Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (3): 433-438.doi: 10.12307/2024.241
Previous Articles Next Articles
Effects of different freezing techniques on the rejection of allogeneic vascular transplantation
Long Jundong, Shi Yehong, Wang Cheng, Chen Shijiu
- Fifth Affiliated (Zhuhai) Hospital of Zunyi Medical University, Zhuhai 519100, Guangdong Province, China
-
Received:2023-02-09Accepted:2023-03-29Online:2024-01-28Published:2023-07-10 -
Contact:Chen Shijiu, Chief physician, Fifth Affiliated (Zhuhai) Hospital of Zunyi Medical University, Zhuhai 519100, Guangdong Province, China -
About author:Long Jundong, Masher, Fifth Affiliated (Zhuhai) Hospital of Zunyi Medical University, Zhuhai 519100, Guangdong Province, China -
Supported by:National Natural Science Foundation of China, No. 81960824 (to WC)
CLC Number:
Cite this article
Long Jundong, Shi Yehong, Wang Cheng, Chen Shijiu. Effects of different freezing techniques on the rejection of allogeneic vascular transplantation[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 433-438.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
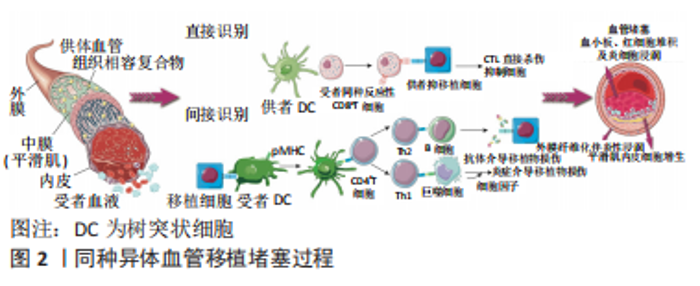
2.1 同种异体血管移植发生排斥反应的机制 同种异体血管移植后的免疫排斥反应主要是由受者T细胞识别供者组织相容性复合MHC-Ⅰ类抗原和MHC-Ⅱ类抗原引发的一系列炎症,最终导致血管堵塞的病理性过程。MHC-Ⅰ在哺乳动物新鲜血管的内皮细胞、平滑肌细胞中高度表达[6],其中内皮细胞被认为是血管移植中免疫刺激最强的成分,在移植排斥过程中起着至关重要的作用[7]。血管移植物内的树突状细胞、巨噬细胞等抗原提呈细胞传递、激活幼稚CD8细胞,使其发生分裂和分化为成熟CD8细胞,成熟CD8细胞到内皮细胞识别MHC-Ⅰ变为效应CD8细胞,同时召集炎症因子对其进行攻击,导致内皮细胞脱落及血管各层细胞坏死,形成累积血管全层的纤维素样坏死[8-9],受损后的血管移植物进行修复、增生,这个过程不断循环以及受者血小板、血细胞黏附于移植段血管等因素,最终导致移植段血管管腔狭窄、堵塞,过程如图2。"
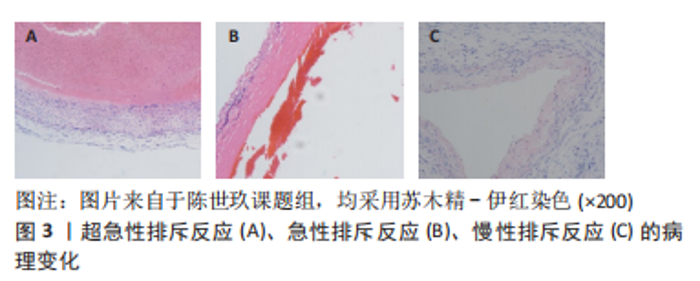
2.1.1 超急性排斥反应 是在同种异体血管移植后24 h内出现的坏死性血管炎表现。主要由于受者体循环中存在的抗体识别移植段血管内膜上残存的ABO血型抗原,发生抗体迅速攻击抗原的免疫效应从而引起血管内皮细胞受损释放凝血因子及血小板活化因子,这激活了内源性凝血系统;同时,抗原-抗体复合物的形成也会激活补体系统[10],这两个系统的激活会导致大量的血小板、炎症递质如C5a聚集于移植段血管;除此之外,供、受者的主要组织相容复合物不符也会加剧这两个系统的激活,例如,活化的单核细胞及嗜酸性粒细胞可以释放促炎因子及促凝因子,这最终导致血栓迅速生长在移植段血管管腔[11-12],病理如图3A。临床级的血管移植物在移植前会经过特定溶液冲净及准确的供、受者配型,故现代同种血管移植几乎不会出现此类反应。 2.1.2 急性排斥反应 包括T细胞介导的细胞免疫、抗体介导的体液免疫[13]。前者首先通过受者T细胞识别血管移植物细胞表面的特异性抗原启动排斥反应,然后T细胞进入激活阶段,自身活化增殖成效性T细胞,同时激活其他免疫细胞,如嗜中性粒细胞、树突状细胞,在这一过程中会释放大量的干扰素γ、肿瘤坏死因子α导致血管移植物内皮细胞损伤及实质细胞损伤[14];后者首先使促炎细胞如自然杀伤(NK)细胞、嗜中性粒细胞和巨噬细胞释放穿孔素、活性氧和肿瘤坏死因子α直接损伤内皮细胞导致血管内膜下层的基底膜裸露出来,从而引起凝血反应形成微小血栓,在血栓形成后,使得补体系统激活产生具有炎症趋化作用的C3a、C5a及攻击内皮细胞的C5b-9[11,15-16]。因此,这两种免疫的级联反应会导致血管移植物内皮细胞丢失、平滑肌细胞丢失、基质增生及血栓形成,如图3B,这是同种异体血管移植后最常见的一类排斥反应。 2.1.3 慢性排斥反应 虽对慢性排斥反应发生的原因众说纷纭,如受者发生感染后导致T细胞的二次介导、自身免疫性疾病的发作、免疫抑制剂的使用不当或剂量不足等,且目前尚无共识性机制,但可以肯定的是炎症浸润贯穿于同种异体血管移植慢性排斥反应全程[17]。在其发生早期的病理过程同急性排斥反应相似,但不同的是其病程明显长于急性排斥反应,病理学检查结果可见新生内膜形成、中层平滑肌结构明显紊乱及大量细胞外基质形成[18]。随着病变的发展,由于长时间的炎症浸润导致移植段血管管壁增厚、管腔狭窄甚至堵塞[19],在临床上,组织病理学检查仍是其诊断的金标准,如图3C,这是影响血管移植物长期存活的主要原因。"
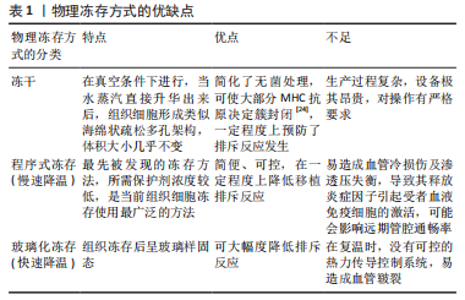
