Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (4): 602-608.doi: 10.12307/2023.866
Previous Articles Next Articles
Progress in epigenetic regulation of vascular smooth muscle cell remodeling in the occurrence and development of aortic aneurysms
He Yuanjie, Chen Yuheng, Zhao Yongchao, Wang Zhenglong
- Department of Cardiology, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2022-11-15Accepted:2023-01-13Online:2024-02-08Published:2023-07-14 -
Contact:Wang Zhenglong, MD, Chief physician, Professor, Master’s supervisor, Department of Cardiology, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China Zhao Yongchao, MD, Associate researcher, Attending physician, Master’s supervisor, Department of Cardiology, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:He Yuanjie, Master candidate, Department of Cardiology, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 81760072 (to WZL); Basic Research Plan of Guizhou Provincial Department of Science and Technology, No. ZK[2022]general671 (to ZYC); Zunyi Excellent Youth Talent Training Project, No. ZSKHZ Zi [2022]366 (to ZYC); Research Initiation Fund of the Affiliated Hospital of Zunyi Medical University, No. YZ(2022)8 (to ZYC)
CLC Number:
Cite this article
He Yuanjie, Chen Yuheng, Zhao Yongchao, Wang Zhenglong. Progress in epigenetic regulation of vascular smooth muscle cell remodeling in the occurrence and development of aortic aneurysms[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 602-608.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
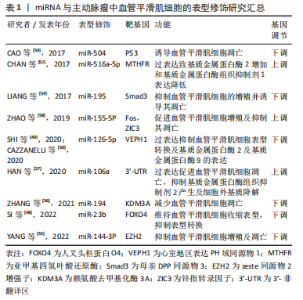
2.1 主动脉瘤与血管平滑肌细胞的相关性概述 主动脉瘤是一种高发病率和高死亡率的致命性心血管疾病,在65岁以上的男性人群中发病率高达 6%[9],在同龄女性人群中约1.3%[10]。与主动脉瘤相关的主要流行病学危险因素包括性别男性、65岁以上年龄和吸烟史[11],其他危险因素包括动脉粥样硬化性疾病、胆固醇、高血压、慢性阻塞性肺部疾病、种族和家族史等[12-14]。此外单核苷酸多态性、病毒感染如巨细胞病毒等其他因素也与动脉瘤的发病相关[15-17]。相反,动脉瘤的易感性与进展与糖尿病呈负相关,特别是2型糖尿病[18]。主动脉瘤可分为胸主动脉瘤及腹主动脉瘤[19],尽管两者发生的位置不同,但具有绝大多数共同的病理生理特征。主动脉壁由3层组成:内膜主要由内皮细胞组成、中膜由血管平滑肌细胞和细胞外基质组成、外膜由结缔组织组成[20]。血管平滑肌细胞功能障碍是主动脉壁结构完整性丧失的关键,在主动脉瘤的发生发展中起核心作用[21]。作为血管中膜的主要组成部分,血管平滑肌细胞具有明显的可塑性,血管平滑肌细胞表型转化可致血管功能障碍,促进主动脉瘤的形成与发展[22]。另一方面,基质金属蛋白酶和基质金属蛋白酶的组织抑制剂分泌的动态平衡是维持血管细胞外基质稳态的重要调节因素,当血管损伤时,血管平滑肌细胞可分泌多种炎症和趋化因子招募炎症细胞浸润并引发基质金属蛋白酶及基质金属蛋白酶的组织抑制剂释放失衡,导致管壁中基质金属蛋白酶的分泌过多,促使细胞外基质不成比例的降解并引发主动脉瘤的发生和破裂,因此,血管平滑肌细胞在血管的稳态维持以及血管的功能保护中发挥着不可或缺的作用[23-24]。 血管平滑肌细胞表型具有多样性和可变性的特点,在胚胎发育过程中血管平滑肌细胞由未分化表型逐渐分化为具有成熟特征的分化表型[25],当血管受到损伤或受到生长因子刺激时血管平滑肌细胞又从分化型转化为去分化型并获得增殖能力这个过程称为表型转化[26]。血管平滑肌细胞表型转化过程中,成熟的、可收缩的血管平滑肌细胞基因包括SM肌球蛋-白重链(smooth muscle myosin heavy chain,SM-MHC)、平滑肌22α(Sm22α)和α-肌动蛋白被“关闭”,而合成的、增殖的基因如骨桥蛋白则被“打开”,进而导致血管平滑肌细胞获得增殖、迁移和合成并分泌大量细胞外基质的能力。因此,血管平滑肌细胞的表型转化在主动脉瘤发生和进展中发挥了不可或缺的关键作用,如何调控相关基因网络的表达对于调节血管平滑肌细胞表型转化决定其细胞命运是诊治主动脉瘤的重要环节。在主动脉瘤的发生过程中,多种表观遗传修饰因素作为基因表达网络的重要调控方式,可调节并导致血管平滑肌细胞发生重塑,使血管平滑肌细胞在主动脉瘤中发生表型转换,即向去分化合成(增殖性)表型或衰老表型转变,最终可促进或抑制主动脉瘤的发生发展。 2.2 DNA甲基化与主动脉瘤 DNA甲基化是基因转录层面的表观遗传机制,在维持DNA结构、染色体稳定性及X染色体失活方面具有重要作用,并能调节转座子和反转录基因等元素和基因表达[27]。DNA甲基化主要与基因的转录抑制有关,其特征是通过 DNA 甲基转移酶(DNA methyltransferase,DNMT)将甲基转移到 DNA 的胞嘧啶环上,形成5-甲基胞嘧啶[28],而DNA双加氧酶TET2通过催化5-甲基胞嘧啶发生去甲基化来进行基因表达的调控[29]。 从主动脉瘤A患者中分离的血管平滑肌细胞发现,包括v-ets禽成红细胞增多症病毒E26癌基因同源物、白细胞介素6受体、赖氨酸甲基转移酶2( Lysine methyltransferase 2,SMYD2)、丝氨酸蛋白酶抑制剂B9在内的4个基因发生DNA甲基化水平改变。其中,主动脉瘤A患者血管平滑肌细胞的SMYD启动子区域甲基化程度明显低于对照组,提示DNA甲基化与主动脉瘤的发生发展密切相关[30]。SMYD2是一个在肌肉细胞中高表达的基因,参与肌原纤维的构成,其可使Hsp90甲基化,从而抑制Hsp90的表达,以提高肌节的稳定性。提示Hsp90甲基化或可成为主动脉瘤的干预靶点[21]。 