Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (8): 1278-1285.doi: 10.12307/2022.236
Previous Articles Next Articles
Inflammatory responses in post-stroke depression
Tang Wenjing1, Wu Siyuan1, Yang Chen1, Tao Xi1, 2
- 1Department of Neurological Rehabilitation, the First Affiliated Hospital of Hunan Normal University, Changsha 410016, Hunan Province, China; 2Hunan Provincial Key Laboratory of Neurorestoratology, Hunan Normal University, Changsha 410003, Hunan Province, China
-
Received:2021-05-26Revised:2021-05-27Accepted:2021-07-10Online:2022-03-18Published:2021-11-02 -
Contact:Tao Xi, MD, Associate chief physician, Master’s supervisor, Department of Neurological Rehabilitation, the First Affiliated Hospital of Hunan Normal University, Changsha 410016, Hunan Province, China; Hunan Provincial Key Laboratory of Neurorestoratology, Hunan Normal University, Changsha 410003, Hunan Province, China -
About author:Tang Wenjing, Master candidate, Department of Neurological Rehabilitation, the First Affiliated Hospital of Hunan Normal University, Changsha 410016, Hunan Province, China -
Supported by:the Natural Science Foundation of Hunan Province, No. 2018JJ6104 (to TX); Scientific Research Project of Hunan Education Department, No. 20C1172 (to TX); Scientific Research Project of Hunan Health Committee, No. 20200176 (to TX)
CLC Number:
Cite this article
Tang Wenjing, Wu Siyuan, Yang Chen, Tao Xi. Inflammatory responses in post-stroke depression[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1278-1285.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
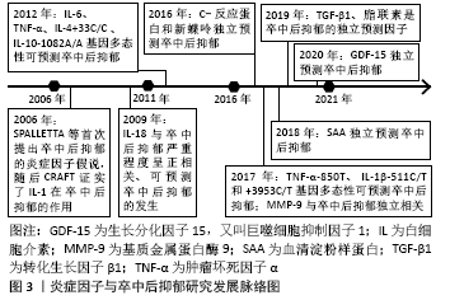
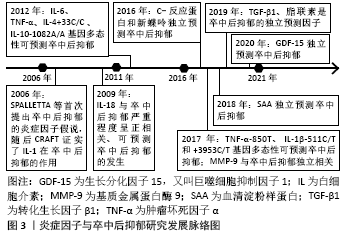
2.1 促炎性细胞因子 2.1.1 白细胞介素1β 白细胞介素1是趋化因子家族中的常见因子,有白细胞介素1α和白细胞介素1β两种亚型。在缺血性脑卒中病程中,后者比前者发挥更重要的作用,如促炎性反应及神经退变。在慢性应激诱导的抑郁大鼠模型,海马区白细胞介素1β表达较健康对照组明显升高[9-10]。而脑室注射白细胞介素1受体拮抗剂(白细胞介素1ra)可以恢复缺血性脑卒中小鼠的蔗糖偏嗜消耗,见表3[11]。该研究从侧面验证了阻断白细胞介素1受体有益于抑郁症状缓解。众所周知,小胶质细胞是中枢神经系统的重要常驻免疫细胞,在介导炎症反应中起枢纽作用。研究发现,小胶质细胞所分泌的白细胞介素1β可被激活的NLRP3炎症小体所诱导,从而促进抑郁炎症反应进程[12]。上述研究提示白细胞介素1β作为促炎因子在小胶质细胞介导的炎症反应中扮演重要角色,其表达增加促进抑郁样行为表现。 临床研究也发现,卒中后抑郁患者不仅血清白细胞介素1β较非卒中后抑郁患者升高,且白细胞介素1β-511C/T和+ 3953C/T基因多态性可增加脑卒中后2周抑郁发生率[4],见表1,提示白细胞介素1β基因位点的遗传变异可能与人类卒中后抑郁关联。但是,该作者5年前的研究不支持此观点[13],这可能与纳入样本量及抑郁严重程度不同有关。因此,早期基因筛查或卒中后白细胞介素1β靶向干预,或可有助于卒中后抑郁早期诊断和治疗。 2.1.2 白细胞介素6 在中枢,白细胞介素6主要来源于神经元、星形胶质细胞、小胶质细胞和内皮细胞。相较于其他细胞因子,白细胞介素6在炎症反应中的促炎作用最为明确,临床上常被用于评价炎症反应的严重程度。有一项基于48例急性缺血性脑卒中患者血清的分析发现,卒中后抑郁患者白细胞介素6水平较非卒中后抑郁患者明显升高[14];而另一项纳入286例缺血性脑卒中患者的纵向研究发现,急性期升高的血清白细胞介素6与卒中后2周及1年的抑郁发生独立相关[6],见表2。这两项研究分别从不同角度证实了白细胞介素6与卒中后抑郁之间存在关联。下丘脑-垂体-肾上腺轴在调节人体内分泌过程中发挥重要作用,血浆皮质醇代谢异常被认为与卒中后抑郁相关。相较于非卒中后抑郁和正常对照组,卒中后抑郁组患者8:00血清白细胞介素6水平明显升高,而皮质醇升高主要发生在16:00 [15]。李庆丽等[16]研究发现卒中后抑郁患者空腹血清白细胞介素6水平可被中药解郁通络方与帕罗西汀联合治疗所缓解,同时,皮质醇等激素也呈降低趋势,见表3。抗抑郁治疗从侧面验证了白细胞介素6作为卒中后抑郁疗效预判中的价值。然而,白细胞介素6与皮质醇的升高在时间上并不一致,两者在介导卒中后抑郁患病机制中是否存在内在联系目前尚无报道。 白细胞介素6介导卒中后抑郁的炎症机制在基础研究中进一步被证实。慢性应激可促使脑缺血大鼠海马及血清白细胞介素6表达上调[17],见表3。切除卵巢的雌性抑郁大鼠海马组织白细胞介素6、干扰素γ及吲哚胺2,3-双加氧酶(indoleamine 2,3-dioxygenase,IDO)表达增加,而5-羟色胺表达减少[18]。色氨酸是5-羟色胺的前体分子。IDO作为色氨酸降解酶,促进色氨酸降解,减少5-羟色胺合成。另有研究发现[19],增加的白细胞介素6和干扰素γ可以上调IDO表达,进一步减少5-羟色胺合成,从而介导抑郁及卒中后抑郁的发生。有意思的是,白细胞介素6除了促炎作用外,其自身还能抑制其他促炎性细胞因子(如肿瘤坏死因子)的产生,间接刺激抗炎性细胞因子(如白细胞介素1ra和白细胞介素10)的表达增加[20]。在炎症反应中,如何定位并阻断白细胞介素6在脑组织细胞依赖性表达,从而影响卒中后抑郁发展方向是亟待解决的难题。 2.1.3 白细胞介素8 白细胞介素8主要来源于中性粒细胞以及活化的小胶质细胞、星形胶质细胞和内皮细胞,是中枢神经系统发育和功能调节的重要因子。