2.2 冻存处理对血管移植排斥反应的影响 目前针对血管移植物的现代冻存处理主要包括物理方法和化学试剂。在此研究中,物理方法包括冻干、程序式冻存及玻璃化冻存,化学方法主要涉及冻存保护剂的种类。其中程序式冻存慢速降温、玻璃化冻存及非渗透性保护剂是重要部分,将做重点阐述。 2.2.1 物理方法 冷冻能够长时间储存同种异体血管,同时也能降低排斥反应的发生,其机制有以下几点:①供者的血管移植物会被受者免疫系统识别为外来异物引发排斥反应,在冷冻过程中,血管移植物的细胞膜或细胞质等结构会受到一定损伤,导致其免疫原性下降,从而减少被受者免疫系统识别的概率[20]。②冻存会抑制血管移植物的新陈代谢,减少了细胞膜脂质的流动性,从而降低其细胞因子、炎症递质及活性氧等分子的释放[21],进一步减少了移植排斥反应的发生。③在同种异体血管移植中,内皮细胞的损伤是导致排斥反应发生的原因之一,而冻存在一定程度上保护了血管内皮细胞的完整性及功能[22],如冻存通过降低细胞钙离子内流来减少内皮细胞氧化应激的发生及代谢活性,从而减轻其损伤程度[23]。 由于液体有不同的物理状态,因此不同的冷冻方式对同种异体血管排斥反应的影响也不同,结合临床,各自优缺点如表1。"
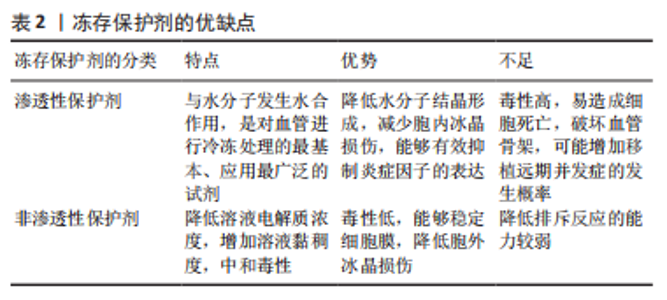
冻干:指冷冻干燥,又称升华干燥,是一种使细胞或组织脱水的物理过程。 OLMOS-Zú?IGA等[25]在对冻干犬肺动脉移植和低温冻存犬肺动脉移植的对照研究中发现,在术后4周冻干组肺动脉通畅率明显高于低温冻存组,减少了炎症细胞从平滑肌迁移到血管内膜和中膜的数量,降低了血管移植物产生无序胶原蛋白和内膜增生、钙化的发生。MORATO等[26]在同种异体骨植入试验中印证了冻干具有移植降低移植排斥反的作用。但限于设备昂贵,过程复杂,尚未普遍开展,临床中仍以低温冻存为主。 程序式冻存慢速降温:水作为血管的主要成分,在低温冻存中,降温速率的控制尤为关键。程序降温是同种异体血管传统的冷冻保存方法。 在基础研究中,HRUBY等[27]报道经程序式冻存后的同种异体大鼠腹主动脉在移植后,内皮及平滑肌所表达的MHC类分子表达受到抑制,CD4、CD8、IgG等免疫效应分子的浸润量降低。HIDI等[28]在研究中发现程序冻存对血小板相关抗原(GpIIb/IIIa受体)的形成有抑制作用,能减少血管壁纤维蛋白的沉积,降低血管移植物的血栓的形成。程序降温法使移植排斥反应下降的原因可能与冻存过程内皮细胞的丢失以及表面抗原被破坏、移植后平滑肌细胞的退化有关[29-30]。 与上述研究不同,一项35例接受冷冻保留异基因动脉搭桥术患者进行的前瞻性临床试验中发现,所有被研究的患者在干预1个月后都表现出抗MHC抗体的增加[30]。WEISS等[31]回顾了2000-2015年进行的同种异体髂主动脉冷冻保存原位移植的长期结果,其中2例患者在血管重建后30 d因排斥反应死亡,余31例中的13例患者在47个月因移植物相关并发症死亡,4例患者有移植物血栓形成,3例患者出现髂主动脉假性血管瘤。 最近的研究指出,复温过程也是一个对血管移植排斥反应有着重要影响但未被重视的方面,常规复温冻存血管移植物的方法是将其放入37 ℃的水浴快速升温[32],但过快复温速率及较高的温度会对血管移植物细胞在复温过程造成额外的损伤,在移植后灌注恢复时将产生大量的氧自由基,增强内皮细胞-白细胞的相互作用,引起严重的炎症导致移植排斥反应加重[33-34]。相反,HRUBY等[27]指出在解冻血管移植物时,先将其放入温度为+4 ℃的冰箱中约60 min,然后在室温下保存约30 min后进行缓慢解冻,血管移植物的MHC类分子表达量会显著下降,从而减轻移植排斥反应。 不难发现,基础实验与临床研究的结果尚有差异,作者推断其中与基础实验样本量低,术后追踪时间短及复温条件不同相关。 玻璃化冻存快速降温:是将血管移植物直接投入液氮,以极快的速度(15 000-30 000 ℃/ min)使细胞内的水分子没有足够时间形成冰晶而直接转变为黏稠不结晶的玻璃化状态[35]。大量研究采用了玻璃化冻存,其主要原因在于可能有助于进一步保护血管结构,降低排斥反应。 在基础研究中,魏民等[36]进行了程序式冻存、玻璃化冻存兔股动脉的同种异体移植对照研究,尽管术后股动脉内膜/中膜比值提示均发生排斥反应,但玻璃化组的血管阻塞率及淋巴细胞浸润数量明显低于程序式冻存组,这表示玻璃化冻存可以更进一步的降低血管移植排斥反应。SCHNEIDER等[37]表示玻璃化冻存也可减少血管移植物代谢水平及促炎因子、趋化因子的释放水平从而降低排斥反应。但受限于没有合适的热力传导控制系统,在复温时容易造成冻存组织皲裂[38],故未能在临床广泛开展。在临床研究中,玻璃化冻存降低免疫原性的作用在瓣膜、肌腱、精子等组织细胞中同样得到证实[39]。 用于玻璃化冻存的容器对排斥反应的改善也有一定作用。尽管塑料低温冻存管最常用,但它对排斥反应没有改善。GALBINSKI等[40]发现用不锈钢制成的封闭容器能激活热休克蛋白(HSP70),HSP70可升高抗炎因子白细胞介素10的表达水平及通过Toll样受体对Treg细胞进行正性调节来预防移植排斥反应[41-42]。XIAO等[43]对比了不锈钢和银封闭玻璃化容器,后者热传导速率比前者高,可使玻璃化过程更快,能够降低移植物细胞的冷损伤及细胞凋亡,从而减少排斥反应的发生。因此,在玻璃化冻存中通过改进冻存容器可以进一步降低排斥反应,但是目前体内研究相对不足,以上结果以基础研究和体外实验为主,尚不能为实际临床应用提供指导。 综上所述,在基础研究中已经证实玻璃化冻存较程序式冻存能进一步降低血管移植排斥反应,尽管前者现在在临床中的使用受限于复温条件,但这仍是未来血管冻存的优选方式之一。 2.2.2 化学冻存保护剂 从血管移植物的冻存到移植,其细胞会受到冷冻剂解冻的双重打击,容易造成细胞膜破裂甚至死亡,为了降低在冷冻处理对细胞的损伤,冻存保护剂在低温冻存中被广泛应用[44]。当前常用的冻存保护剂主要分为两大类:即渗透性保护剂与非渗透性保护剂[45]。冻存保护剂主要通过减少细胞在冻融过程中死亡、维持细胞内外水分平衡、减少血栓形成及炎症反应、修饰细胞膜表面分子等方面减轻排斥反应,对其研究结果和优缺点论述如表2。"
