质细胞也可激活小胶质细胞加重对少突胶质细胞前体细胞的损害[35]。如何促进脊髓损伤后的再髓鞘化一直是需要攻克的难题,少突胶质细胞前体细胞在其中扮演了重要角色,减少反应性星形胶质细胞对少突胶质细胞前体细胞的影响可有效改善脊髓损伤后神经功能,相关研究在未来会持续开展,这对脊髓损伤后轴突的再髓鞘化有着重要意义。 2.2.2 反应性星形胶质细胞在脊髓损伤中的积极作用 OKADA等[39]发现反应性星形胶质细胞在脊髓损伤急性期可迅速迁移至损伤部位限制炎性细胞浸润并改善神经功能。在脊髓损伤后,神经暴露在含有高水平炎性物质的微环境中,这极其不利于受损脊髓的恢复,反应性星形胶质细胞形成的瘢痕组织则可阻断这一微环境,胶质瘢痕也可参与神经干细胞和神经前体细胞增殖及分化的调节,还可与细胞外基质共同募集成纤维细胞和内皮细胞,帮助损伤部位快速形成微血管[40]。星形胶质细胞限制脊髓损伤后炎症扩散与STAT3信号相关,脊髓损伤后星形胶质细胞及邻近细胞中STAT3表达明显上调,在STAT3缺失后胶质纤维酸性蛋白表达减弱,瘢痕形成明显中断,这使病变区域炎症反应扩散,轴突再生进一步被抑制,这表明STAT3是反应性星形胶质细胞瘢痕形成的关键调节因子[41]。CHEN等[42]发现亮氨酸拉链激酶(LZK,MAP3K13)促进星形胶质细胞形成胶质瘢痕,成年小鼠星形胶质细胞中LZK基因缺失减少胶质瘢痕的形成,导致脊髓损伤后损伤范围扩大。相反,LZK在星形胶质细胞中过表达可缩小损伤范围,这表明LZK是星形胶质细胞活性的关键细胞内调节因子。ROTHSTEIN等[43]则发现了星形胶质细胞能够吸收谷氨酸,从而减少其兴奋性毒性损伤。综上所述,在脊髓损伤后单纯消除星形胶质细胞可导致炎症扩散、神经元损害,并不利于神经功能的恢复。 2.3 治疗策略 "

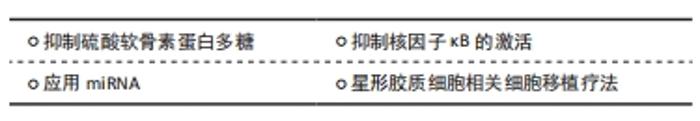
2.3.1 抑制硫酸软骨素蛋白多糖 糖胺聚糖侧链的蛋白质核心组成了硫酸软骨素蛋白多糖,消除糖胺聚糖侧链可抑制硫酸软骨素蛋白多糖活性,进而促进轴突生长,帮助神经功能恢复。2004年BRADBURY等[44]将硫酸软骨素酶ABC注入脊髓损伤后的成年大鼠,并首次发现鞘内注射硫酸软骨素酶ABC可去除糖胺聚糖并降解损伤部位的硫酸软骨素蛋白多糖,从而促进轴突再生,恢复突触活动。这一开创性研究引起人们对硫酸软骨素酶ABC的广泛关注,在这之后,硫酸软骨素酶ABC被证明对轴突的可塑性有促进作用,可以导致损伤处巨噬细胞呈抗炎表型,限制损伤区的炎症,其与其他疗法结合可进一步促进脊髓损伤后轴突的可塑性和神经功能的恢复[45]。最近BURNSIDE等[46]将硫酸软骨素酶ABC基因以酶传递的方法转入宿主细胞表达,消除了大量糖胺聚糖,有效改善了鼠脊髓损伤后神经功能,并可严格控制硫酸软骨素酶ABC基因的表达。此外,SUZUKI等[47]证明应用硫酸软骨素酶ABC之后移植神经干细胞可增加移植物存活率,并减少胶质瘢痕形成,从而更好地改善小鼠神经功能。硫酸软骨素酶ABC对于脊髓损伤的治疗研究日益深入,在未来的研究中应进一步了解硫酸软骨素酶ABC与其他疗法的协同效应,这可更好地改善脊髓损伤后的神经功能,还应进一步提高硫酸软骨素酶ABC的安全性,尽量降低其使用成本,寻找更经济的递送载体,以更好地发展硫酸软骨素酶ABC相关治疗方法。GRIMPE等[48]还发现敲除木糖基转移酶1的mRNA,可阻止其催化糖胺聚糖添加到硫酸软骨素蛋白多糖的核心蛋白,促进了脊髓损伤后神经功能的恢复。HART等[49]则证明了骨形态发生蛋白4的活性在大鼠脊髓损伤急性期强烈而短暂地上调,可导致反应性星形胶质细胞中硫酸软骨素蛋白多糖的产生增加,而阻断骨形态发生蛋白4后硫酸软骨素蛋白多糖的产生减少并促进了少突胶质细胞前体细胞的再生,这一发现证明骨形态发生蛋白4在调节继发性脊髓损伤中发挥作用,并且可作为改善内源性细胞反应和髓鞘再生的潜在靶点。更多的抑制硫酸软骨素蛋白多糖的治疗方法依然等待着发现,有理由相信未来会有更多的实验致力于此。 2.3.2 抑制核因子κB的激活 核因子κB蛋白在几乎所有的动物细胞中都有表达,它参与细胞对外界刺激的响应,在炎症反应、免疫应答等过程中核因子κB起关键作用。BRAMBILLA等[50]发现星形胶质细胞可介导核因子κB的激活,以核因子κB为靶点的治疗策略有望促进脊髓损伤后神经功能的恢复。VISMARA等[51]使用承载Rolipram的纳米凝胶抑制核因子κB,减少了A1型星形胶质细胞中脂质运载蛋白2和一氧化氮合酶等促炎物质的表达。骨髓间充质干细胞源性外泌体通过抑制核因子κB减少A1型星形胶质细胞的活化,减轻脊髓损伤的炎症反应[52-53]。这些研究结果证明,有效调节核因子κB可调控星形胶质细胞的活化过程,减轻脊髓损伤后神经炎症,并最终改善神经功能,核因子κB将成为脊髓损伤治疗的重要靶点。 2.3.3 应用miRNA 近年来miRNA被认为是参与脊髓损伤病理机制的重要因素,其中miR-21被证明可能是反应性星形胶质细胞的活化开关[54]。SU等[55]发现下调miR-21可促A1型星形胶质细胞转变为A2型星形胶质细胞,减少脊髓损伤后炎症反应,促进神经功能恢复。近期实验发现,miR-219/miR-338也可减少促炎性细胞因子表达,并抑制星形胶质细胞的活化,增强少突胶质细胞前体细胞的分化和成熟,可促进受损神经的功能恢复[56]。利用miRNA抑制A1型星形胶质细胞活化,减轻A1型星形胶质细胞促炎效应,有望成为脊髓损伤新的治疗靶点。 2.3.4 星形胶质细胞相关细胞移植疗法 大量研究表明反应性星形胶质细胞可与移植细胞产生协同作用促进轴突生长并发挥保护神经的作用[57-58]。KIM等[59]最近报道了一种3D培养系统,他们将星形胶质细胞限制在水凝胶微纤维中,并使星形胶质细胞不与神经元发生物理接触,促进了轴突的生长和形成,这一发现为脊髓损伤的细胞疗法提供了一种新的培养支架。