Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (36): 5778-5784.doi: 10.3969/j.issn.2095-4344.0542
Previous Articles Next Articles
Endoplasmic reticulum stress participates in the process of high glucose-induced apoptosis in nucleus pulposus cells
Li Xianzhou, Zhang Cunxin
- (Department of Spine Surgery, East Branch of Jining No.1 People’s Hospital, Jining 272111, Shandong Province, China)
-
Received:2018-04-27Online:2018-12-28Published:2018-12-28 -
Contact:Zhang Cunxin, Master, Physician, Department of Spine Surgery, East Branch of Jining No.1 People’s Hospital, Jining 272111, Shandong Province, China -
About author:Li Xianzhou, Chief physician, Department of Spine Surgery, East Branch of Jining No.1 People’s Hospital, Jining 272111, Shandong Province, China
CLC Number:
Cite this article
Li Xianzhou, Zhang Cunxin. Endoplasmic reticulum stress participates in the process of high glucose-induced apoptosis in nucleus pulposus cells[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(36): 5778-5784.
share this article
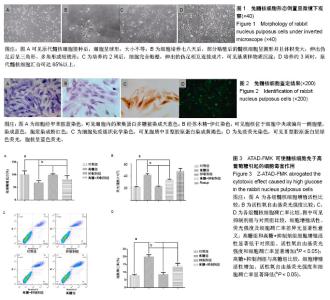
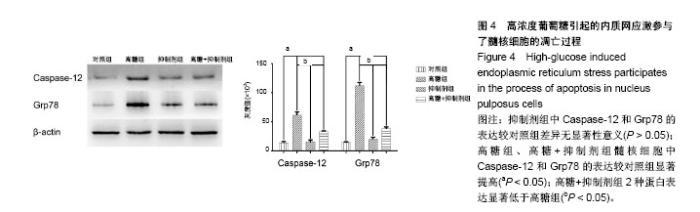
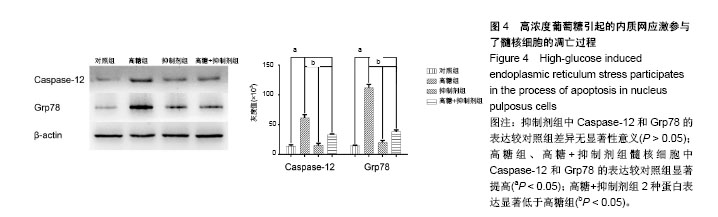
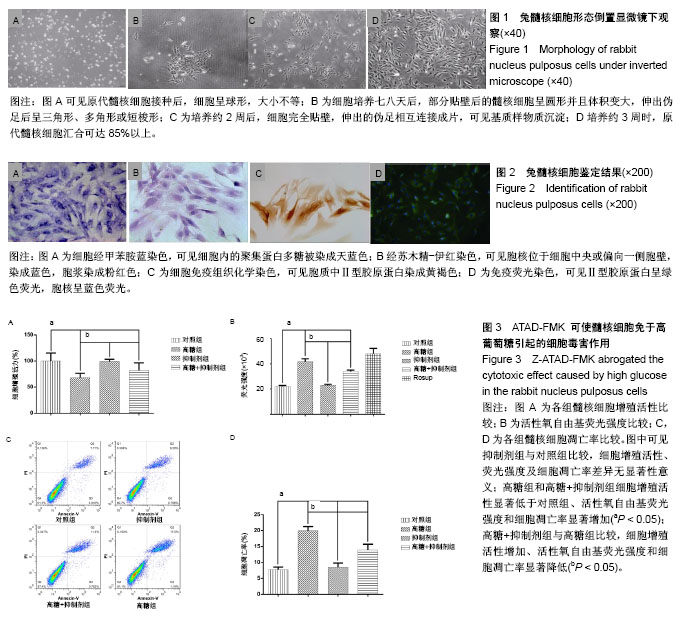
2.1 髓核细胞形态学观察 原代髓核细胞接种后,经倒置显微镜下观察,可见细胞呈球形,大小不等,有的吸附在培养瓶底,有的悬浮在培养液中。培养七八天后,可见有部分髓核细胞贴壁。贴壁后的髓核细胞呈圆形并且体积变大,伸出伪足后呈三角形、多角形或短梭形。细胞核较大,有时可见双核,核仁明显,胞浆中可见分泌颗粒,细胞折光性好。约2周后,髓核细胞完全贴壁,细胞伸出的伪足相互连接成片,细胞周围可见基质样物质沉淀。培养约3周时,原代髓核细胞汇合可达85%以上,见图1。 2.2 髓核细胞鉴定结果 2.2.1 髓核细胞经甲苯胺蓝染色 可见细胞内的聚集蛋白多糖被染成天蓝色,越靠近细胞核,着色越深,见图2A。 2.2.2 苏木精-伊红染色 可见髓核细胞呈三角形、多角形或短梭形等不规则形状,胞核位于细胞中央或偏向一侧胞壁,染成蓝色,胞浆染成粉红色,越靠近胞核,胞浆着色越深,见图2B。 2.2.3 细胞免疫组织化学染色 可见胞质中Ⅱ型胶原蛋白染成黄褐色,越靠近细胞核着色越深,见图2C。 2.2.4 免疫荧光染色 可见Ⅱ型胶原蛋白呈绿色荧光,大部分位于胞质中,且靠近胞核处荧光强度越强,胞核呈蓝色荧光,见图2D。 2.3 ATAD-FMK可使髓核细胞免于高葡萄糖引起的细胞毒害作用 CCK-8检测发现高浓度葡萄糖可显著抑制髓核细胞的增殖活性,使用Z-ATAD-FMK预处理2 h发现,Z-ATAD-FMK可显著拮抗高浓度葡萄糖所致的细胞毒性,见图3A。这种变化趋势同样发现在细胞内总活性氧的检测结果中,见图3B。流式细胞术检测发现,高糖组髓核细胞凋亡率显著增加,Z-ATAD-FMK预处理髓核细胞后,与对照组比较,虽然不能显著降低髓核细胞凋亡率,但与高糖组比较,其差异有显著性意义(P < 0.05) ,见图3C,D。 2.4 高浓度葡萄糖引起的内质网应激参与了髓核细胞的凋亡过程 Western blot技术用来分析Caspase-12和Grp78表达变化,使用β-actin作为内参。灰度分析发现,高浓度葡萄糖可显著增加Caspase-12和Grp78的表达,而Z-ATAD-FMK可抑制高糖引起的Caspase-12和Grp78的表达增加,见图4。"
| [1] Mcclelland AD, Kantharidis P.microRNA in the development of diabetic complications.Clin Sci (Lond).2014;126(2):95-110.[2] Teraguchi M,Yoshimura N,Hashizume H,et al.Metabolic Syndrome Components Are Associated with Intervertebral Disc Degeneration: The Wakayama Spine Study.PLoS One.2016;11(2): e147565.[3] Park JB,Byun CH,Park EY.Rat Notochordal Cells Undergo Premature Stress-Induced Senescence by High Glucose. Asian Spine J.2015;9(4):495-502.