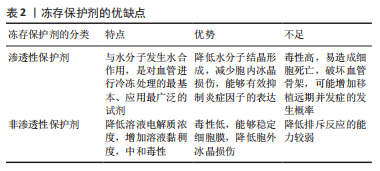

渗透性冻存保护剂:因其分子质量小,能够自由渗透到细胞内,又称为细胞内液保护剂,比如甘油、二甲基亚砜(DSMO)。它们可与水分子发生水合作用,具有减少胞内冰晶的形成、防止细胞内水分外渗、避免细胞在冷冻处理时过度皱缩而死亡的作用。 FAHNER等[46]用甘油作为保护剂冻存山羊颈动脉后进行同种异体移植,在术后3个月的随访中发现甘油组的血管外膜炎症反应弱于自体移植组,且两组管腔通畅率相近。GAO等[47]发现甘油缩水化后聚合而成的超支化聚甘油可以减少血管移植物中性粒细胞的浸润,延长移植物的存活时间。 VAN DOORMAAL等[48]展开了用生理盐水、甘油及DMSO作为保护剂储存兔主动脉的对比研究,结果显示DMSO组的主动脉壁厚度/直径比明显优于其他组。由高浓度的DMSO所配成冻存保护剂被称为“VS83”,H?GERLE等[49]用VS83对心血管移植物进行冷冻处理后,发现心血管移植物白细胞介素6、肿瘤坏死因子α的释放及促炎M1型巨噬细胞的增殖均被抑制,显著降低移植术后的炎症反应。同时,LIN等[50]首次证实DMSO有通过STAT5的表观遗传效应诱导幼稚T细胞向Treg细胞分化的作用,降低了移植物白细胞介素17α、白细胞介素23、干扰素γ等致炎因子的表达水平,这有助于减少炎症与坏死的发生。 非渗透性冻存保护剂:其作为大分子团,能溶于水但不能自由出入细胞膜,故又称为细胞外液保护剂,比如羟乙基淀粉、聚乙二醇、海藻糖、中药等物质,其具有低毒性的优点;同时也有增加细胞外液黏度、提高渗透压、使细胞内水分迅速向外析出、减少胞内水分从而降低冻存过程中胞内冰晶形成而导致细胞损伤的作用。 羟乙基淀粉能够吸收膜外水分子,增加胞外液体黏度来动态抑制冰晶形成[51];同时,羟乙基淀粉还可黏附在血管内膜上,在移植后通过降低血小板糖蛋白Ⅰb、Ⅱb及Ⅲa的表达,阻断纤维蛋白原与活化血小板的结合来抑制凝血和血小板的聚集从而减少血栓的形成[52]。聚乙二醇能稳定血管内皮细胞的血栓调节蛋白(TM),在低温环境下可以避免血栓调节蛋白4/5/6类结构域失活[53],减少凝血酶与炎蛋白的结合位点,从而降低血管移植物炎症及血栓的发生[54]。 海藻糖可以激活细胞自噬、减轻细胞氧化应激、降低组织中白细胞介素6、COX-2、肿瘤坏死因子α、单核细胞趋化因子1等炎症因子的释放水平[55],减少炎症因子对树突状细胞抗原提呈作用的激活[56]。有研究者将纳米颗粒封装的海藻糖与VS83技术相结合对大鼠心脏瓣膜进行保存后发现,白细胞介素10分泌增多,肿瘤坏死因子α释放减少[56]。WANG等[57]通过加入NaOH调节pH值及环氧氯丙烷等化学修饰合成的海藻糖共聚物,既保留了海藻糖的优点又具有良好的抑冰性,减少冰损伤引发细胞坏死进一步减轻移植后的炎症,达到预防排斥反应发生的作用。 最近的研究表明中药提取物可以有效预防同种异体移植排斥反应。如雷公藤内酯在低温冻存时能够降低移血管植物MHC-Ⅰ类分子及共刺激分子的表达水平,削弱了受者树突状细胞的抗原提呈作用及效应T细胞的激活[58];山茱萸通过降低白细胞介素12、干扰素γ 的表达来减轻排斥反应[59];丹参酮和龟板可以提高移植物Treg细胞数的量来抑制T细胞的毒性作用[60];大黄素可诱导FoxP3、CD122等调节性T细胞来抑制同种异体移植排斥反应[61]。 除了上述几种渗透性冻存保护剂,目前还研究出抗冻蛋白类、含血清类的冻存保护剂。抗冻蛋白是生物界自然选择进化产生的一种防止生物体结冰而导致死亡的功能性蛋白,如林蛙、南北极鱼类、昆虫等体内含有大量抗冻蛋白,其具有冰洁合面和非冰结合面两种官能团,它通过非冰结合面上存在的带电荷侧链及疏水性侧链,使得非冰结合面上的界面水无,来抑制冰晶形成从而减少细胞在冻融过程中的死亡[62]。含血清类的冻存保护剂,如含胎牛血清的冻存保护剂中有大量的生长因子、免疫球蛋白,可以通过给冻存的血管移植物提供营养、调节细胞应激反应来维持细胞代谢,减少细胞死亡[63]。从理论上说,这两种冻存保护剂均可通过减少冻融期间血管移植物细胞死亡来降低移植后的排斥反应。但是,对于目前来说,本课题组不建议将这两种冻存保护剂纳入降低同种异体血管移植排斥反应冻存保护剂的范畴,具体原因如下:①由于当前冻存保护剂洗脱技术仍存在一定弊端,不能够及时、全面地洗脱血管移植物上黏附的冻存保护剂;②这两种冻存保护剂是除了供、受者之外的第三类异物蛋白,容易引起受者免疫系统攻击引发炎症导致移植后排斥反应的加重。 无论是哪种类型的冻存保护剂都是为了减少血管移植物在冻融中的损伤,KURO等[64]开发了一种电压施加装置,即在冻存血管移植物时,通过予冻存保护剂1 000 V的电压能够使内皮细胞的损伤进一步降低,改善移植后结果。冻存保护剂都具有一定的细胞毒性,为了解决这一问题,NAGAYA等[65]报道了一种“中空纤维法”,可使冻存保护剂的有效体积达到最小值,通过减少冻存保护剂对细胞的毒性,能够降低移植后的炎症反应。以上方法仅分别在基础研究中的鼠股动脉移植及胰岛冻存中实现,在人类血管冻存中是否有同样效果有待进一步试验。 考虑以上因素,作者认为应联合使用渗透性及非渗透性冻存保护剂,但使用时应充分考量其物理、化学、生物特性及两者的混合比例,以期既能降低细胞毒性又可抑制排斥反应。"