与正常人比较,抑郁症患者脑脊液白细胞介素8水平明显增高[21]。而一项针对重度抑郁症死亡患者脑组织病理的研究发现,与奖赏行为有关的脑区(BA10)不仅白细胞介素8表达增加,而且抗凋亡因子Y-box结合蛋白1(YBX1)、caspase-1抑制蛋白pseudo- ICE(COP1)和FKGS2也过度表达[22]。这些研究提示白细胞介素8可能通过介导神经元变性和凋亡促进抑郁的发生。因此,有人提议把白细胞介素8作为抗抑郁治疗的潜在靶点[34]。等位基因变异性筛查对抑郁遗传易感性分析具有重要价值。研究发现,抑郁症患者多存在白细胞介素8-251T/A等位基因的遗传变异[23];而另一项纳入276例脑卒中患者的研究则发现,该等位基因变异在卒中后抑郁患者中也广泛存在,但与卒中后抑郁严重程度无显著关联[13]。增加样本量或与血清白细胞介素8浓度进行交互作用分析或可有助于分析该因子在卒中后抑郁患病中的价值。进一步研究发现,急性缺血性脑卒中患者血清白细胞介素8水平普遍升高,且与病情严重程度呈正相关[24]。与白细胞介素6相同,血清白细胞介素8也被用于抗卒中后抑郁治疗疗效评价[25],见表3。虽然神经功能缺损程度是卒中后抑郁的一项重要危险因子,但是脑卒中急性期患者血清或者脑脊液白细胞介素8对继发性抑郁是否具有预测价值,还缺少相关临床数据。 2.1.4 白细胞介素18 白细胞介素18 是属于白细胞介素1家族的促炎因子,在神经炎症和退行性病变中发挥重要作用。缺血性脑卒中患者血清白细胞介素18水平不仅与卒中的严重程度呈正相关,还与卒中后2周、甚至1年后的抑郁发生相关,可作为远期卒中后抑郁发生的有效预测因子[6],见表2。杏仁核是抑郁环路的重要组成部分。在卒中后抑郁小鼠杏仁核注射重组白细胞介素18会加重其抑郁行为;相反,注射特异性内源性抑制剂(白细胞介素18结合蛋白)可缓解抑郁样行为[26]。钠-钾-氯共转运体NKCC1可使细胞内氯离子超载,并与γ-氨基丁酸能神经元关联,后者在介导抑制性神经递质传导及抑郁样行为中起重要作用[27]。慢性应激不仅可以激活大鼠海马区白细胞介素18R/NKCC1信号通路[27],还可以上调大脑皮质IDO[28],从而增加γ-氨基丁酸能神经元活性并降低5-羟色胺利用效率,最终增加代谢产物的抑制效应。而在白细胞介素18敲除小鼠模型中,这种应激效应则不明显[26]。这些研究进一步支持白细胞介素18及其受体介导慢性应激在卒中后抑郁模型的可能作用机制。 2.1.5 肿瘤坏死因子α 肿瘤坏死因子家族包括肿瘤坏死因子α和肿瘤坏死因子β。在内外因素刺激下,中枢神经元、星形胶质细胞及小胶质细胞均可分泌肿瘤坏死因子α,参与细胞信号传导、转录和促炎及凋亡等作用。MU等[29]纳入了120例受试者,发现卒中后抑郁患者血清肿瘤坏死因子α显著高于非卒中后抑郁患者,且与抑郁程度呈正相关。提示肿瘤坏死因子α在卒中后抑郁诊断方面具有应用前景。慢性应激可促使脑缺血大鼠海马和血清肿瘤坏死因子α表达增 加[17]。在慢性应激和脂多糖诱导小鼠制作抑郁样模型中,ZHAO等[30]发现两组小鼠大脑前额叶皮质和海马肿瘤坏死因子α及IDO均明显增高。过表达肿瘤坏死因子α可通过激活神经元突触前和突触后膜上肿瘤坏死因子受体,调节突触信号传导和可塑性,最终改变神经元功能[31]。反之,腹腔注射肿瘤坏死因子α抑制剂英夫利昔单抗或IDO抑制剂(1-MT)可缓解小鼠抑郁样行为,且前者具有小胶质细胞依赖性抑制IDO表达[28]。以上研究提示肿瘤坏死因子α与IDO在抑郁病理生理学机制中可能具有协同作用,增加的肿瘤坏死因子α可能从神经突触受体、可塑性及递质降解等途径介导慢性应激诱导抑郁样行为,是卒中后抑郁干预的重要潜在靶点。 此外,研究发现肿瘤坏死因子α?850C/T的基因多态性与脑卒中后2周抑郁发生独立相关[4],见表2。肿瘤坏死因子受体1(TNFR1)由TNFRSF1A和TNFRSF1B基因编码,在抑郁症患者血清中,上述基因的表达较正常人明显增高[32]。WANG等[33]发现,卒中后抑郁大鼠小脑组织肿瘤坏死因子α蛋白和TNFRSF1A mRNA表达在小脑顶核电刺激后下降,且抑郁程度好转,见表3。肿瘤坏死因子α不仅在治疗后的啮齿类抑郁模型海马和皮质区表达下降[10],在抑郁症治愈患者血清中也呈下降趋势[34]。所以,检测肿瘤坏死因子α基因多态性或血清浓度有助于早期卒中后抑郁诊断及疗效判断。 2.1.6 干扰素γ 干扰素γ是Ⅱ型干扰素家族中的唯一成员,在神经系统炎症反应中由CD4+ T辅助细胞1分泌,介导星形胶质细胞和小胶质细胞在中枢发挥免疫活性。研究发现[35-36],移植粪便微生物群或灌胃和厚朴酚可降低海马干扰素γ水平,并缓解小鼠抑郁样行为。相反,给小鼠侧脑室注射干扰素γ可诱发抑郁样行为,机制可能与促进海马和前额叶皮质小胶质细胞激活及延缓海马颗粒细胞层神经元成熟有关[37]。CAMARGOS等[38]研究发现,在小鼠全脑缺血模型,米诺环素缓解抑郁样行为与大脑干扰素γ表达降低有关,见表3。干扰素γ是IDO的强力激活剂。在C57BL/6小鼠腹腔连续注射干扰素γ,可增强其脑组织IDO和喹啉酸活性[39]。激活的IDO减少5-羟色胺合成,并降低其利用效率。不过,该研究缺乏对小鼠行为学的描述。在机制层面,关于干扰素γ在动物模型的研究仍局限于普通抑郁表型,尚缺少针对卒中后抑郁动物模型的深入研究。SU等[40]对104例缺血性脑卒中患者追踪随访1年,其中12例被诊断为卒中后抑郁,其血清干扰素γ水平显著高于非卒中后抑郁患者。但是,另一项研究发现卒中后抑郁患者血清干扰素γ水平与抑郁程度无显著关联,而血清干扰素γ/白细胞介素10比值在诊断卒中后中度抑郁中具有更重要价值[41],该研究进一步提示促炎与抗炎失调在卒中后抑郁患病中的可能作用。不过,干扰素γ在卒中后抑郁诊断中的潜在价值需要更多临床数据去证实。 "
2.2 抗炎性细胞因子 2.2.1 白细胞介素4 在中枢,白细胞介素4可由星形胶质细胞及小胶质细胞产生,但外周T细胞分泌的白细胞介素4同样影响学习和记忆功能。相较于一般脑卒中患者,白细胞介素4+33C/C基因多态性改变的患者在卒中后1年表现出更高的卒中后抑郁患病率[13],提示白细胞介素4的遗传变异与卒中后抑郁关联,见表1。PARK等[42]在注射重组白细胞介素1β的大鼠侧脑室内,同时注射不同(高、低)浓度的重组白细胞介素4或生理盐水,结果发现重组白细胞介素4组的抑郁行为不仅明显改善,而且效果呈浓度依赖性差异;此外,两种浓度的重组白细胞介素4均可促使海马和前额叶皮质5-羟色胺表达降低及5-羟色胺转运体表达增加,而色氨酸羟化酶仅在前额叶皮质降低,提示白细胞介素4可能通过抑制白细胞介素1β并调节中枢神经递质来缓解大鼠抑郁样行为。较野生型C57/BL6小鼠,白细胞介素4敲除对小鼠脑缺血后空间学习及记忆障碍的影响更为明显,且伴有M2型小胶质细胞标记的CD206和白细胞介素10表达减少[43]。血管性抑郁与血管性认知损伤共病非常多见,可能具有相似的分子生物学通路。然而,尚无白细胞介素4在卒中后抑郁炎症反应中的研究报道。胶质细胞依赖性分泌的白细胞介素4如何在抑郁或卒中后抑郁病理生理学机制中发挥活性,需要进一步研究。 2.2.2 白细胞介素10 白细胞介素10是目前研究最多的抗炎因子,主要来源于中枢小胶质细胞。一项对脑卒中早期患者的遗传变异分析发现,白细胞介素10-1082 A/A 基因多态性改变与卒中后1年卒中后抑郁患病显著相关[13]。虽然卒中后抑郁患者血清白细胞介素10水平有所增加,但是,其增加比例显著低于促炎性细胞因子。