[4] Fields AJ,Berg-Johansen B,Metz L N,et al. Alterations in intervertebral disc composition, matrix homeostasis and biomechanical behavior in the UCD-T2DM rat model of type 2 diabetes.J Orthop Res.2015;33(5):738-746.[5] Mobbs RJ,Newcombe RL,Chandran KN.Lumbar discectomy and the diabetic patient: incidence and outcome. J Clin Neurosci.2001; 8(1):10-13.[6] Sakellaridis N.The influence of diabetes mellitus on lumbar intervertebral disk herniation. Surg Neurol.2006;66(2):152-154.[7] Gruber HE,Watts JA,Hoelscher GL,et al.Mitochondrial gene expression in the human annulus: in vivo data from annulus cells and selectively harvested senescent annulus cells.Spine J.2011; 11(8):782-791.[8] Kim KW,Chung HN,Ha KY,et al.Senescence mechanisms of nucleus pulposus chondrocytes in human intervertebral discs. Spine J.2009;9(8):658-666.[9] Kong JG, Park JB, Lee D,et al.Effect of high glucose on stress-induced senescence of nucleus pulposus cells of adult rats.Asian Spine J.2015;9(2):155-161.[10] Schwarz DS,Blower MD.The endoplasmic reticulum: structure, function and response to cellular signaling.Cell Mol Life Sci.2016; 73(1):79-94.[11] Pluquet O, Pourtier A, Abbadie C. The unfolded protein response and cellular senescence. A review in the theme: cellular mechanisms of endoplasmic reticulum stress signaling in health and disease. Am J Physiol Cell Physiol.2015;308(6):C415-C425.[12] Li Z,Yu C,Chen X,et al. Research on olaquindox induced endoplasmic reticulum stress related apoptosis on nephrotoxicity. Wei Sheng Yan Jiu.2015;44(3):444-450.[13] Peng P,Ma Q,Wang L,et al.Preconditioning With Tauroursodeoxycholic Acid Protects Against Contrast-Induced HK-2 Cell Apoptosis by Inhibiting Endoplasmic Reticulum Stress. Angiology.2015;66(10):941-949.[14] Ye W,Zhu W,Xu K,et al.Increased macroautophagy in the pathological process of intervertebral disc degeneration in rats. Connect Tissue Res.2013;54(1):22-28.[15] Mern DS,Beierfuss A,Thome C, et al. Enhancing human nucleus pulposus cells for biological treatment approaches of degenerative intervertebral disc diseases: a systematic review.J Tissue Eng Regen Med.2014;8(12):925-936.[16] Colombier P, Clouet J, Hamel O, et al. The lumbar intervertebral disc: from embryonic development to degeneration.Joint Bone Spine.2014,81(2):125-129.[17] Chen X,Zhu L,Wu G,et al.A comparison between nucleus pulposus-derived stem cell transplantation and nucleus pulposus cell transplantation for the treatment of intervertebral disc degeneration in a rabbit model.Int J Surg.2016;28:77-82.[18] Buchmaier BS,Bibi A,Muller GA,et al.Renal cells express different forms of vimentin: the independent expression alteration of these forms is important in cell resistance to osmotic stress and apoptosis.PLoS One.2013;8(7):e68301.