| [1] GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the global burden of disease Study 2019. Lancet. 2020;96(10258):1204-1222. [2] 饶蓁蓁,傅晏红,李若曈,等.2030年我国常见危险因素所致心脑血管疾病死亡和早死概率预测研究[J]. 中华预防医学杂志,2022, 56(5):567-573. [3] JOUDA H, LARREA MURILLO L, WANG T. Current Progress in Vascular Engineering and Its Clinical Applications. Cells. 2022;11(3):493. [4] L’HEUREUX N, DUSSERRE N, KONIG G, et al. Human tissue-engineered blood vessels for adult arterial revascularization. Nat Med. 2006;12(3):361-365. [5] LIU S, ZHI J, LI S, et al. Progress on Precise Regulation of Vascular Intimal Repair by a Surface Coating of Vascular Stent. Curr Drug Deliv. 2021;18(7):862-873. [6] WILCOX EC, EDELMAN ER. Substratum interactions determine immune response to allogeneic transplants of endothelial cells. Front Immunol. 2022;13:946794. [7] PEELEN DM, HOOGDUIJN MJ, HESSELINK DA, et al. Advanced in vitro Research Models to Study the Role of Endothelial Cells in Solid Organ Transplantation. Front Immunol. 2021;12:607953. [8] MORIS D, CENDALES LC. Sensitization and Desensitization in Vascularized Composite Allotransplantation. Front Immunol. 2021;12: 682180. [9] MO F, WATANABE N, MCKENNA MK, et al. Engineered off-the-shelf therapeutic T cells resist host immune rejection. Nat Biotechnol. 2021; 39(1):56-63. [10] MINNELLI C, RIAZY M, OHASHI R, et al. Early Transplant Arteriopathy in Kidney Transplantation. Transplant Proc. 2021;53(5):1554-1561. [11] STITES E, RENNER B, LASKOWKI J, et al. Complement fragments are biomarkers of antibody-mediated endothelial injury. Mol Immunol. 2020;118:142-152. [12] BENTALL A, JEVAKANTHAN M, BRAITCH M, et al. Characterization of ABH-subtype donor-specific antibodies in ABO-A-incompatible kidney transplantation. Am J Transplant. 2021;21(11):3649-3662. [13] BESTARD O, GRINYÓ J. Refinement of humoral rejection effector mechanisms to identify specific pathogenic histological lesions with different graft outcomes. Am J Transplant. 2019;19(3):952-953. [14] KUMMER L, ZARADZKI M, VIJAYAN V, et al. Vascular Signaling in Allogenic Solid Organ Transplantation - The Role of Endothelial Cells. Front Physiol. 2020;11:443. [15] VALLAKATI A, REDDY S, DUNLAP ME, et al. Impact of Statin Use After Heart Transplantation: A Meta-Analysis. Circ Heart Fail. 2016;9(10): e003265. [16] CUMPELIK A, HEEGER PS. Effects of the complement system on antibody formation and function: implications for transplantation. Curr Opin Organ Transplant. 2022;27(5):399-404. [17] ENNS W, VON ROSSUM A, CHOY J. Mouse model of alloimmune-induced vascular rejection and transplant arteriosclerosis. J Vis Exp. 2015;(99):e52800. [18] KAUFMAN CL, KANITAKIS J, WEISSENBACHER A, et al. Defining chronic rejection in vascularized composite allotransplantation-The American Society of Reconstructive Transplantation and International Society of Vascularized Composite Allotransplantation chronic rejection working group: 2018 American Society of Reconstructive Transplantation meeting report and white paper Research goals in defining chronic rejection in vascularized composite allotransplantation. SAGE Open Med. 2020;8:2050312120940421. [19] JUSTIZ VAILLANT AA, MOHSENI M. Chronic Transplantation Rejection//StatPearls. Treasure Island (FL): StatPearls Publishing; 2022. [20] SONG G, WANG S, BARKESTANI MN, et al. Membrane attack complexes, endothelial cell activation, and direct allorecognition. Front Immunol. 