Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (1): 147-152.doi: 10.12307/2023.906
Previous Articles Next Articles
Effects of different exercise types on human DNA damage, DNA methylation and telomere length
Yang Pei
- College of Physical Education, Guangxi University of Science and Technology, Liuzhou 545006, Guangxi Zhuang Autonomous Region, China
-
Received:2022-12-12Accepted:2023-02-11Online:2024-01-08Published:2023-06-29 -
Contact:Yang Pei, College of Physical Education, Guangxi University of Science and Technology, Liuzhou 545006, Guangxi Zhuang Autonomous Region, China -
About author:Yang Pei, Master, Lecturer, College of Physical Education, Guangxi University of Science and Technology, Liuzhou 545006, Guangxi Zhuang Autonomous Region, China -
Supported by:the Guangxi Philosophy and Social Science Planning Research Project, No. 21FTY011 (to YP)
CLC Number:
Cite this article
Yang Pei. Effects of different exercise types on human DNA damage, DNA methylation and telomere length[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 147-152.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 不同运动方式对DNA损伤、DNA甲基化和端粒长度的影响 2.1.1 运动方式的简述 此文共包含4种运动方式:急性运动、长期有氧运动、长期无氧运动和长期抗阻运动。其中有氧运动是一种以有氧代谢为主要供能方式的运动方式,而无氧运动则以无氧代谢为主要供能方式;抗阻训练指的是肌肉在对抗外部阻力时进行的主动运动,通常有利于增加肌肉力量、张力、质量和耐力;急性运动也叫一次性运动,可在有氧、无氧和抗阻条件下进行。 2.1.2 急性运动对DNA损伤、DNA甲基化和端粒长度的影响 (1) DNA损伤:国内外学者对急性运动与DNA损伤之间的关系进行了广泛的研究。总的来说,急性运动会造成DNA损伤,其原因与氧化应激水平增高有关[16-17]。氧化应激引起的 DNA 损伤常被认为是对健康不利的因素,而BELVIRANLI等[18]和REICHHOLD等[19]认为急性运动导致氧化应激是正常生理反应,并不会给身体健康带来长期不良后果。TRYFIDOU等[20]也指出急性有氧运动导致的DNA损伤不应该被视为有害结果。DNA损伤仅在急性有氧运动后1 d才显著,并且在运动后5-28 d内不会增加。