研究显示,平滑肌22α(Sm22α)缺陷小鼠表现出妊娠诱导的T主动脉瘤,而Sm22α缺陷使马凡综合征Fbn1C1039G/+小鼠的主动脉瘤加重,验证了该基因产物是主动脉瘤效应物,而赖氨酸甲基转移酶Zeste 基因增强子人类同源物2(Zeste gene enhancer human homolog 2,EZH2)活性的增加增强了Sm22α的抑制作用,EZH2的抑制改善了Fbn1C1039G/+小鼠的主动脉性能,这与收缩蛋白表达(包括Sm22α)的恢复有关[31]。类似的研究显示,血管紧张素Ⅱ和CaCl2诱导的主动脉瘤A小鼠组织中Sm22α基因启动子的甲基化程度显著高于对照组织,两种模型的血管平滑肌细胞中Sm22α的基因敲除导致主动脉组织中主动脉瘤A形成加速,而过度表达则导致相反的效果,这表明基因特异性甲基化与主动脉瘤A发生之间可能存在关联[32]。 DNA甲基化是研究最广泛的对DNA的表观遗传修饰。以上研究表明,通过DNA甲基转移酶将甲基添加到胞嘧啶5’碱基到鸟嘌呤(CpG二核苷酸)上,这可以抑制基因转录,进而通过靶向血管平滑肌细胞重塑相关基因,调控主动脉瘤的发生和发展,目前围绕DNA甲基化在主动脉瘤中作用的研究非常有限,文章论证了主动脉瘤中特定基因的甲基化变化以及在主动脉瘤中的潜在作用,可能在未来的表观遗传学疗法中有用。 2.3 组蛋白修饰与主动脉瘤 组蛋白是真核生物染色体的结构蛋白,负责染色质的压缩。组蛋白修饰酶可以通过添加或移除甲基或乙酰基来调节基因表达,组蛋白的甲基化和去甲基化分别通过组蛋白甲基转移酶(histone methyltransferase,HMT)和组蛋白去甲基酶实现[33]。组蛋白乙酰转移酶(histone acetyltransferase,HAT)和组蛋白脱乙酰酶(histone deacetylase,HDAC)分别促进乙酰化和脱乙酰化,组蛋白乙酰化与转录活性有关,而组蛋白去乙酰化与转录抑制相关[34]。 组蛋白甲基化既可以诱导转录激活,也可以诱导转录抑制,这取决于甲基的数量和位置,活跃的转录状态以阳性标记为特征,如H3K4,H3K36和H3K79的二甲基化或三甲基化(me2/me3),抑制转录状态表现为H3K9me2/me3和H3K27me3 上标记的增加[35]。HDAC是基因转录的有效表观遗传调节因子,它们由18个分子组成,分为4类:Ⅰ、Ⅱ(包括Ⅱa和Ⅱb)、Ⅲ和Ⅳ类,Ⅰ类和Ⅱa类HDAC已被证明可调节参与主动脉瘤A的血管平滑肌细胞基因的表达,并调节分化、收缩、增殖、炎症和细胞外基质沉积,从而影响主动脉瘤A的发生和发展[36]。在血管紧张素Ⅱ注入ApoE-/-小鼠的主动脉瘤表现出类似的HDAC表达谱。使用Ⅰ类HDAC抑制剂(MS-275)或Ⅱa类抑制剂(MC-1568)治疗后可提高小鼠生存率,降低主动脉瘤A的发生率和严重程度[37]。 Smad2启动子上特定组蛋白残基的甲基化与乙酰化的增加与从马凡氏综合征、主动脉瓣二叶畸形和非遗传性T主动脉瘤患者的主动脉组织样本分离的血管平滑肌细胞中Smad2蛋白的增加相关[38]。Smad2在这些血管平滑肌细胞中的过度表达被证明受组蛋白乙酰转移酶(p300/PCAF)的调节 [39]。T主动脉瘤与影响转化生长因子β信号通路成员或血管平滑肌细胞肌动球蛋白细胞骨架成分和调节因子的突变有关。影响转化生长因子β信号和血管平滑肌细胞骨架的突变都会导致形成一种三元复合物,包括组蛋白脱乙酰酶HDAC9、染色质重塑酶BRG1和MALAT1,HDAC9-MALAT1-BRG1复合物结合染色质并抑制收缩蛋白基因表达,与 H3K27me3的获得相关,破坏MALAT1或HDAC9可恢复收缩蛋白表达,改善主动脉壁结构,抑制实验性动脉瘤生长[40]。最近一项比较主动脉瘤和健康主动脉样本的研究报告了多种组蛋白乙酰化转移酶在心血管疾病中显著升高,KAT2B,KAT3A,KAT3B和KAT6B 在主动脉瘤组织中的表达量最高,KAT6A 表达与收缩性血管平滑肌细胞标志物肌球蛋白重链 11 (myosin heavy chain 11,MYH11) 以及血管细胞黏附分子 1及内皮细胞相关[4]。综上,组蛋白修饰相关酶可通过诱导转录激活或抑制在血管平滑肌细胞基因调控中发挥重要作用,并参与血管平滑肌细胞表型和功能的改变,以应对环境压力,是主动脉瘤发生发展过程中重要的调控环节和调控方式,通过靶向相关酶类分子,或可成为主动脉瘤治疗的相关靶点。 2.4 非编码RNA与主动脉瘤 非编码RNA(Non-coding RNA,ncRNA)是指转录组中不直接编码产生蛋白质的RNA分子,包括核糖体RNA(rRNA)、转运RNA(tRNA) 、微小RNA(miRNA) 及长链非编码RNA(long noncoding RNA,lncRNA)等多种 RNAs类型,这些RNAs的共同特点是都能从基因组上转录而来,但是不翻译成蛋白。研究显示,非编码RNAs在通过调节血管平滑肌细胞凋亡、表型转换、细胞外基质降解等在主动脉瘤的发生发展中同样起着关键的作用[41-44]。文章主要讨论miRNAs及lncRNA在主动脉瘤发生发展中扮演的角色及其分子机制。 2.4.1 miRNAs miRNA 是长约 22 nt 的内源性 RNA,通常通过与信使 RNA 的 3’-非翻译区(3’-UTR)结合,在转录后抑制靶基因的表达[45]。研究表明,血管平滑肌细胞的稳态受到大量miRNA的强烈调节[46]。在主动脉瘤的相关研究中报道了多种miRNAs,这些miRNAs可通过靶向特定基因以调节血管平滑肌细胞可塑性以及细胞外基质降解,进而促进或改善主动脉瘤的发病及进展[47-59],见表1。miRNAs作为非编码RNAs家族中的重要成员,其研究较为深入、作用机制明确,在主动脉瘤发生发展中的功能较为清晰。尽管当前对于miRNAs的大规模转化及临床研究尚未取得令人满意的效果。未来,通过相关基因工程学手段递送或抑制相关miRNAs的表达,有望在主动脉瘤的临床诊疗过程中发挥重要作用。"
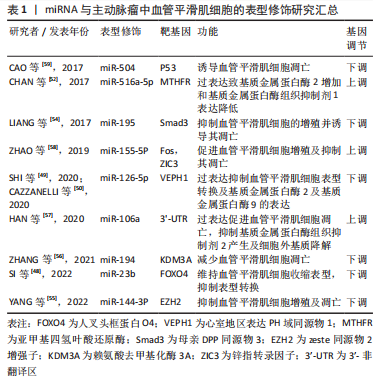
miRNA通过调节血管平滑肌细胞表型转换调节主动脉瘤的发生发展:miRNA被证明可抑制血管平滑肌细胞表型转换,从而稳定血管平滑肌细胞收缩表型,抑制主动脉瘤的进展[47]。例如miR-23b最近被证明靶向转录因子叉头框O4(transcription factor forkhead box O4,FoxO4)抑制血管平滑肌细胞表型转换,在AngⅡ诱导的血管平滑肌细胞表型转换过程中,miR-23b敲除显著增加了FoxO4的表达,揭示了miR-23b通过维持血管平滑肌细胞收缩表型在预防主动脉瘤形成中的关键作用[48]。同样,miR-126-5p 通过直接负调控心室地区表达PH 域同源物1(ventricular zone expressed PH domain homolog 1,VEPH1)表达来促进人主动脉 血管平滑肌细胞分化,从而导致成熟、收缩基因(如Sma和Myh11)的表达增加,以及合成基因(如波形蛋白和增殖细胞核抗原)的表达减少[49],进而抑制主动脉瘤的发生、发展。 miRNA调节血管平滑肌细胞基质成分参与主动脉瘤的发生发展:主动脉壁由内膜、中膜和外膜组成,内侧平滑肌和细胞外基质结构蛋白(主要是胶原蛋白和弹性蛋白)的排列对维持主动脉的结构和功能至关重要。在主动脉瘤发育过程中,这些层和蛋白质组分的完整性被破坏,细胞外基质的降解是主动脉瘤的特征之一,但这一过程的机制尚未完全了解。miRNA已被证明可调节参与血管壁基质合成和降解的关键成分的表达[50]。miR-126a-5p除了被证明能维持血管平滑肌细胞成熟表型外,也改善了血管紧张素Ⅱ诱导的弹性片段和细胞外基质降解。 miR-126a-5p抑制了主动脉中膜血管平滑肌细胞中观察到ADAM金属肽酶与血小板反应蛋白1型基序4(Adam metallopeptidase and thrombospondin type 1 motif 4,ADAMTS-4;一种调节基质降解的分泌蛋白酶)的强烈上调,抑制了主动脉瘤的形成[51]。