研究发现,白细胞介素6/白细胞介素10和肿瘤坏死因子α/白细胞介素10比值升高对卒中后1年卒中后抑郁诊断具有预测作用[40],见表2;而白细胞介素18/白细胞介素10、干扰素γ/白细胞介素10与白细胞介素1β/白细胞介素10比值增加在中度卒中后抑郁患者中更为明显[41]。这些临床研究提示先天性的白细胞介素10转录活性异常或促炎/抗炎因子比例失调与卒中后抑郁患病密切关联。尽管在卒中后抑郁动物模型中,关于脑组织白细胞介素10表达的研究并不多见,而在双侧颈总动脉闭塞诱发的抑郁样小鼠模型,脑组织白细胞介素10表达明显增高[38]。在脂多糖诱导的抑郁小鼠模型中,白细胞介素10抗体滴鼻后前额叶皮质IDO1 mRNA表达增加,抑郁程度加重;相反,使用重组白细胞介素10滴鼻后,IDO1 mRNA表达降低,抑郁样行为缓解[44]。在BV2小胶质细胞,白细胞介素10也能显著降低干扰素γ诱导的IDO1 mRNA表达[44]。由此推理,小胶质细胞依赖的白细胞介素10/IDO1通路可能介导卒中后抑郁发生发展。 2.2.3 转化生长因子β1 转化生长因子β1是转化生长因子家族的一种亚型,是维持小胶质细胞发育与成熟的关键因子。较假手术组,卒中后抑郁小鼠海马转化生长因子β表达显著降低,而侧脑室注射重组转化生长因子β后,抑郁行为明显被逆转[45],见表3。研究发现,转化生长因子β1介导的小鼠抑郁行为与miR-192-5p及其靶基因Fbln2信号通路有关[46]。而经RNA测序及基因富集分析,进一步证实了海马转化生长因子β1信号通路呈小胶质细胞依赖性参与抑郁患病机制[47]。临床研究也发现抑郁症患者血浆转化生长因子β1水平较正常对照组显著降低,且与抑郁程度成负相关[48]。而重症抑郁患者血浆转化生长因子β1水平在治疗后显著升高,这从侧面验证了其与抑郁关联及在治疗效果评判中的价值[49]。然而,关于转化生长因子β1在卒中后抑郁的研究还非常少见。作为一种新型抗炎性细胞因子,转化生长因子β1能否作为卒中后抑郁诊治标志物及如何介导小胶质细胞参与卒中后抑郁病理生理学机制,需要更多研究。 "

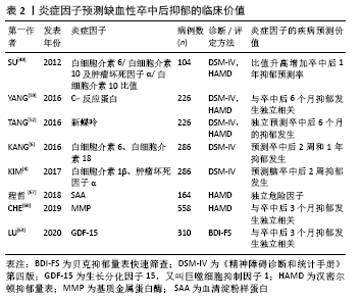
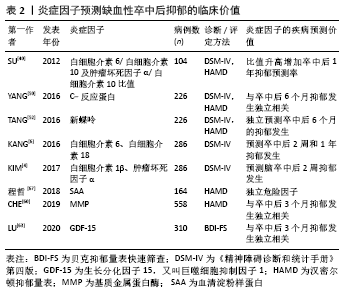
2.3 其他炎症因子 2.3.1 C-反应蛋白 C-反应蛋白是由肝细胞产生的非特异性炎症反应标志物,在脑卒中及卒中后精神障碍患者的血浆或脑脊液中显著升高。一项纳入120例缺血性脑卒中患者的横断面研究发现,卒中后抑郁患者血清C-反应蛋白水平显著高于非卒中后抑郁患者[29]。而另一项研究前瞻性的纳入226例急性期缺血性脑卒中患者,发现早期C-反应蛋白与卒中后6个月的抑郁发生独立相关[50],见表2,所以C-反应蛋白可能是卒中后抑郁早期诊断及判断预后的重要敏感标志物。血氧水平依赖性的功能核磁共振成像对研究抑郁环路机制具有独特的优势。研究者对抑郁患者的全脑功能连接进行分析,发现血浆C-反应蛋白水平与腹侧纹状体及前额叶皮质之间的连接强度呈负相关[51],提示C-反应蛋白可能与皮质纹状体奖赏回路的改变及促进抑郁发生存在联系。但是,这项研究局限于普通抑郁患者。卒中后抑郁患者脑结构存在血管性基础病变,其全脑功能连接或抑郁环路可能有别于普通抑郁患者。奖赏回路机制作为抑郁患病的重要节点是否同样与卒中后抑郁 关联,外周性分泌蛋白C-反应蛋白是如何与中枢环路建立联系?关于C-反应蛋白在抑郁动物模型的研究还非常有限。 2.3.2 新蝶呤 新蝶呤主要由巨噬细胞和小胶质细胞产生,是机体细胞免疫激活的标志,与缺血性脑卒中的复发独立相关。较非卒中后抑郁患者,卒中后抑郁患者入院时血清新蝶呤显著增高,对卒中后6个月的抑郁发生有独立预测价值[52],见表2。在免疫激活状态下,小胶质细胞M1极化在卒中后抑郁患病机制中起重要作用。四氢生物蝶呤不仅是新喋呤前体,同时也是多巴胺、去甲肾上腺素和5-羟色胺等单胺类神经递质合成的重要辅助因子。慢性炎症时,巨噬细胞来源的干扰素γ促使活性氧输出增加,四氢生物蝶呤的氧化损失会减少单胺类神经递质的生物合成[53];而氧化应激生成的新蝶呤可激活小胶质细胞,导致中枢免疫稳态失衡,促进抑郁发生[53]。然而,关于新蝶呤在卒中后抑郁的基础研究仍几乎空白。 2.3.3 脂联素 脂联素是脂肪细胞分泌的炎症因子,具有抗炎、抗氧化及胰岛素拮抗作用,对心脑血管疾病具有重要的调节作用。较非卒中后抑郁患者,卒中后抑郁患者血清脂联素水平显著降低,且与抑郁程度呈负相关,低水平脂联素是卒中后抑郁独立危险因子[54],见表1。选择性敲除5-羟色胺神经元脂联素特异性受体1可诱发小鼠抑郁样行为,同时下调海马和内侧前额叶皮质色氨酸羟化酶2和5-羟色胺表达[55]。而侧脑室注射重组脂联素可抑制海马成纤维细胞生长因子受体3表达,减轻小鼠抑郁样行为[56]。这些研究均提示脂联素对抑郁模型具有保护作用。但是,尚不清楚脂联素如何介导卒中后抑郁模型的患病机制,补充脂联素是否对卒中后抑郁患者有益,未来仍需要进行更多研究。 2.3.4 NLRP3炎症小体 NLRP3炎症小体由核苷酸结合寡聚化结构域样受体3、凋亡相关斑点样蛋白及半胱氨酸天冬氨酸酶1组合而成,是调节先天免疫和炎症反应的特殊信号分子。研究发现,慢性应激不仅增加正常大鼠海马区NLRP3炎症小体表达[9],而且对缺血性卒中大鼠海马区炎症反应及细胞凋亡同样有激活作用[57],见表3。作为炎症反应节点的重要标志物,NLRP3炎症小体可促进活化的小胶质细胞分泌白细胞介素1β和白细胞介素18[58],两者通过免疫调节及促炎反应参与卒中后抑郁发生[11,26]。ATP依赖的离子通道型嘌呤样受体P2X7R是NLRP3炎症小体的重要上游分子,在抑郁症的患病机制中起重要作用。脑室内注射P2X7R抑制剂可以缓解卒中后抑郁大鼠抑郁样行为[59]。P2X7R/NLRP3炎症小体信号通路能否具有小胶质细胞依赖性地调节卒中后抑郁样行为,值得进一步探索。 2.3.5 基质金属蛋白酶9(matrix metalloproteinase 9,MMP-9) MMP-9是细胞外基质降解的关键决定因子,参与炎症反应及神经元可塑性,在脑损伤及抑郁发展中起作用。研究发现,缺血性脑卒中急性期血清MMP-9升高与3个月后抑郁发生独立相关[60],见表2。而联合MMP-9、生长分化因子15(growth differentiation factor-15,GDF-15)、抗心磷脂抗体及抗磷脂酰丝氨酸抗体可从不同病理途径增加卒中后抑郁预测的增量效应[61]。然而,作为5-羟色胺和去甲肾上腺素再摄取抑制剂,文拉法辛改善小鼠情绪及记忆功能与促进海马MMP-9表达增加有关[62]。MMP-9在临床和基础研究中呈现截然相反的表达趋势,相关机制不明。 2.3.6 GDF-15 又被称为巨噬细胞抑制因子1,属于转化生长因子β超家族,作为一种应激调节因子,其受体GFRAL表达于脑干孤束核及后脑区,对食物摄入及情绪反应具有调节作用。研究人员对310例急性缺血性脑卒中患者进行追踪随访,发现入院时血清GDF-15水平与3个月后抑郁发生明显相关,且卒中后抑郁患者较非卒中后抑郁者高出近1倍[63],见表2。联合血清GDF-15及其他标志物对卒中后抑郁预测更具有增量效应[61]。相较于临床研究,GDF15在脑血管病和抑郁模型的基础研究中也有报道。急性缺血不仅可引起大鼠脑组织GDF-15表达增加,而且与神经功能损伤程度呈正相关[64]。在小鼠侧脑室注射GDF15可诱发厌食行为和体质量下降,同时观察到孤束核和后脑区域Fos表达增加,神经损害加重;而单独切除后脑或后脑和孤束核,则消除GDF15对食物摄入和体质量的负面影响[65]。提示脑组织缺血诱导的GDF-15表达增加可能与厌食行为及体质量下降的抑郁行为特征存在关联。