2022;13:1020889. [21] LEN JS, KOH WSD, TAN SX. The roles of reactive oxygen species and antioxidants in cryopreservation. Biosci Rep. 2019;39(8):BSR20191601. [22] PARIHAR A, KUMAR A, PANDA U, et al. Cryopreservation: A Comprehensive Overview, Challenges, and Future Perspectives. Adv Biol (Weinh). 2023;e2200285. doi: 10.1002/adbi.202200285. [23] REN L, WANG MR, WANG QC. ROS-induced oxidative stress in plant cryopreservation: occurrence and alleviation. Planta. 2021;254(6):124. [24] MOORE TM, GENDLER E, GENDLER E. Viruses adsorbed on musculoskeletal allografts are inactivated by terminal ethylene oxide disinfection. J Orthop Res. 2004;22(6):1358-1361. [25] OLMOS-ZÚÑIGA JR, JASSO-VICTORIA R, DÍAZ-MARTÍNEZ NE, et al. Lyophilized allografts without pre-treatment with glutaraldehyde are more suitable than cryopreserved allografts for pulmonary artery reconstruction. Braz J Med Biol Res. 2016;49(2):e5001. [26] MORATO GO, ROCHA A G, CHUNG DG, et al. Lyophilized and gamma-sterilized allogeneic bone implant used as a spacer for advancement of a modified tibial tuberosity in the treatment of cranial cruciate ligament disease in dogs. PLoS One. 2019;14(8):e0220291. [27] HRUBY J, SPUNDA R, MERICKA P, et al. Influence of the new standardized clinical cryopreservation/slow thawing protocol on immunogenicity of arterial allografts in rats. PLoS One. 2020;15(3): e0230234. [28] HIDI L, KOMOROWICZ E, KOVÁCS GI, et al. Cryopreservation moderates the thrombogenicity of arterial allografts during storage. PLoS One. 2021;16(7):e0255114. [29] COTI I, WENDA S, ANDREEVA A, et al. Donor-specific HLA antibodies after fresh decellularized vs cryopreserved native allograft implantation. HLA. 2020;96(5):580-588. [30] GONZÁLEZ-GAY M, LÓPEZ-MARTÍNEZ R, BUSTO-SUÁREZ S, et al. Immunological Aspects Involved in the Degeneration of Cryopreserved Arterial Allografts. Front Surg. 2020;7:616654. [31] WEISS S, BACHOFEN B, WIDMER MK, et al. Long-term results of cryopreserved allografts in aortoiliac graft infections. J Vasc Surg. 2021; 74(1):268-275. [32] ANTONOPOULOS CN, PAPAKONSTANTINOU NA, HARDY D, et al. Editor’s Choice - Cryopreserved Allografts for Arterial Reconstruction after Aorto-Iliac Infection: A Systematic Review and Meta-Analysis. Eur J Vasc Endovasc Surg. 2019;58(1):120-128. [33] MINOR T, VON HORN C. Rewarming Injury after Cold Preservation. Int J Mol Sci. 2019;20(9):2059. [34] BOGERT NV, WERNER I, KORNBERGER A, et al. Effect of Rewarming on Leukocyte-Endothelial Interaction After Deep Hypothermic Preservation. Ann Transplant. 