[19] Tajeddine N.How do reactive oxygen species and calcium trigger mitochondrial membrane permeabilisation?. Biochim Biophys Acta. 2016;1860(6):1079-1088.[20] Roy N,Bagchi S,Raychaudhuri P.Damaged DNA binding protein 2 in reactive oxygen species (ROS) regulation and premature senescence.Int J Mol Sci.2012;13(9):11012-11026.[21] Wu Q, Ni X. ROS-mediated DNA methylation pattern alterations in carcinogenesis.Curr Drug Targets.2015;16(1):13-19.[22] Kroese LJ,Scheffer PG.8-hydroxy-2'-deoxyguanosine and cardiovascular disease: a systematic review. Curr Atheroscler Rep.2014;16(11):452.[23] Liu J, Du L. PERK pathway is involved in oxygen-glucose-serum deprivation-induced NF-kB activation via ROS generation in spinal cord astrocytes. Biochem Biophys Res Commun. 2015; 467(2): 197-203. [24] Liu D,Zhang H,Gu W,et al.Effects of exposure to high glucose on primary cultured hippocampal neurons: involvement of intracellular ROS accumulation. Neurol Sci.2014;35(6):831-837.[25] Zhao Y,Yan Y,Zhao Z,et al.The dynamic changes of endoplasmic reticulum stress pathway markers GRP78 and CHOP in the hippocampus of diabetic mice.Brain Res Bull.2015;111:27-35.[26] Fabrizio G,Di Paola S,Stilla A,et al. ARTC1-mediated ADP- ribosylation of GRP78/BiP: a new player in endoplasmic-reticulum stress responses. Cell Mol Life Sci.2015;72(6):1209-1225.[27] Park YJ,Yoo SA,Kim WU.Role of endoplasmic reticulum stress in rheumatoid arthritis pathogenesis.J Korean Med Sci.2014;29(1):2-11.[28] Zheng YZ,Cao ZG,Hu X,et al.The endoplasmic reticulum stress markers GRP78 and CHOP predict disease-free survival and responsiveness to chemotherapy in breast cancer.Breast Cancer Res Treat.2014;145(2):349-358.[29] Iurlaro R, Munoz-Pinedo C.Cell death induced by endoplasmic reticulum stress. FEBS J. 2016;283(14):2640-2652.[30] Lin P,Chen F,Sun J,et al.Mycotoxin zearalenone induces apoptosis in mouse Leydig cells via an endoplasmic reticulum stress- dependent signalling pathway. Reprod Toxicol.2015;52:71-77.[31] Shalini S,Dorstyn L,Dawar S,et al.Old, new and emerging functions of caspases.Cell Death Differ.2015;22(4):526-539.[32] Uchiyama R,Tsutsui H.Caspases as the key effectors of inflammatory responses against bacterial infection. Arch Immunol Ther Exp (Warsz).2015;63(1):1-13.[33] Yu M,Chen R,Jia Z,et al.MWCNTs Induce ROS Generation, ERK Phosphorylation, and SOD-2 Expression in Human Mesothelial Cells.Int J Toxicol.2016;35(1):17-26.[34] Vuleta A,Manitasevic JS,Tucic B.Adaptive flexibility of enzymatic antioxidants SOD, APX and CAT to high light stress: The clonal perennial monocot Iris pumila as a study case.Plant Physiol Biochem.2016;100:166-173.