最新研究发现,氟西汀可诱导大鼠原代星形胶质细胞GDF-15表达增加,后者具有促进抑郁小鼠血脑屏障周围星形胶质重构及紧密连接蛋白claudin-5表达增多的作用[66]。然而,关于GDF15在脑组织表达或下游信号通路的研究仍局限于普通抑郁模型。尚无GDF15在卒中后抑郁模型的相关报道。 2.3.7 血清淀粉样蛋白A 是一种高密度载脂蛋白。慢性炎症反应时,其血浆水平可适度升高,而急性炎症期可上升至正常水平的1 000倍,在炎症控制和传播中起关键作用。睡眠减少是卒中后抑郁早期临床表现之一,严重的睡眠障碍可激活中枢免疫失调及炎症反应。程哲等[67]纳入164例首发急性缺血性脑卒中患者,发现卒中后抑郁患者血清淀粉样蛋白A水平较非卒中后抑郁患者显著升高,认为血清淀粉样蛋白A可作为卒中后抑郁预测的独立危险因子,见表2。同样,睡眠剥夺的负面效应在健康人群也得到验证。较正常对照组,连续2晚全部睡眠剥夺的志愿者清晨血清淀粉样蛋白A水平明显升高,而睡眠恢复正常后血清淀粉样蛋白A也恢复至正常水平;有意思的是,这种血清淀粉样蛋白A变化在连续4晚快相睡眠剥夺组则不明显[68]。提示睡眠障碍不仅可能诱发机体慢性炎症反应,而且这种炎症反应与慢波睡眠时相关联,但是这种关联的生物机制尚不明确。 不仅如此,睡眠剥夺的生物学效应在基础研究中也被证实。对C57BL/6小鼠进行15 d睡眠限制(每天仅睡3 h)或连续睡眠剥夺72 h后,血清淀粉样蛋白A浓度、肝脏Saa1.1/Saa2.1 mRNA和脂肪组织Saa3 mRNA水平均显著上 调[68]。作为初始生化指标改变的一部分,血清淀粉样蛋白A在长期睡眠障碍模型或人群中持续影响脂蛋白转运效率,有可能导致胰岛素抵抗和体质量增加等有害事件。血清淀粉样蛋白A在小胶质细胞NLRP3炎症小体激活过程中起重要作用,敲除血清淀粉样蛋白A可减轻脑缺血小鼠模型的炎症反应及脑梗死体积[69],提示干预血清淀粉样蛋白A有希望成为脑缺血保护和减轻炎症反应的靶点。然而,尚无血清淀粉样蛋白A在卒中后抑郁模型的相关报道。 "

| [1] 陶希,王佳,刘楚娟,等.缺血性卒中后抑郁动物模型构建及核心症状评定研究进展[J].中国康复医学杂志,2020,10(35):1264-1269. [2] MITCHELL AJ, SHETH B, GILL J, et al. Prevalence and predictors of post-stroke mood disorders: A meta-analysis and meta-regression of depression, anxiety and adjustment disorder. Gen Hosp Psychiatry. 2017;47:48-60. [3] 赵兵兵,李涛,王豆,等.缺血再灌注联合慢性不可预知刺激构建卒中后抑郁新模型[J].中国组织工程研究,2019,23(11):1755-1760. [4] KIM JM, KANG HJ, KIM JW, et al. Associations of tumor necrosis factor-α and interleukin-1β levels and polymorphisms with post-stroke depression. Am J Geriatr Psychiatry. 2017;25(12):1300-1308. [5] 陶希,刘佳,邓景贵,等.低频重复经颅磁刺激对卒中后抑郁的远期疗效及血5-羟色胺表达的影响[J].中华物理医学与康复杂志,2014,36(6): 416-420. [6] KANG HJ, BAE KY, KIM SW, et al. Effects of interleukin-6, interleukin-18, and statin use, evaluated at acute stroke, on post-stroke depression during 1-year follow-up. Psychoneuroendocrinology. 2016;72:156-160. [7] WANG C, WANG M, XIA K, et al. A bioactive injectable self-healing anti-inflammatory hydrogel with ultralong extracellular vesicles release synergistically enhances motor functional recovery of spinal cord injury. Bioact Mater. 2021;6(8):2523-2534. [8] MORIYAMA Y. TAKAGI N, Tanonaka K. Intravenous injection of neural progenitor cells improved depression-like behavior after cerebral ischemia. Transl Psychiatry. 2011;1(8):e29. [9] YUE N, LI B, YANG L, et al. Electro-acupuncture alleviates chronic unpredictable stress-induced depressive- and anxiety-like behavior and hippocampal neuroinflammation in rat model of depression. Front Mol Neurosci. 2018;11:149. [10] JIA M, LI C, ZHENG Y, et al. Leonurine exerts antidepressant-like effects in the chronic mild stress-induced depression model in mice by inhibiting neuroinflammation. Int J Neuropsychopharmacol. 2017;20(11):886-895. [11] CRAFT TK, DEVRIES AC. Role of IL-1 in poststroke depressive-like behavior in mice. Biol Psychiatry. 2006;60(8):812-818. [12] Zhang Y, Liu L, Liu Y, et al. NLRP3 inflammasome mediates chronic mild stress-induced depression in mice via neuroinflammation. Int J Neuropsychopharmacol. 2015;18(8):pyv006. [13] KIM JM, STEWART R, KIM SW, et al. Associations of cytokine gene polymorphisms with post-stroke depression. World J Biol Psychiatry. 2012;13(8):579-587. [14] SPALLETTA G, CRAVELLO L, IMPERIALE F, et al. Neuropsychiatric symptoms and interleukin-6 serum levels in acute stroke. J Neuropsychiatry Clin Neurosci. 