2020;25:e919540. [35] FINGER EB, BISCHOF JC. Cryopreservation by vitrification: a promising approach for transplant organ banking. Curr Opin Organ Transplant. 2018;23(3):353-360. [36] 魏民,张伯勋,刘郑生,等.玻璃化法保存异体动脉的实验研究[J].中国修复重建外科杂志,2005,19(4):251-254. [37] SCHNEIDER M, STAMM C, BROCKBANK KGM, et al. The choice of cryopreservation method affects immune compatibility of human cardiovascular matrices. Sci Rep. 2017;7(1):17027. [38] GANGWAR L, PHATAK SS, ETHERIDGE M, et al. A guide to successful mL to L scale vitrification and rewarming. Cryo Letters. 2022;43(6):316-321. [39] WANG M, TODOROV P, WANG W, et al. Cryoprotectants-Free Vitrification and Conventional Freezing of Human Spermatozoa: A Comparative Transcript Profiling. Int J Mol Sci. 2022;23(6):3047. [40] GALBINSKI S, KOWALEWSKI LS, GRIGOLO GB, et al. Comparison between two cryopreservation techniques of human ovarian cortex: morphological aspects and the heat shock response (HSR). Cell Stress Chaperones. 2021;27(2):97-106. [41] BORGES TJ, LANG BJ, LOPES RL, et al. Modulation of Alloimmunity by Heat Shock Proteins. Front Immunol. 2016;7:303. [42] GUO P, ZHANG H, LI C, et al. Research progress on Toll-like receptors pathways regulating function of regulatory T cells. Sheng Wu Gong Cheng Xue Bao. 2020;36(9):1701-1712. [43] XIAO Z, ZHANG Y, FAN W. Cryopreservation of human ovarian tissue using the silver closed vitrification system. J Assist Reprod Genet. 2017;34(11):1435-1444. [44] LIN M, CAO H, MENG Q, et al. Insights into the crystallization and vitrification of cryopreserved cells. Cryobiology. 2022;106:13-23. [45] YONG KW, LAOUAR L, ELLIOTT JAW, et al. Review of non-permeating cryoprotectants as supplements for vitrification of mammalian tissues. Cryobiology. 2020;96:1-11. [46] FAHNER PJ, IDU MM, VAN GULIK TM, et al. Glycerol-preserved arterial allografts evaluated in the infrarenal rat aorta. Eur Surg Res. 2009;42(2):78-86. [47] GAO S, GUAN Q, CHAFEEVA I, et al. Hyperbranched polyglycerol as a colloid in cold organ preservation solutions. PLoS One. 2015;10(2): e0116595. [48] VAN DOORMAAL TPC, SLUIJS JH, VINK A, et al. Comparing five simple vascular storage protocols. J Surg Res. 2014;192(1):200-205. [49] HÖGERLE BA, SCHNEIDER M, SUDROW K, et al. Effects on human heart valve immunogenicity in vitro by high concentration cryoprotectant treatment. J Tissue Eng Regen Med. 2018;12(2):e1046-e1055. [50] LIN GJ, WU CH, YU CC, et al. Adoptive transfer of DMSO-induced regulatory T cells exhibits a similar preventive effect compared to an in vivo DMSO treatment for chemical-induced experimental encapsulating peritoneal sclerosis in mice. Toxicol Appl Pharmacol. 