一项研究检测了miR-516a-5p和亚甲基四氢叶酸还原酶(Methylenetetrahydrofolate reductase,MTHFR)/同型半胱氨酸途径的作用,该途径与主动脉瘤A发病机制有关,研究表明,miR-516a-5p过表达抑制了MTHFR的表达,还导致基质金属蛋白酶2的表达增加和基质金属蛋白酶抑制剂1的表达降低[52],从而促进主动脉瘤A的形成。 miRNA调节血管平滑肌细胞凋亡和增殖参与主动脉瘤的发生发展:miRNA可以调节血管平滑肌细胞凋亡,这可能是主动脉瘤形成的另一种机制[53]。MiR-195通过靶向Smad3抑制血管平滑肌细胞的增殖并诱导其凋亡,在主动脉瘤进展中促进血管平滑肌细胞重塑 [54]。另一种miRNA,miR-144-3p过表达通过与EZH2的 3’-UTR结合,从而抑制EZH2表达,减少血管平滑肌细胞增殖,此外,EZH2在p21的启动子区域高度富集,p21表达的下调可以挽救miR-144-3p对血管平滑肌细胞增殖和凋亡的影响,miR-144-3p作为血管平滑肌细胞凋亡的启动子,抑制主动脉瘤A的发生和发展 [55]。在类似的研究中,miR-194通过赖氨酸去甲基化酶3A(lysine demethylase 3A,KDM3A)抑制BCL2 相互作用蛋白3的表达,减少血管平滑肌细胞凋亡,对主动脉瘤A的进展起到抑制作用[56]。 miR-106a的表达增加促进血管平滑肌细胞凋亡,并通过直接靶向其3’-UTR下调基质金属蛋白酶抑制剂2,从而恢复基质金属蛋白酶的产生并最终加速细胞外基质的降解,从而促进主动脉瘤的进展[57]。miR-155-5p可通过靶向3’-UTR抑制血管平滑肌细胞中Fos和锌指转录因子3(Zinc finger transcription factor 3,ZIC3)的表达,进而抑制血管平滑肌细胞活力及凋亡[58]。最后研究还表明,miR-504抑制p53依赖性血管平滑肌细胞凋亡,并防止主动脉瘤形成[59]。 2.4.2 lncRNAs lncRNAs通常被定义为长度大于200 nt且不编码蛋白质的RNA。它们参与许多生物事件,例如通过支架蛋白发出信号、形成核结构、压印基因组位点和调节酶活性。lncRNA基因的异常表达或突变与多种人类疾病有关。表2显示了在主动脉瘤发育过程中参与调节血管平滑肌细胞的lnc-RNAs的概况以及这些lncRNA表达水平如何影响血管平滑肌细胞功能[60-69]。"
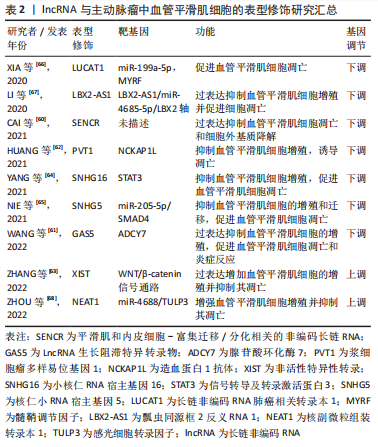

平滑肌和内皮细胞-富集迁移/分化相关的非编码长链RNA (Smooth muscle and endothelial cell-enriched migration/differentiation- associated long noncoding RNA,SENCR)是从人冠状动脉平滑肌细胞的RNA测序中被鉴定出的。该研究发现,在主动脉瘤A组织和血管紧张素Ⅱ刺激的血管平滑肌细胞中SENCR下调,SENCR的过度表达可以抑制血管紧张素Ⅱ诱导的血管平滑肌细胞凋亡,而SENCR的抑制促进血管紧张素Ⅱ诱发的血管平滑肌细胞凋亡。此外,SENCR过度表达后,血管紧张素Ⅱ诱导的血管平滑肌细胞中基质金属蛋白酶2和基质金属蛋白酶9的表达减少,而基质金属蛋白酶抑制剂1的表达增加。在体内,SENCR的过度表达改善了血管紧张素Ⅱ诱导的主动脉组织的病理变化和动脉壁弹性纤维的损伤,并抑制了血管紧张素Ⅱ诱导的细胞凋亡和细胞外基质的降解。总体而言,SENCR的过度表达通过抑制血管平滑肌细胞凋亡和细胞外基质降解来抑制主动脉瘤A的形成,为SENCR作为主动脉瘤A治疗的潜在靶点提供了可靠的证据[60]。 LncRNA生长阻滞特异转录物5(growth arrest specific 5,Gas5)被发现在主动脉瘤A组织和血管紧张素Ⅱ诱导的人主动脉平滑肌细胞中上调。GAS5的过表达抑制了血管紧张素Ⅱ诱导的人主动脉平滑肌细胞的增殖,促进了凋亡和炎症反应。MiR-185-5p可被GAS5吸收,其抑制剂可逆转GAS5沉默对血管紧张素Ⅱ诱导的人主动脉平滑肌细胞的增殖、凋亡和炎症反应的影响。腺苷酸环化酶7是miR-185-5p的靶点。腺苷酸环化酶7敲低增加了血管紧张素Ⅱ诱导的人主动脉平滑肌细胞的增殖,同时减少了凋亡和炎症反应。此外,过表达的腺苷酸环化酶7逆转了miR-185-5p过表达对血管紧张素Ⅱ诱导的人主动脉平滑肌细胞的增殖、凋亡和炎症反应的影响。GAS5通过吸收miR-185-5p来积极调节腺苷酸环化酶7的表达,以抑制AKT信号通路的活性。GAS5通过调节人主动脉平滑肌细胞的增殖、凋亡和炎症反应促进主动脉瘤A的形成,这可能为主动脉瘤A的治疗提供新的思路[61]。 LncRNA浆细胞瘤变异易位1(plasmacytoma variant translocation 1,PVT1)也被证明与主动脉瘤A的进展有关。研究表明,PVT1作为miR-3127-5p的海绵调节造血蛋白1抗体(hematopoietic protein 1 antibody,NCKAP1L)的表达,从而抑制血管平滑肌细胞增殖、诱导凋亡和激活炎症。PVT1通过miR-3127-5p/NCKAP1L轴参与主动脉瘤A进展,可能是主动脉瘤A的一个有前途的生物标志物和治疗靶点[62]。另有学者研究了lncRNA非活性特异性转录物(X-inactive specific transcript,XIST)在血管平滑肌细胞进化中的功能和分子机制,XIST通过miR-1264靶向WNT/β-catenin信号通路抑制细胞凋亡并刺激HA血管平滑肌细胞的增殖,表明XIST在主动脉瘤A中的潜在作用[63]。核仁RNA宿主基因16 (Small nucleolar RNA host gene 16,SNHG16)通过吸附miR-106b-5p释放信号转导转录激活蛋白3(Signal transducer and Transcription activator 3,STAT3)使其不受miR-106b-5p的抑制作用,SNHG16/miR-106b-5p/STAT3通过抑制血管平滑肌细胞增殖,促进其凋亡,参与调节主动脉瘤A的进展,这表明了主动脉瘤A发病机制的可能靶点[64]。 核仁小RNA宿主基因5(small nucleolar RNA host gene 5,SNHG5)被报道可通过调节miR-205-5p/SMAD4轴调节血管平滑肌细胞的增殖和凋亡,从而抑制主动脉瘤A的发生发展[65]。一项研究揭示了长链非编码RNA肺癌相关转录本1(long strand non coding RNA lung cancer related transcript 1,LUCAT1)通过直接靶向miR-199a-5p提高髓鞘调节因子(myelin regulatory factors,MYRF)表达促进血管平滑肌细胞凋亡,为预防主动脉瘤A的形成提供有价值的信息[66]。同样也有实验研究了瓢虫同源框2反义RNA 1(ladybird homeobox 2 antisense RNA 1,LBX2-AS1)或瓢虫同源框2(ladybird homeobox 2,LBX2)对血管平滑肌细胞生物学过程的影响,研究证实LBX2-AS1和LBX2在主动脉瘤A中均具有促血管平滑肌细胞凋亡和抗增殖功能,LBX2-AS1通过ceRNA机制隔离miR-4685-5p以释放LBX2表达,由LBX2-AS1,miR-4685-5p和LBX2形成正反馈环,促使主动脉瘤A形成和进展恶化,这意味着LBX2-AS1/miR-4685-5p/LBX2轴在主动脉瘤A治疗中的潜在用途[67]。此外,抑STAT3可促进血管平滑肌细胞增殖,抑制血管平滑肌细胞凋亡。