此外,星形胶质细胞可在性别决定区Y框蛋白2(SOX2)和锌指蛋白521(Zfp521)转录因子刺激下重新编程并去分化为诱导神经干细胞(iNSCs)用于脊髓损伤治疗,但这一过程是不稳定且低效的,YANG等[60]发现持续的声波刺激可增强星形胶质细胞重编程过程,更有效地生成诱导神经干细胞。ZAREI-KHEIRABADI等[61]则发现Zfp521在重编程星形胶质细胞为诱导神经干细胞方面比SOX2更有效,在成年大鼠损伤脊髓中,驻留的星形胶质细胞可以被Zfp521重新编程为诱导神经干细胞,且这种治疗改善了大鼠的神经功能,其后肢、步长、脚趾伸展、足长和爪区的运动能力得到了增强,运动诱发电位记录也表明了大鼠的脊髓功能完整性。将星形胶质细胞重新编程的疗法在临床上面临的障碍较少,因为其无需外源性细胞移植,显著降低了排斥反应相关风险,但再编程效率目前依然较低,未来应进一步研究如何提高转化为诱导神经干细胞数量,这一疗法将成为脊髓损伤体内治疗的一把利器。 2.4 问题与展望 目前为止,对星形胶质细胞的认识仍不全面,还需克服如以下诸多问题:①目前研究只发现了A1和A2两种极化类型的反应性星形胶质细胞,是否还有其他极化类型仍需进一步探究;②A1型星形胶质细胞是否只有神经毒性,A2型星形胶质细胞是否只有神经保护功能,A1型星形胶质细胞和A2型星形胶质细胞之间有无中间形式;③A1型星形胶质细胞的活化机制现已初步明了,其主要由小胶质细胞分泌的白细胞介素1α、肿瘤坏死因子和C1q所诱导,但A2型星形胶质细胞的活化机制依然未知;④对于脊髓损伤,针对星形胶质细胞的众多治疗方法之间是否有协同性有待进一步论证。针对这些问题,更精密仪器、更先进的显微镜技术、单细胞测序技术和空间转录技术也许会提供新的方向。"

| [1] BELROSE JL, PRASAD A, SAMMONS MA, et al. Comparative gene expression profiling between optic nerve and spinal cord injury in Xenopus laevis reveals a core set of genes inherent in successful regeneration of vertebrate central nervous system axons. BMC Genomics. 2020;21(1):540. [2] MCRAE J, SMITH C, EMMANUEL A, et al. The experiences of individuals with cervical spinal cord injury and their family during post-injury care in non-specialised and specialised units in UK. BMC Health Serv Res. 2020; 20(1):783. [3] HACHEM LD, MOTHE AJ, TATOR CH. Unlocking the paradoxical endogenous stem cell response after spinal cord injury. Stem Cells. 2020;38(2):187-194. [4] HALASSA MM, FELLIN T, TAKANO H, et al. Synaptic islands defined by the territory of a single astrocyte. J Neurosci. 2007;27(24):6473-6477. [5] ROGERS RC, HASSER EM, HERMANN GE. Thrombin action on astrocytes in the hindbrain of the rat disrupts glycemic and respiratory control. Am J Physiol Regul Integr Comp Physiol. 2020;318(6):R1068-R1077. [6] DANEMAN R, PRAT A. The blood-brain barrier. Cold Spring Harb Perspect Biol. 2015;7(1):a020412. [7] ISRAELOV H, RAVID O, ATRAKCHI D,et al. Caspase-1 has a critical role in blood-brain barrier injury and its inhibition contributes to multifaceted repair. J Neuroinflammation. 2020;17(1):267. [8] GOVIER-COLE AE, WOOD RJ, FLETCHER JL, et al. Inhibiting Bone Morphogenetic Protein 4 Type I Receptor Signaling Promotes Remyelination by Potentiating Oligodendrocyte Differentiation. eNeuro. 2019;6(2):ENEURO.0399-18.2019. [9] BAUER HC, BAUER H, LAMETSCHWANDTNER A, et al. Neovascularization and the appearance of morphological characteristics of the blood-brain barrier in the embryonic mouse central nervous system. Brain Res Dev Brain Res. 1993;75(2):269-278. [10] LIU LR, LIU JC, BAO JS, et al. Interaction of Microglia and Astrocytes in the Neurovascular Unit. Front Immunol. 