2019;378:114641. [51] KÖRBER C, SCHEIWE MW. The cryoprotective properties of hydroxyethyl starch investigated by means of differential thermal analysis. Cryobiology. 1980;17(1):54-65. [52] XU L, LI L, ZU J, et al. Effects of Different Types of Early Restrictive Fluid Resuscitation on Immune Function and Multiorgan Damage on Hemorrhagic Shock Rat Model in a Hypothermic Environment. Comput Math Methods Med. 2022;2022:4982047. [53] LIU X, BORON M, ZHAO Y, et al. End-point modification of recombinant thrombomodulin with enhanced stability and anticoagulant activity. Eur J Pharm Sci. 2019;139:105066. [54] WATANABE-KUSUNOKI K, NAKAZAWA D, ISHIZU A, et al. Thrombomodulin as a Physiological Modulator of Intravascular Injury. Front Immunol. 2020;11:575890. [55] LIU S, YANG Y, GAO H, et al. Trehalose attenuates renal ischemia-reperfusion injury by enhancing autophagy and inhibiting oxidative stress and inflammation. Am J Physiol Renal Physiol. 2020;318(4): F994-F1005. [56] AHMAD T, EAPEN MS, ISHAQ M, et al. Anti-Inflammatory Activity of Fucoidan Extracts In Vitro. Mar Drugs. 2021;19(12):702. [57] WANG J, SHI X, XIONG M, et al. Trehalose glycopolymers for cryopreservation of tissue-engineered constructs. Cryobiology. 2022; 104:47-55. [58] WANG Y, ZHANG S, LI Z, et al. The effects of triptolide on the cellular activity of cryopreserved rat sciatic nerves and nerve regeneration after allotransplantation. Int J Neurosci. 2020;130(1):83-96. [59] LU C, ZENG Y Q, LIU H, et al. Tanshinol suppresses cardiac allograft rejection in a murine model. J Heart Lung Transplant. 2017;36(2): 227-236. [60] LIU X, ZENG YQ, LIANG YZ, et al. Medicinal herbs Fructus corni and Semen cuscutae suppress allograft rejection via distinct immune mechanisms. Oncotarget. 2016;7(24):35680-35691. [61] QIU F, LIU H, LIANG CL, et al. Corrigendum: A New Immunosuppressive Molecule Emodin Induces both CD4+FoxP3+ and CD8+CD122+ Regulatory T Cells and Suppresses Murine Allograft Rejection. Front Immunol. 2020;11:1381. [62] LIU K, WANG C, MA J, et al. Janus effect of antifreeze proteins on ice nucleation. Proc Natl Acad Sci U S A. 2016;113(51):14739-14744. [63] JAHANBAN-ESFAHLAN A, OSTADRAHIMI A, JAHANBAN-ESFAHLAN R, et al. Recent developments in the detection of bovine serum albumin. Int J Biol Macromol. 2019;138:602-617. [64] KURO A, MORIMOTO N, HARA T, et al. Protection of rat artery grafts from tissue damage by voltage-applied supercooling. Med Mol Morphol. 