核副微粒组装转录本1(nuclear paraspeckle assembly transcript1,NEAT1)被认为是多种疾病的致癌基因,然而有研究发现敲低NEAT1增强了血管平滑肌细胞的增殖并损害了细胞凋亡,在机制上,STAT3充当转录因子促成NEAT1转录,由STAT3诱导的lncRNA NEAT1被鉴定为ceRNA并且通过靶向miR-4688/TULP3轴促进主动脉瘤形成[68]。 与miRNAs通过互补结合调节其靶mRNAs的相对直接的机制不同,lncRNAs已被证明是转录和翻译的调控因子,例如通过充当分子诱饵和支架,或通过蛋白质结合将核糖核蛋白复合体引导到其靶标。除此之外,它们还可以作为宿主基因转录miRNAs(在细胞核中)或作为miRNA海绵(在细胞质中)。而仅仅是lncRNA转录过程本身就能调节基因表达,进一步增加了lncRNAs作用机制的复杂性。上述研究表明,lncRNA在主动脉瘤的发展和扩张机制中(例如血管平滑肌细胞的增殖和凋亡)起着至关重要的作用,其主要通过多种分子机制调节血管平滑肌细胞重塑相关基因的表达,在文章引用的文献报道能够提供体内证据,揭示了lncRNAs本身的多种多样的调控机制,也反应了主动脉瘤的发生发展过程中lncRNAs的表观转录后修饰的复杂性。许多lncRNA已被确定为主动脉瘤的关键调节因子,而且还在不断增加,但需要更多的证据来确定并证明它们在主动脉瘤中的作用机制及其诊断和治疗潜力。"

| [1] BOSSONE E, EAGLE KA. Epidemiology and management of aortic disease: aortic aneurysms and acute aortic syndromes. Nat Rev Cardiol. 2021;18(5):331-348. [2] GUO DC, PAPKE CL, HE R, et al. Pathogenesis of thoracic and abdominal aortic aneurysms. Ann N Y Acad Sci. 2006;1085:339-352. [3] RYER EJ, RONNING KE, ERDMAN R, et al. The potential role of DNA methylation in abdominal aortic aneurysms. Atherosclerosis. 2015;241(1): 121-129. [4] HAN Y, TANIOS F, REEPS C, et al. Histone acetylation and histone acetyltransferases show significant alterations in human abdominal aortic aneurysm. Clin Epigenetics. 2016;8:3. [5] MAEGDEFESSEL L, SPIN JM, RAAZ U, et al. miR-24 limits aortic vascular inflammation and murine abdominal aneurysm development. Nat Commun. 2014;5:5214. [6] OSPELT C. A brief history of epigenetics. Immunol Lett. 2022;249:1-4. [7] PEIXOTO P, CARTRON PF, SERANDOUR AA, et al. From 1957 to nowadays: a brief history of epigenetics. Int J Mol Sci. 2020;21(20):7571. [8] ROMBOUTS KB, VAN MERRIENBOER TAR, KET JCF, et al. The role of vascular smooth muscle cells in the development of aortic aneurysms and dissections. Eur J Clin Invest. 2022;52(4):e13697. [9] GOLLEDGE J, MULLER J, DAUGHERTY A, et al. Abdominal aortic aneurysm: pathogenesis and implications for management. Arterioscler Thromb Vasc Biol. 2006;26(12):2605-2613. [10] WANHAINEN A, VERZINI F, VAN HERZEELE I, et al. Editor’s Choice -European Society for Vascular Surgery (ESVS). 2019 Clinical Practice Guidelines on the Management of Abdominal Aorto-iliac Artery Aneurysms. Eur J Vasc Endovasc Surg. 2019;57(1):8-93. [11] ALTOBELLI E, RAPACCHIETTA L, PROFETA VF, et al. Risk factors for abdominal aortic aneurysm in population-based studies: a systematic review and meta-analysis. Int J Environ Res Public Health. 2018;15(12):2805. [12] GRØNDAL N, SØGAARD R, LINDHOLT JS. Baseline prevalence of abdominal aortic aneurysm, peripheral arterial disease and hypertension in men aged 65-74 years from a population screening study (VIVA trial). Br J Surg. 2015;102(8):902-906. [13] TANG W, YAO L, ROETKER NS, et al. Lifetime risk and risk factors for abdominal aortic aneurysm in a 24-year prospective study: theARIC Study (Atherosclerosis Risk in Communities). Arterioscler Thromb Vasc Biol. 2016;36(12):2468-2477. [14] SAKALIHASAN N, DEFRAIGNE JO, KERSTENNE MA, et al. Family members of patients with abdominal aortic aneurysms are at increased risk for aneurysms: analysis of 618 probands and their families from the Liège AAA Family Study. Ann Vasc Surg. 2014;28(4):787-797. [15] VATS S, SUNDQUIST K, WANG X, et al. Associations of global DNA methylation and homocysteine levels with abdominal aortic aneurysm: a cohort study from a population-based screening program in Sweden. Int J Cardiol. 