2020;11:1024. [11] GONZÁLEZ J, PINZÓN A, ANGARITA-RODRÍGUEZ A, et al. Advances in Astrocyte Computational Models: From Metabolic Reconstructions to Multi-omic Approaches. Front Neuroinform. 2020;14:35. [12] PHELPS CH. Barbiturate-induced glycogen accumulation in brain. An electron microscopic study. Brain Res. 1972;39(1):225-234. [13] BROWN AM, TEKKÖK SB, RANSOM BR. Glycogen regulation and functional role in mouse white matter. J Physiol. 2003;549(Pt 2):501-512. [14] GOLLIHUE JL, NORRIS CM. Astrocyte mitochondria: Central players and potential therapeutic targets for neurodegenerative diseases and injury. Ageing Res Rev. 2020;59:101039. [15] ISAKOVIĆ J, GORUP D, MITREČIĆ D. Molecular mechanisms of microglia- and astrocyte-driven neurorestoration triggered by application of electromagnetic fields. Croat Med J. 2019;60(2):127-140. [16] ZHANG L, ZHOU Z, ZHAI W, et al. Safflower yellow attenuates learning and memory deficits in amyloid β-induced Alzheimer’s disease rats by inhibiting neuroglia cell activation and inflammatory signaling pathways. Metab Brain Dis. 2019;34(3):927-939. [17] MISHRA A. Binaural blood flow control by astrocytes: listening to synapses and the vasculature. J Physiol. 2017;595(6):1885-1902. [18] DIVOLIS G, STAVROPOULOS A, MANIOUDAKI M, et al. Activation of both transforming growth factor-β and bone morphogenetic protein signalling pathways upon traumatic brain injury restrains pro-inflammatory and boosts tissue reparatory responses of reactive astrocytes and microglia. Brain Commun. 2019;1(1):fcz028. [19] LISJAK M, POTOKAR M, ZOREC R, et al. Indirect Role of AQP4b and AQP4d Isoforms in Dynamics of Astrocyte Volume and Orthogonal Arrays of Particles. Cells. 2020;9(3):735. [20] HERRMANN JE, IMURA T, SONG B, et al. STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci. 2008; 28(28):7231-7243. [21] SABIN KZ, ECHEVERRI K. The role of the immune system during regeneration of the central nervous system. J Immunol Regen Med. 2020;7:100023. [22] LEE S, JHA MK, SUK K. Lipocalin-2 in the Inflammatory Activation of Brain Astrocytes. Crit Rev Immunol. 2015;35(1):77-84. [23] ZAMANIAN JL, XU L, FOO LC, et al. Genomic analysis of reactive astrogliosis. J Neurosci. 2012;32(18):6391-6410. [24] ZHANG HY, WANG Y, HE Y, et al. A1 astrocytes contribute to murine depression-like behavior and cognitive dysfunction, which can be alleviated by IL-10 or fluorocitrate treatment. J Neuroinflammation. 