[35] Koretsi V,Kirschneck C,Proff P,et al.Expression of glutathione peroxidase 1 in the spheno-occipital synchondrosis and its role in ROS-induced apoptosis.Eur J Orthod.2015;37(3):308-313.[36] Tsuji T,Watanabe K,Hosogane N,et al.Risk factors of radiological adjacent disc degeneration with lumbar interbody fusion for degenerative spondylolisthesis.J Orthop Sci.2016;21(2):133-137.[37] Vergroesen PP,Kingma I,Emanuel KS,et al.Mechanics and biology in intervertebral disc degeneration: a vicious circle. Osteoarthritis Cartilage.2015;23(7):1057-1070.[38] Ma JF,Zang LN,Xi YM,et al.MiR-125a Rs12976445 Polymorphism is Associated with the Apoptosis Status of Nucleus Pulposus Cells and the Risk of Intervertebral Disc Degeneration.Cell Physiol Biochem.2016;38(1):295-305.[39] Battie MC,Videman T.Lumbar disc degeneration: epidemiology and genetics.J Bone Joint Surg Am.2006;88 Suppl 2:3-9. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [3] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Li Shibin, Lai Yu, Zhou Yi, Liao Jianzhao, Zhang Xiaoyun, Zhang Xuan. Pathogenesis of hormonal osteonecrosis of the femoral head and the target effect of related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 935-941. |
| [6] | Xu Yinqin, Shi Hongmei, Wang Guangyi. Effects of Tongbi prescription hot compress combined with acupuncture on mRNA expressions of apoptosis-related genes,Caspase-3 and Bcl-2, in degenerative intervertebral discs [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 713-718. |
| [7] | Zhang Wenwen, Jin Songfeng, Zhao Guoliang, Gong Lihong. Mechanism by which Wenban Decoction reduces homocysteine-induced apoptosis of myocardial microvascular endothelial cells in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 723-728. |
| [8] | Liu Qing, Wan Bijiang. Effect of acupotomy therapy on the expression of Bcl-2/Bax in synovial tissue of collagen-induced arthritis rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 729-734. |
| [9] | Xie Chongxin, Zhang Lei. Comparison of knee degeneration after anterior cruciate ligament reconstruction with or without remnant preservation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 735-740. |
| [10] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [11] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [12] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [13] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [14] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [15] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||