2020;321:137-142. [16] TOGHILL BJ, SARATZIS A, HARRISON SC, et al. The potential role of DNA methylation in the pathogenesis of abdominal aortic aneurysm. Atherosclerosis. 2015;241(1):121-129. [17] JABŁOŃSKA A, ZAGRAPAN B, PARADOWSKA E, et al. Abdominal aortic aneurysm and virus infection: a potential causative role for cytomegalovirus infection? J Med Virol. 2021;93(8):5017-5024. [18] YUAN Z, LU Y, WEI J, et al. Abdominal aortic aneurysm: roles of inflammatory cells. Front Immunol. 2021;11:609161. [19] QIAN G, ADEYANJU O, OLAJUYIN A, et al. Abdominal aortic aneurysm formation with a focus on vascular smooth muscle cells. Life (Basel). 2022; 12(2):191. [20] MAZUREK R, DAVE JM, CHANDRAN RR, et al. Vascular cells in blood vessel wall development and disease. Adv Pharmacol. 2017;78:323-350. [21] QUINTANA RA, TAYLOR WR. Cellular mechanisms of aortic aneurysm formation. Circ Res. 2019;124(4):607-618. [22] PAN L, BAI P, WENG X, et al. Legumain is an endogenous modulator of integrin αvβ3 triggering vascular degeneration, dissection, and rupture. Circulation. 2022;145(9):659-674. [23] KHANAFER K, GHOSH A, VAFAI K. Correlation between MMP and TIMP levels and elastic moduli of ascending thoracic aortic aneurysms. Cardiovasc Revasc Med. 2019;20(4):324-327. [24] KOULLIAS GJ, RAVICHANDRAN P, KORKOLIS DP, et al. Increased tissue microarray matrix metalloproteinase expression favors proteolysis in thoracic aortic aneurysms and dissections. Ann Thorac Surg. 2004;78(6): 2106-2111. [25] FRISMANTIENE A, PHILIPPOVA M, ERNE P, et al. Smooth muscle cell-driven vascular diseases and molecular mechanisms of VSMC plasticity. Cell Signal. 2018;52:48-64. [26] ALLEN CL, BAYRAKTUTAN U. Differential mechanisms of angiotensin II and PDGF-BB on migration and proliferation of coronary artery smooth muscle cells. J Mol Cell Cardiol. 2008;45(2):198-208. [27] GREENBERG MVC, BOURC’HIS D. The diverse roles of DNA methylation in mammalian development and disease. Nat Rev Mol Cell Biol. 2019;20(10):590-607. [28] SCHMITZ RJ, LEWIS ZA, GOLL MG. DNA methylation: shared and divergent features across eukaryotes. Trends Genet. 2019;35(11):818-827. [29] CHEN LL, LIN HP, ZHOU WJ, et al. SNIP1 recruits TET2 to regulate c-MYC target genes and cellular DNA damage response. Cell Rep. 2018;25(6): 1485-1500.e4. [30] TOGHILL BJ, SARATZIS A, FREEMAN PJ, et al. SMYD2 promoter DNA methylation is associated with abdominal aortic aneurysm (AAA) and SMYD2 expression in vascular smooth muscle cells. Clin Epigenetics. 2018;10:29. [31] LINO CARDENAS CL, KESSINGER CW, MACDONALD C, et al. Inhibition of the methyltranferase EZH2 improves aortic performance in experimental thoracic aortic aneurysm. JCI Insight. 2018;3(5):e97493. [32] ZHONG L, HE X, SI X, et al. SM22α (smooth muscle 22α) Prevents aortic aneurysm formation by inhibiting smooth muscle cell phenotypic switching through suppressing reactive oxygen species/NF-κB (nuclear factor-κB). Arterioscler Thromb Vasc Biol. 2019;39(1):e10-e25. [33] VAN DEN BOSSCHE J, NEELE AE, HOEKSEMA MA, et al. Macrophage polarization: theepigenetic point of view. Curr Opin Lipidol. 2014;25(5): 367-373. [34] STERNER DE, BERGER SL. Acetylation of histones and transcription-related factors. Microbiol Mol Biol Rev. 2000;64(2):435-459. [35] GREER EL, SHI Y. Histone methylation: a dynamic mark in health, disease and inheritance. Nat Rev Genet. 