2013;25(4):255-263. [15] ZHANG XF, ZOU W, YANG Y. Effects of IL-6 and cortisol fluctuations in post-stroke depression. J Huazhong Univ Sci Technolog Med Sci. 2016;36(5):732-735. [16] 李庆丽,王贵贤,王艳捷,等.解郁通络方对脑卒中后抑郁神经免疫炎症指标的影响[J].中国实验方剂学杂志,2018,24(6):204-209. [17] 王建楠,乔嘉璐,张磊,等.芬戈莫德对卒中后抑郁大鼠的治疗作用[J].神经解剖学杂志,2020,36(4):376-382. [18] XU Y, SHENG H, TANG Z, et al. Inflammation and increased IDO in hippocampus contribute to depression-like behavior induced by estrogen deficiency. Behav Brain Res. 2015;288:71-78. [19] NAGY EE, FRIGY A, SZÁSZ JA, et al. Neuroinflammation and microglia/macrophage phenotype modulate the molecular background of post-stroke depression: a literature review. Exp Ther Med. 2020;20(3):2510-2523. [20] BENATTI FB, PEDERSEN BK. Exercise as an anti-inflammatory therapy for rheumatic diseases-myokine regulation. Nat Rev Rheumatol. 2015;11(2): 86-97. [21] KUZIOR H, FIEBICH BL, YOUSIF NM, et al. Increased IL-8 concentrations in the cerebrospinal fluid of patients with unipolar depression. Compr Psychiatry. 2020;102:152196. [22] SHELTON RC, CLAIBORNE J, Sidoryk-Wegrzynowicz M, et al. Altered expression of genes involved in inflammation and apoptosis in frontal cortex in major depression. Mol Psychiatry. 2011;16(7):751-762. [23] JANELIDZE S, SUCHANKOVA P, EKMAN A, et al. Low IL-8 is associated with anxiety in suicidal patients:genetic variation and decreased protein levels. Acta Psychiatr Scand. 2015;131(4):269-278. [24] SHAHEEN HA, DAKER LI, ABBASS MM, et al. The relationship between the severity of disability and serum IL-8 in acute ischemic stroke patients. Egypt J Neurol Psychiatr Neurosurg. 2018;54(1):26. [25] 梁婉桂,汤瑞珠,傅晓芸,等.行气解郁方对脑卒中后抑郁患者神经功能及炎症因子水平的影响[J].辽宁中医杂志,2020,47(11):130-133. [26] WU D, ZHANG G, ZHAO C, et al. Interleukin-18 from neurons and microglia mediates depressive behaviors in mice with post-stroke depression. Brain Behavi Immun. 2020;88:411-420. [27] TSUKAHARA T, MASUHARA M, IWAI H, et al. Repeated stress-induced expression pattern alterations of the hippocampal chloride transporters KCC2 and NKCC1 associated with behavioral abnormalities in female mice. Biochem Biophys Res Commun. 2015;465(1):145-151. [28] LIU YN, PENG YL, LIU L, et al. TNFα mediates stress-induced depression by upregulating indoleamine 2,3-dioxygenase in a mouse model of unpredictable chronic mild stress. Eur Cytokine Netw. 2015;26(1):15-25. [29] MU Y, WANG Z, ZHOU J, et al. Correlations of post-stroke depression with inflammatory response factors. Iran J Public Health. 2018;47(7): 988-993. [30] ZHAO X, CAO F, LIU Q, et al. Behavioral, inflammatory and neurochemical disturbances in LPS and UCMS-induced mouse models of depression. Behav Brain Res. 2019;364:494-502. [31] O’CONNOR JJ. Targeting tumour necrosis factor-α in hypoxia and synaptic signalling. Ir J Med Sci. 2013;182(2):157-162. [32] BOBIŃSKA K, GAŁECKA E, SZEMRAJ J, et al. Is there a link between TNF gene expression and cognitive deficits in depression? Acta Biochim Pol. 2017;64(1):65-73. [33] WANG M, GUO J, DONG LN, et al. Cerebellar fastigial nucleus stimulation in a chronic unpredictable mild stress rat model reduces post-stroke depression by suppressing brain inflammation via the microRNA-29c/TNFRSF1A signaling pathway. Med Sci Monit. 