2020;17(1):200. [25] SOFRONIEW MV. Astrocyte Reactivity: Subtypes, States, and Functions in CNS Innate Immunity. Trends Immunol. 2020;41(9):758-770. [26] LIDDELOW SA, GUTTENPLAN KA, CLARKE LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541(7638): 481-487. [27] LIDDELOW SA, BARRES BA. Reactive Astrocytes: Production, Function, and Therapeutic Potential. Immunity. 2017;46(6):957-967. [28] LI K, LI J, ZHENG J, et al. Reactive Astrocytes in Neurodegenerative Diseases. Aging Dis. 2019;10(3):664-675. [29] FUJITA A, YAMAGUCHI H, YAMASAKI R, et al. Connexin 30 deficiency attenuates A2 astrocyte responses and induces severe neurodegeneration in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride Parkinson’s disease animal model. J Neuroinflammation. 2018;15(1):227. [30] SONN I, NAKAMURA M, RENAULT-MIHARA F, et al. Polarization of Reactive Astrocytes in Response to Spinal Cord Injury is Enhanced by M2 Macrophage-Mediated Activation of Wnt/β-Catenin Pathway. Mol Neurobiol. 2020;57(4):1847-1862. [31] HUANG Y, LI SN, ZHOU XY, et al. The Dual Role of AQP4 in Cytotoxic and Vasogenic Edema Following Spinal Cord Contusion and Its Possible Association With Energy Metabolism via COX5A. Front Neurosci. 2019;13: 584. [32] OH SJ, HAN KS, PARK H, et al. Protease activated receptor 1-induced glutamate release in cultured astrocytes is mediated by Bestrophin-1 channel but not by vesicular exocytosis. Mol Brain. 2012;5:38. [33] O’REILLY ML, TOM VJ. Neuroimmune System as a Driving Force for Plasticity Following CNS Injury. Front Cell Neurosci. 2020;14:187. [34] MCKEON RJ, SCHREIBER RC, RUDGE JS, et al. Reduction of neurite outgrowth in a model of glial scarring following CNS injury is correlated with the expression of inhibitory molecules on reactive astrocytes. J Neurosci. 1991;11(11):3398-3411. [35] GAUDET AD, FONKEN LK. Glial Cells Shape Pathology and Repair After Spinal Cord Injury. Neurotherapeutics. 2018;15(3):554-577. [36] WANG Y, CHENG X, HE Q, et al. Astrocytes from the contused spinal cord inhibit oligodendrocyte differentiation of adult oligodendrocyte precursor cells by increasing the expression of bone morphogenetic proteins. J Neurosci. 2011;31(16):6053-6058. [37] SIEBERT JR, STELZNER DJ, OSTERHOUT DJ. Chondroitinase treatment following spinal contusion injury increases migration of oligodendrocyte progenitor cells. Exp Neurol. 2011;231(1):19-29. [38] HAMMOND TR, GADEA A, DUPREE J, et al. Astrocyte-derived endothelin-1 inhibits remyelination through notch activation. Neuron. 