此外,MORENO-VILLANUEVA等[21]报道急性运动可以保护受试者免疫细胞免受辐射诱导后DNA链断裂。综上,急性运动虽然会造成短暂的DNA损伤,但不会对身体造成长期危害。 (2) DNA甲基化:急性运动会促进骨骼肌的结构和代谢适 应,从而使机体在健康方面获益[22]。急性运动通常会诱导骨骼肌基因启动子区域的DNA去甲基化,有研究对健康久坐的男性和女性进行了急性运动干预并获取了骨骼肌活检样本[14],结果发现急性运动诱导过氧化物酶体增殖物激活受体γ共激活因子1α (peroxisome proliferator-activated receptor-gamma co-activator-1alpha,PGC-1α)等局部基因的mRNA表达显著增高,且全基因组甲基化水平降低;甲基化水平的降低程度受运动强度的影响,其中以高强度最为显著。FABRE等[23]研究了急性运动后人体脂肪组织的转录和表观遗传反应,结果显示急性运动降低了白色脂肪组织DNA甲基化程度。 (3)端粒长度:不同的急性运动模式对端粒长度的影响有不同的结果。WERNER等[24]通过交叉研究的方式探索了急性有氧耐力与急性抗阻运动对端粒酶活性的影响,结果显示急性有氧耐力训练可增加白细胞的端粒酶活性;相反,急性抗阻训练后没有观察到端粒酶活性的变化。此外,SIMPSON等[25]和LUDLOW等[26]也发现急性有氧运动介导的早期适应性反应可通过上调端粒保护基因进而维持端粒长度。然而,MASTALOUDIS等[27]和BORGHINI等[28]研究有相反的结论,即暴露于急性耐力运动会增加氧自由基的生成和氧化应激反应,进而导致端粒长度缩短。以上研究表明急性运动在调节端粒长度方面存在争议。 综上,急性运动可以降低DNA甲基化并增加DNA损伤,然而对于急性运动调节端粒长度方面仍存在争议。因此,需要进一步探索急性运动(有氧运动和抗阻训练)对端粒长度、端粒酶活性和其他端粒调节分子(如端粒蛋白复合物和miRNA)的影响,以确定该运动方式对端粒长度和端粒酶活性是否具有积极意义。 2.1.3 长期有氧运动对DNA损伤、DNA甲基化和端粒长度的影响 (1) DNA损伤:共有3项研究报告了长期有氧运动对DNA损伤的影响[29-31],其中2项研究指出长期有氧运动有利于提高抗氧化能力,减少DNA损伤[29-30];而RAHNAMA等[31]表明8周的有氧运动未能改善大学生的氧化应激水平。通过比较上述研究发现,RAHNAMA的研究仅开展了8周的运动干预,而其他2项研究的干预时间则在12-24周,故干预时间的长短可能影响了运动干预效果。另外,RAHNAMA在其研究中纳入的受试者为健康大学生,而其余2项研究的受试者为肠易激综合征患者和老年人。既往研究指出肠易激综合征患者和老年人自身的氧化应激水平较高,故运动可能更容易发挥其抗氧化的能力[32-33]。综上,长期有氧运动在改善DNA损伤方面具有积极意义,但干预效果易受人群和运动持续时间因素的影响。 (2) DNA甲基化:ROWLANDS等[34]研究发现16周的有氧训练可以降低2型糖尿病患者葡萄糖转运蛋白4基因启动子区的甲基化程度。然而BOYNE等[35]有相反的结果,发现48周的有氧训练未能降低乳腺癌患者多个基因启动子区域的甲基化程度。考虑到纳入研究数量较少,故仍需更多研究进一步探索有氧运动对于DNA甲基化的影响。 (3)端粒长度:一项来自丹麦的横断面研究比较了女性精英足球运动员和同龄普通女性端粒长度的差异,结果发现女性精英足球运动员淋巴细胞的端粒长度比同龄普通女性长22%-24%[36]。LAROCCA等[37]和DENHAM等[11]测量了长期接受有氧耐力运动人群的端粒长度,结果显示与健康非运动人群相比,运动人群组拥有更长的端粒。