2012;13(5):343-357. [36] PONS D, JUKEMA JW. Epigenetic histone acetylation modifiers in vascular remodelling - new targets for therapy in cardiovascular disease. Neth Heart J. 2008;16(1):30-32. [37] GALÁN M, VARONA S, ORRIOLS M, et al. Induction of histone deacetylases (HDACs) in human abdominal aortic aneurysm: therapeutic potential of HDAC inhibitors. Dis Model Mech. 2016;9(5):541-552. [38] GOMEZ D, COYET A, OLLIVIER V, et al. Epigenetic control of vascular smooth muscle cells in Marfan and non-Marfan thoracic aortic aneurysms. Cardiovasc Res. 2011;89(2):446-456. [39] GOMEZ D, KESSLER K, MICHEL JB, et al. Modifications of chromatin dynamics control Smad2 pathway activation in aneurysmal smooth muscle cells. Circ Res. 2013;113(7):881-890. [40] LINO CARDENAS CL, KESSINGER CW, CHENG Y, et al. An HDAC9-MALAT1-BRG1 complex mediates smooth muscle dysfunction in thoracic aortic aneurysm. Nat Commun. 2018;9(1):1009. [41] KUMAR S, BOON RA, MAEGDEFESSEL L, et al. Role of noncoding RNAs in the pathogenesis of abdominal aortic aneurysm. Circ Res. 2019;124(4):619-630. [42] WU ZY, TRENNER M, BOON RA, et al. Long noncoding RNAs in key cellular processes involved in aortic aneurysms. Atherosclerosis. 2020;292:112-118. [43] KALAYINIA S, ARJMAND F, MALEKI M, et al. MicroRNAs: roles in cardiovascular development and disease. Cardiovasc Pathol. 2021;50: 107296. [44] LI Y, MAEGDEFESSEL L. Non-coding RNA contribution to thoracic and abdominal aortic aneurysm disease development and progression. Front Physiol. 2017;8:429. [45] VISHNOI A, RANI S. MiRNA biogenesis and regulation of diseases: an overview. Methods Mol Biol. 2017;1509:1-10. [46] MANGUM KD, FARBER MA. Genetic and epigenetic regulation of abdominal aortic aneurysms. Clin Genet. 2020;97(6):815-826. [47] ADAM M, RAAZ U, SPIN JM, et al. MicroRNAs in abdominal aortic aneurysm. Curr Vasc Pharmacol. 2015;13(3):280-290. [48] SI X, CHEN Q, ZHANG J, et al. MicroRNA-23b prevents aortic aneurysm formation by inhibiting smooth muscle cell phenotypic switching via FoxO4 suppression. Life Sci. 2022;288:119092. [49] SHI X, MA W, PAN Y, et al. MiR-126-5p promotes contractile switching of aortic smooth muscle cells by targeting VEPH1 and alleviates Ang II-induced abdominal aortic aneurysm in mice. Lab Invest. 2020;100(12):1564-1574. [50] CAZZANELLI P, WUERTZ-KOZAK K. MicroRNAs in intervertebral disc degeneration, apoptosis, inflammation, and mechanobiology. Int J Mol Sci. 2020;21(10):3601. [51] LI L, MA W, PAN S, et al. MiR-126a-5p limits the formation of abdominal aortic aneurysm in mice and decreases ADAMTS-4 expression. J Cell Mol Med. 2020;24(14):7896-7906. [52] CHAN CYT, CHEUK BLY, CHENG SWK. Abdominal aortic aneurysm-associated MicroRNA-516a-5p regulates expressions of methylenetetrahydrofolate reductase, matrix metalloproteinase-2, and tissue inhibitor of matrix metalloproteinase-1 in human abdominal aortic vascular smooth muscle cells. Ann Vasc Surg. 2017;42:263-273. [53] DING W, LIU Y, SU Z, et al. Emerging role of non-coding RNAs in aortic dissection. Biomolecules. 