2014;81(3): 588-602. [39] OKADA S, NAKAMURA M, KATOH H, et al. Conditional ablation of Stat3 or Socs3 discloses a dual role for reactive astrocytes after spinal cord injury. Nat Med. 2006;12(7):829-834. [40] HE Y, LIU X, CHEN Z. Glial Scar-a Promising Target for Improving Outcomes After CNS Injury. J Mol Neurosci. 2020;70(3):340-352. [41] SRIRAM K, BENKOVIC SA, HEBERT MA, et al. Induction of gp130-related cytokines and activation of JAK2/STAT3 pathway in astrocytes precedes up-regulation of glial fibrillary acidic protein in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of neurodegeneration: key signaling pathway for astrogliosis in vivo? J Biol Chem. 2004;279(19):19936-19947. [42] CHEN M, GEOFFROY CG, MEVES JM, et al. Leucine Zipper-Bearing Kinase Is a Critical Regulator of Astrocyte Reactivity in the Adult Mammalian CNS. Cell Rep. 2018;22(13):3587-3597. [43] ROTHSTEIN JD, DYKES-HOBERG M, PARDO CA, et al. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron. 1996;16(3):675-686. [44] BRADBURY EJ, MOON LD, POPAT RJ, et al. Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature. 2002;416(6881):636-640. [45] NUTMA E, VAN GENT D, AMOR S, et al. Astrocyte and Oligodendrocyte Cross-Talk in the Central Nervous System. Cells. 2020;9(3):600. [46] BURNSIDE ER, DE WINTER F, DIDANGELOS A, et al. Immune-evasive gene switch enables regulated delivery of chondroitinase after spinal cord injury. Brain. 2018;141(8):2362-2381. [47] SUZUKI H, AHUJA CS, SALEWSKI RP, et al. Neural stem cell mediated recovery is enhanced by Chondroitinase ABC pretreatment in chronic cervical spinal cord injury. PLoS One. 2017;12(8):e0182339. [48] GRIMPE B, SILVER J. A novel DNA enzyme reduces glycosaminoglycan chains in the glial scar and allows microtransplanted dorsal root ganglia axons to regenerate beyond lesions in the spinal cord. J Neurosci. 2004;24(6): 1393-1397. [49] HART CG, DYCK SM, KATARIA H, et al. Acute upregulation of bone morphogenetic protein-4 regulates endogenous cell response and promotes cell death in spinal cord injury. Exp Neurol. 2020;325:113163. [50] BRAMBILLA R, DVORIANTCHIKOVA G, BARAKAT D, et al. Transgenic inhibition of astroglial NF-κB protects from optic nerve damage and retinal ganglion cell loss in experimental optic neuritis. J Neuroinflammation. 2012;9:213. [51] VISMARA I, PAPA S, VENERUSO V, et al. Selective Modulation of A1 Astrocytes by Drug-Loaded Nano-Structured Gel in Spinal Cord Injury. ACS Nano. 2020;14(1):360-371. [52] LIU W, WANG Y, GONG F, et al. Exosomes Derived from Bone Mesenchymal Stem Cells Repair Traumatic Spinal Cord Injury by Suppressing the Activation of A1 Neurotoxic Reactive Astrocytes. J Neurotrauma. 