2022;55(2):91-99. [65] NAGAYA M, MATSUNARI H, KANAI T, et al. An Effective New Cryopreservation Procedure for Pancreatic Islets Using Hollow Fiber Vitrification. Horm Metab Res. 2016;48(8):540-549. [66] IWAKI R, SHOJI T, MATSUZAKI Y, et al. Current status of developing tissue engineering vascular technologies. Expert Opin Biol Ther. 2022; 22(3):433-440. [67] LEJAY A, VENTO V, KUNTZ S, et al. Current status on vascular substitutes. J Cardiovasc Surg (Torino). 2020;61(5):538-543. [68] MAZUR P. Kinetics of water loss from cells at subzero temperatures and the likelihood of intracellular freezing. J Gen Physiol. 1963;47(2): 347-369. |
| [1] | Wang Xinyi, Xie Xianrui, Chen Yujie, Wang Xiaoyu, Xu Xiaoqing, Shen Yihong, Mo Xiumei. Electrospun nanofiber scaffolds for soft and hard tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 426-432. |
| [2] | Yang Jie, Hu Haolei, Li Shuo, Yue Wei, Xu Tao, Li Yi. Application of bio-inks for 3D printing in tissue repair and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 445-451. |
| [3] | Dai Xinyu, Yan Jihong, Hua Lingjun, Zheng Xiaohong. Resistance exercise improves body composition in overweight and obese people: an umbrella review [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 267-271. |
| [4] | Meng Zhicheng, Qiao Weiping, Zhao Yang, Liu Hongfei, Li Kaijie, Ma Bo. Effects of immune cells and related cytokines in the pathogenesis and treatment of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 280-287. |
| [5] | Long Yi, Yang Jiaming, Ye Hua, Zhong Yanbiao, Wang Maoyuan. Extracellular vesicles in sarcopenic obesity: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 315-320. |
| [6] | Chang Wanpeng, Zhang Zhongwen, Yang Yulin, Zi Yang, Yang Mengqi, Du Bingyu, Wang Nan, Yu Shaohong. Efficacy of rehabilitation exoskeleton robots on post-stroke lower limb motor dysfunction: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 321-328. |
| [7] | Long Qingxi, Zhang Pingshu, Liu Qing, Ou Ya, Zhang Lili, Yuan Xiaodong. Single-cell RNA sequencing reveals the heterogeneity of astrocytes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 139-146. |
| [8] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [9] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [10] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [11] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [12] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [13] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [14] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [15] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||