2019;25:5594-5605. [34] LIU JJ, WEI YB, STRAWBRIDGE R, et al. Peripheral cytokine levels and response to antidepressant treatment in depression:a systematic review and meta-analysis. Mol Psychiatry. 2019;25(2):339-350. [35] LI N, WANG Q, WANG Y, et al. Fecal microbiota transplantation from chronic unpredictable mild stress mice donors affects anxiety-like and depression-like behavior in recipient mice via the gut microbiota-inflammation-brain axis. Stress. 2019;22(5):592-602. [36] ZHANG B, WANG PP, HU KL, et al. Antidepressant-like effect and mechanism of action of honokiol on the mouse lipopolysaccharide (LPS) depression model. Molecules. 2019;24(11):2035. [37] ZHANG J, HE H, QIAO Y, et al. Priming of microglia with IFN-γ impairs adult hippocampal neurogenesis and leads to depression-like behaviors and cognitive defects. Glia. 2020;68(12):2674-2692. [38] CAMARGOS QM, SILVA BC, SILVA DG, et al. Minocycline treatment prevents depression and anxiety-like behaviors and promotes neuroprotection after experimental ischemic stroke. Brain Res Bull. 2020;155:1-10. [39] SAITO K, MARKEY SP, HEYES MP. Chronic effects of gamma-interferon on quinolinic acid and indoleamine-2,3-dioxygenase in brain of C57BL6 mice. Brain Res. 1991;546(1):151-154. [40] SU JA, CHOU SY, TSAI CS, et al. Cytokine changes in the pathophysiology of poststroke depression. Gen Hosp Psychiatry. 2012;34(1):35-39. [41] BENSIMON K, HERRMANN N, SWARDFAGER W, et al. Kynurenine and depressive symptoms in a poststroke population. Neuropsychiatr Dis Treat. 2014;10:1827-1835. [42] PARK HJ, SHIM HS, AN K, et al. IL-4 inhibits il-1β-induced depressive-like behavior and central neurotransmitter alterations. Mediators Inflamm. 2015;2015:941413. [43] LIU X, LIU J, ZHAO S, et al. Interleukin-4 is essential for microglia/macrophage m2 polarization and long-term recovery after cerebral ischemia. Stroke. 2016;47(2):498-504. [44] LAUMET G, EDRALIN JD, CHIANG AC, et al. Resolution of inflammation-induced depression requires T lymphocytes and endogenous brain interleukin-10 signaling. Neuropsychopharmacology. 2018;43(13): 2597-2605. [45] ZHANG Y, YU P, LIU H, et al. Hyperforin improves post-stroke social isolation‑induced exaggeration of PSD and PSA via TGF-β. Int J Mol Med. 2019;43(1):413-425. [46] TANG CZ, YANG JT, LIU QH, et al. Up-regulated miR-192-5p expression rescues cognitive impairment and restores neural function in mice with depression via the Fbln2-mediated TGF-β1 signaling pathway. FASEB J. 2019;33(1):606-618. [47] ZHANG K, YANG C, CHANG L, et al. Essential role of microglial transforming growth factor-β1 in antidepressant actions of (R)-ketamine and the novel antidepressant TGF-β1. Transl Psychiatry. 2020;10(1):32. [48] 邱文静,杨欢,肖波,等.抑郁症中糖皮质激素受体与CD4+CD25+调节性T细胞的关系及其所致免疫失衡机制的初步探讨[J].中国神经精神疾病杂志,2009,35(6):350-354. [49] AL-HAKEIM HK, AL-KUFI SN, AL-DUJAILI AH, et al. Serum interleukin levels and insulin resistance in major depressive disorder. CNS Neurol Disord Drug Targets. 2018;17(8):618-625. [50] YANG RR, LU BC, LI T, et al. The relationship between high-sensitivity C-reactive protein at admission and post stroke depression:a 6-month follow-up study. Int J Geriatr Psychiatry. 