2019;36(3):469-484. [53] 周燕,王琳,裴双,等.骨髓间充质干细胞外泌体可减少脊髓损伤后A1型星形胶质细胞的活化[J].中国组织工程研究,2019,23(21):3294-3301. [54] LIU R, WANG W, WANG S, et al. microRNA-21 regulates astrocytic reaction post-acute phase of spinal cord injury through modulating TGF-β signaling. Aging (Albany NY). 2018;10(6):1474-1488. [55] SU Y, CHEN Z, DU H, et al. Silencing miR-21 induces polarization of astrocytes to the A2 phenotype and improves the formation of synapses by targeting glypican 6 via the signal transducer and activator of transcription-3 pathway after acute ischemic spinal cord injury. FASEB J. 2019;33(10):10859-10871. [56] NGUYEN LH, ONG W, WANG K, et al. Effects of miR-219/miR-338 on microglia and astrocyte behaviors and astrocyte-oligodendrocyte precursor cell interactions. Neural Regen Res. 2020;15(4):739-747. [57] ZHENG Y, MAO YR, YUAN TF, et al. Multimodal treatment for spinal cord injury: a sword of neuroregeneration upon neuromodulation. Neural Regen Res. 2020;15(8):1437-1450. [58] LI Z, YU Y, KANG J, et al. MicroRNA-124 Overexpression in Schwann Cells Promotes Schwann Cell-Astrocyte Integration and Inhibits Glial Scar Formation Ability. Front Cell Neurosci. 2020;14:144. [59] KIM BJ, CHOI JY, CHOI H, et al. Astrocyte-Encapsulated Hydrogel Microfibers Enhance Neuronal Circuit Generation. Adv Healthc Mater. 2020;9(5): e1901072. [60] YANG H, LIU C, FAN H, et al. Sonic Hedgehog Effectively Improves Oct4-Mediated Reprogramming of Astrocytes into Neural Stem Cells. Mol Ther. 2019;27(8):1467-1482. [61] ZAREI-KHEIRABADI M, HESARAKI M, KIANI S, et al. In vivo conversion of rat astrocytes into neuronal cells through neural stem cells in injured spinal cord with a single zinc-finger transcription factor. Stem Cell Res Ther. 2019; 10(1):380. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [5] | Kan Houming, Fan Lijun, Chen Xuetai, Shen Wen. Application of platelet-rich plasma in neuropathic pain [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1286-1292. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [8] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [9] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [10] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [11] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [12] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [13] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [14] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [15] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||