2022;12(10):1336. [54] LIANG B, CHE J, ZHAO H, et al. MiR-195 promotes abdominal aortic aneurysm media remodeling by targeting Smad3. Cardiovasc Ther. 2017. doi: 10.1111/1755-5922.12286. [55] YANG Z, ZHANG L, LIU Y, et al. Potency of miR-144-3p in promoting abdominal aortic aneurysm progression in mice correlates with apoptosis of smooth muscle cells. Vascul Pharmacol. 2022;142:106901. [56] ZHANG H, WANG Y, BIAN X, et al. MicroRNA-194 acts as a suppressor during abdominal aortic aneurysm via inhibition of KDM3A-mediated BNIP3. Life Sci. 2021;277:119309. [57] HAN ZL, WANG HQ, ZHANG TS, et al. Up-regulation of exosomal miR-106a may play a significant role in abdominal aortic aneurysm by inducing vascular smooth muscle cell apoptosis and targeting TIMP-2, an inhibitor of metallopeptidases that suppresses extracellular matrix degradation. Eur Rev Med Pharmacol Sci. 2020;24(15):8087-8095. [58] ZHAO L, OUYANG Y, BAI Y, et al. miR-155-5p inhibits the viability of vascular smooth muscle cell via targeting FOS and ZIC3 to promoteb aneurysm formation. Eur J Pharmacol. 2019;853:145-152. [59] CAO X, CAI Z, LIU J, et al. miRNA504 inhibits p53dependent vascular smooth muscle cell apoptosis and may prevent aneurysm formation. Mol Med Rep. 2017;16(3):2570-2578. [60] CAI Z, HUANG J, YANG J, et al. LncRNA SENCR suppresses abdominal aortic aneurysm formation by inhibiting smooth muscle cells apoptosis and extracellular matrix degradation. Bosn J Basic Med Sci. 2021;21(3):323-330. [61] WANG Y, ZHAI S, XING J, et al. LncRNA GAS5 promotes abdominal aortic aneurysm formation through regulating the miR-185-5p/ADCY7 axis. Anticancer Drugs. 2022;33(3):225-234. [62] HUANG Y, REN L, LI J, et al. Long non-coding RNA PVT1/microRNA miR-3127-5p/NCK-associated protein 1-like axis participates in the pathogenesis of abdominal aortic aneurysm by regulating vascular smooth muscle cells. Bioengineered. 2021;12(2):12583-12596. [63] ZHANG D, LU D, XU R, et al. Inhibition of XIST attenuates abdominal aortic aneurysm in mice by regulating apoptosis of vascular smooth muscle cells through miR-762/MAP2K4 axis. Microvasc Res. 2022;140: 104299. [64] YANG B, WANG X, YING C, et al. Long noncoding RNA SNHG16 facilitates abdominal aortic aneurysm progression through the miR-106b-5p/STAT3 feedback loop. J Atheroscler Thromb. 2021;28(1):66-78. [65] NIE H, ZHAO W, WANG S, et al. Based on bioinformatics analysis lncrna SNHG5 modulates the function of vascular smooth muscle cells through mir-205-5p/SMAD4 in abdominal aortic aneurysm. Immun Inflamm Dis. 2021;9(4):1306-1320. [66] XIA Q, ZHANG L, YAN H, et al. LUCAT1 contributes to MYRF-dependent smooth muscle cell apoptosis and may facilitate aneurysm formation via the sequestration of miR-199a-5p. Cell Biol Int. 2020;44(3):755-763. [67] LI H, ZHANG H, WANG G, et al. LncRNA LBX2-AS1 facilitates abdominal aortic aneurysm through miR-4685-5p/LBX2 feedback loop. Biomed Pharmacother. 2020;129:109904. [68] ZHOU F, ZHENG Z, ZHA Z, et al. Nuclear paraspeckle assembly transcript 1 enhances hydrogen peroxide-induced human vascular smooth muscle cell injury by regulating miR-30d-5p/A disintegrin and metalloprotease 10. Circ J. 2022;86(6):1007-1018. [69] GOLLEDGE J. Abdominal aortic aneurysm: update on pathogenesis and medical treatments. Nat Rev Cardiol. 2019;16(4):225-242. [70] LEMAIRE SA, WANG X, WILKS JA, et al. Matrix metalloproteinases in ascending aortic aneurysms: bicuspid versus trileaflet aortic valves. J Surg Res. 2005;123(1): 40-48. [71] GURUNG R, CHOONG AM, WOO CC, et al. Genetic and epigenetic mechanisms underlying vascular smooth muscle cell phenotypic modulation in abdominal aortic aneurysm. Int J Mol Sci. 2020;21(17):6334. |