2016;31(3):231-239. [51] FELGER JC, LI Z, HAROON E, et al. Inflammation is associated with decreased functional connectivity within corticostriatal reward circuitry in depression. Mol Psychiatry. 2016;21(10):1358-1365. [52] TANG CZ, ZHANG YL, WANG WS, et al. Elevated serum levels of neopterin at admission predicts depression after acute ischemic stroke: a 6-month follow-up study. Mol Neurobiol. 2016;53(5):3194-3204. [53] KALKMAN HO, FEUERBACH D. Antidepressant therapies inhibit inflammation and microglial M1-polarization. Pharmacol Ther. 2016;163:82-93. [54] 陈钊, 侯乐,李丹,等.血清脂联素预测缺血性卒中患者的卒中后抑郁[J].国际脑血管病杂志,2018,26(8):577-582. [55] LI C, MENG F, GARZA JC, et al. Modulation of depression-related behaviors by adiponectin AdipoR1 receptors in 5-HT neurons. Mol Psychiatry. 2020. doi: 10.1038/s41380-020-0649-0. [56] WANG XQ, LI WH, TANG YH, et al. The correlation between adiponectin and FGF9 in depression disorder. Brain Res. 2020;1729:146596. [57] 李丛琴,王强,罗路,等.运动训练对卒中后抑郁大鼠PTEN诱导的炎症通路及海马神经元凋亡的影响[J].中华物理医学与康复杂志,2020, 42(7):577-582. [58] HANAMSAGAR R, TORRES V, KIELIAN T. Inflammasome activation and IL-1β/IL-18 processing are influenced by distinct pathways in microglia. J Neurochem. 2011;119(4):736-748. [59] WANG Z, REN W, ZHAO F, et al. Curcumin amends Ca(2+) dysregulation in microglia by suppressing the activation of P2X7 receptor. Mol Cell Biochem. 2020;465(1-2):65-73. [60] CHE B, ZHONG C, GE J, et al. Serum Matrix Metalloproteinase-9 Is Associated With Depression After Acute Ischemic Stroke. Circ J. 2019;83(11):2303-2311. [61] CHE B, ZHU Z, BU X, et al. Multiple biomarkers covering several pathways for the prediction of depression after ischemic stroke. J Affect Disord. 2021; 280(Pt A):442-449. [62] ALAIYED S, BOZZELLI PL, CACCAVANO A, et al. Venlafaxine stimulates PNN proteolysis and MMP-9-dependent enhancement of gamma power;relevance to antidepressant efficacy. J Neurochem. 2019;148(6): 810-821. [63] LU X, DUAN J, CHENG Q, et al. The association between serum growth differentiation factor-15 and 3-month depression after acute ischemic stroke. J Affect Disord. 2020;260:695-702. [64] 焦光美,单海雷,康玲伶,等.脑梗死大鼠脑组织生长分化因子-15的表达及其与神经功能损害的关系[J].中国免疫学杂志,2020,36(2):154-158. [65] TSAI VW, MANANDHAR R, JØRGENSEN SB, et al. The anorectic actions of the TGFβ cytokine MIC-1/GDF15 require an intact brainstem area postrema and nucleus of the solitary tract. PLoS One. 2014;9(6): e100370. [66] MALIK VA, ZAJICEK F, MITTMANN LA, et al. GDF15 promotes simultaneous astrocyte remodeling and tight junction strengthening at the blood-brain barrier. J Neurosci Res. 2020;98(7):1433-1456. [67] 程哲,郭世龙,孟焱,等.缺血性脑卒中后抑郁患者血清淀粉样蛋白A水平检测及其意义研究[J].中华神经医学杂志,2018,17(11): 1143-1146. [68] DE OLIVEIRA EM, VISNIAUSKAS B, TUFIK S, et al. Serum amyloid a production is triggered by sleep deprivation in mice and humans:is that the link between sleep loss and associated comorbidities? Nutrients. 2017;9(3):311. [69] YU J, ZHU H, TAHERI S, et al. Serum amyloid a-mediated inflammasome activation of microglial cells in cerebral ischemia. J Neurosci. 2019;39(47): 9465-9476. |
| [1] | Zhao Jing, Liu Xiaobo, Zhang Yue, Zhang Jiaming, Zhong Dongling, Li Juan, Jin Rongjiang. Visualization analysis of neuromuscular electrical stimulation therapy based on CiteSpace: therapeutic effects, hot spots, and developmental trends [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1234-1241. |