| [1] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [2] | Zhang Ming, Wang Bin, Jia Fan, Chen Jie, Tang Wei. Application of brain-computer interface technology based on electroencephalogram in upper limb motor function rehabilitation of stroke patients [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 581-586. |
| [3] | Ma Sicong, Chen Jing, Li Yunqing. Functions and roles of connective tissue growth factor in nervous systems [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 615-620. |
| [4] | Yan Binghan, Li Zhichao, Su Hui, Xue Haipeng, Xu Zhanwang, Tan Guoqing. Mechanisms of traditional Chinese medicine monomers in the treatment of osteoarthritis by targeting autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 627-632. |
| [5] | Wang Xinyi, Xie Xianrui, Chen Yujie, Wang Xiaoyu, Xu Xiaoqing, Shen Yihong, Mo Xiumei. Electrospun nanofiber scaffolds for soft and hard tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 426-432. |
| [6] | Long Jundong, Shi Yehong, Wang Cheng, Chen Shijiu. Effects of different freezing techniques on the rejection of allogeneic vascular transplantation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 433-438. |
| [7] | Yang Jie, Hu Haolei, Li Shuo, Yue Wei, Xu Tao, Li Yi. Application of bio-inks for 3D printing in tissue repair and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 445-451. |
| [8] | Qi Ming, Wang Lei, Zhang Zhen. CXCL5 participates in carotid plaque formation by inducing vascular calcification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 186-192. |
| [9] | Dai Xinyu, Yan Jihong, Hua Lingjun, Zheng Xiaohong. Resistance exercise improves body composition in overweight and obese people: an umbrella review [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 267-271. |
| [10] | Meng Zhicheng, Qiao Weiping, Zhao Yang, Liu Hongfei, Li Kaijie, Ma Bo. Effects of immune cells and related cytokines in the pathogenesis and treatment of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 280-287. |
| [11] | Long Yi, Yang Jiaming, Ye Hua, Zhong Yanbiao, Wang Maoyuan. Extracellular vesicles in sarcopenic obesity: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 315-320. |
| [12] | Chang Wanpeng, Zhang Zhongwen, Yang Yulin, Zi Yang, Yang Mengqi, Du Bingyu, Wang Nan, Yu Shaohong. Efficacy of rehabilitation exoskeleton robots on post-stroke lower limb motor dysfunction: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 321-328. |
| [13] | Long Qingxi, Zhang Pingshu, Liu Qing, Ou Ya, Zhang Lili, Yuan Xiaodong. Single-cell RNA sequencing reveals the heterogeneity of astrocytes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 139-146. |
| [14] | Yang Pei. Effects of different exercise types on human DNA damage, DNA methylation and telomere length [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 147-152. |
| [15] | Wang Xianfeng, Wang Kun, Sun Han, Sun Xiaoliang, Yan Litao. Mechanism underlying exosomal lncRNA H19 derived from umbilical cord mesenchymal stem cells promotes cartilage injury repair [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 20-25. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||