| [2] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [3] | Zhao Yuwei, Gao Yuting, Li Zhen, Hao Huiqin . Mechanism of Ermiao San in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 742-748. |
| [4] | Yan Nan, Wu Yanlong, Tang Xiaohui, Zhang Xiaoyan, Wang Hui, Yang Tianze, Zhou Maochun, Wang Zhengdong, Yang Xiaoxia. Bone marrow mesenchymal stem cells may alleviate brain damage caused by the microglial overactivation in the cortex around ischemic site of stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3790-3795. |
| [5] | Chen Na, Wang Xiaohan, Zhang Yunke. Effect and mechanism of mesenchymal stem cells on aging-related ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3914-3920. |
| [6] | Liu Chao, Zeng Zhaomu, Wen Xichao, Wu Wensong, Sun Chengyuan, Zheng Kebin. Role and clinical application prospects of exosomal non-coding RNAs in the occurrence and development of glioma [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3928-3936. |
| [7] | Yang Demeng, Wang Changgeng, Hu Xinyuan, Ao Fei. Protective effect of eucommia alcohol extract on articular cartilage of osteoarthritis model rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3756-3761. |
| [8] | Deng Bowen, Li Xiaoye, Jiang Shengyuan, Liu Gan, Zhao Yi, Zhang Houjun, He Feng, Mu Xiaohong. Mechanisms of Huangqi Guizhi Wuwu Decoction in the treatment of cervical spondylosis and anxiety disorder based on the principle of “treating different diseases with the same method”: a network pharmacology analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3650-3656. |
| [9] | Xiakeerzhati•Xiaohalati, Wang Xiaobei, Wang Lin. Nanoparticles: a novel strategy for the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3566-3572. |
| [10] | Li Shulun, Hao Peng, Hao Fei, Duan Hongmei, Zhao Wen, Gao Yudan, Yang Chaoyang, Li Xiaoguang. Pathological changes in rats with ischemic stroke induced by improved photochemical embolization [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 218-224. |
| [11] | Yao Jiawei, Xu Xiongfeng, Yi Peng, Qiu Bo. Screening of differential genes and validation of key genes in synovial tissue of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2881-2887. |
| [12] | Yang Yanwen, Zhang Jing, Mao Yuling, Ni Jun, Mao Libin, Yao Lu, Wang Huiying, Liu Qiuming, Ma Zhenzhen. Establishment of a severe heatstroke pig model under high-temperature and high-humidity environment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2678-2684. |
| [13] | Xiao Xiang, Huang Dongfeng. Virtual reality training improves motor function of the upper limbs in stroke survivors: an evaluation using transcranial magnetic stimulation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2756-2761. |
| [14] | Li Chuanhong, Yu Xing, Yang Yongdong, Zhao He. Microglia in spinal cord injury: M1/M2 phenotypic polarization and neurotoxic/neuroprotective effects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2265-2272. |
| [15] | Chen Na, Fan Feiyan, Li Shuangli, Zhang Yunke. Effect and mechanism of traditional Chinese medicine regulating mesenchymal stem cell-derived exocrine for ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2081-2086. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||