然而,有3项研究指出24-48周的有氧运动干预未能改变端粒长度和端粒酶活 性[38-40]。以上文献提示长期有氧运动与端粒长度之间的关联存在一定争议。通过比较可发现前3项研究的运动持续时间在2-14年,而后3项研究则持续24-48个月,故运动持续时间可能是导致争议的主要影响因素。SCHELLNEGGER等[41]在其综述中指出运动与端粒长度存在一定剂量关系,较短的运动持续时间无法引起端粒长度的长期变化。因此,更长的运动持续时间可能是有氧运动提高端粒长度的重要方法。 综上,长期有氧运动对改善DNA损伤和端粒长度具有积极意义,但运动干预效果受人群和运动持续时间影响。另外,值得一提的是,基于当前文献可知有氧运动持续2年以上可以增加端粒长度,见表1,未来的研究也应进一步明确最佳的运动持续时间。"


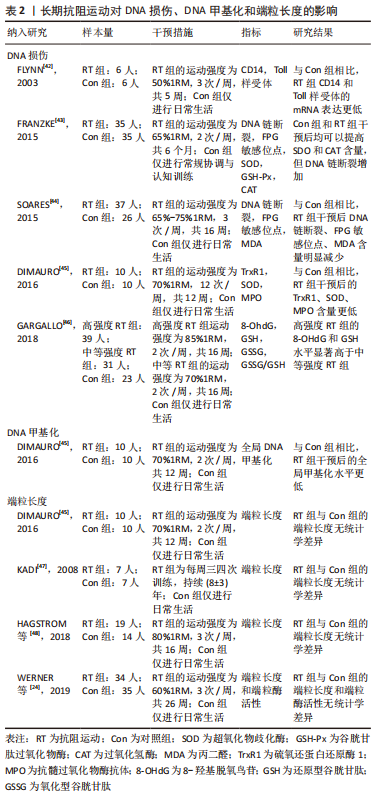
2.1.4 长期抗阻训练对DNA损伤、DNA甲基化和端粒长度的影响 (1) DNA损伤:4项研究探索了长期抗阻运动对DNA损伤的影响[42-45],结果均显示抗阻运动可以提高机体的抗氧化应激能力,进而减少DNA损伤。通过分析运动参数可发现纳入研究均采用了中等运动强度,即50%-75%1RM;这也提示该强度的抗阻运动会对DNA损伤产生有益影响。GARGALLO 等[46]比较了不同强度的抗阻训练(中等强度:70% 1RM;高强度:85%1RM)对久坐妇女DNA损伤的影响,结果显示高强度抗阻训练组的氧化应激水平显著高于中等强度组。该文献表明高强度的抗阻训练会导致氧化应激增加,而适度训练可以减少 DNA 损伤,进而支持了中等强度的有效性。另外,纳入研究的其他运动参数存在一定差异,其中运动频率的范围是2-12次/周,运动持续时间是5-24周不等,因此仍需更多研究进一步探索最佳的运动参数水平。 (2) DNA甲基化:DIMAURO等[45]研究了长期抗阻训练对DNA甲基化的影响,结果显示抗阻训练可以显著降低全局DNA甲基化水平。 (3)端粒长度:从表2可得,4项研究均证实长期的抗阻训练均未能增加端粒长度和端粒酶活性,这提示单独的抗阻训练 可能不足以引起端粒长度的任何变化[24,45,47-48]。"
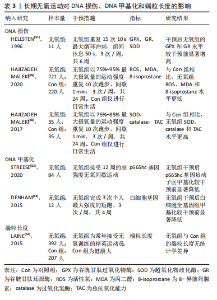
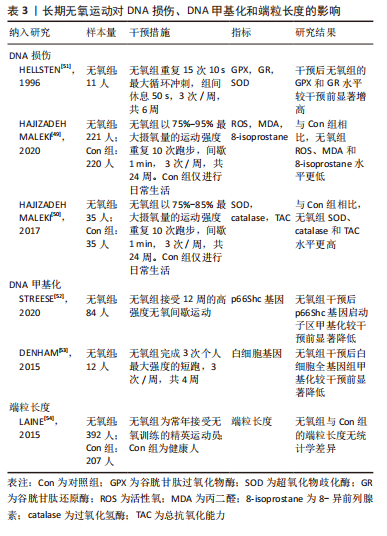
综上,基于当前证据可得长期抗阻训练可以改善DNA损伤并降低DNA甲基化程度,见表2,然而未能增加端粒长度和端粒酶活性。 2.1.5 长期无氧训练对DNA损伤、DNA甲基化和端粒长度的影响 (1) DNA损伤:3项研究描述了长期无氧运动对降低DNA损伤具有积极意义[49-51]。通过分析运动参数可发现除运动持续时间外,以上研究其余的运动参数具有较高的一致性。其中运动强度为75%-95%最大摄氧量,间歇时间在50-60 s之间,运动频率均为3次/周,这些共同的参数可作为制定运动处方的重要依据。另外,对于运动持续时间而言,应通过进一步的实验确定最佳持续时间。 (2) DNA甲基化:STREESE等[52]一项针对久坐人群运动的研究显示,高强度无氧间歇运动可以显著降低p66Shc 基因启动子区甲基化。DENHAM等[53]对12名健康男性开展了4周的高强度短跑干预,结果发现干预后白细胞全基因组甲基化较干预前显著降低。以上结果提示长期无氧运动对于改善DNA甲基化具有积极效果。 (3)端粒长度:LAINE等[54]调查了精英级运动员和健康人的白细胞端粒长度的差别,运动项目以无氧运动为主,包括足球、冰球、篮球等。有趣的是作者发现所有运动组和对照组之间没有显著差异,提示长期无氧运动对端粒长度可能没有改善作用。 综上,长期无氧运动在降低DNA损伤和甲基化方面具有积极作用,见表3。"
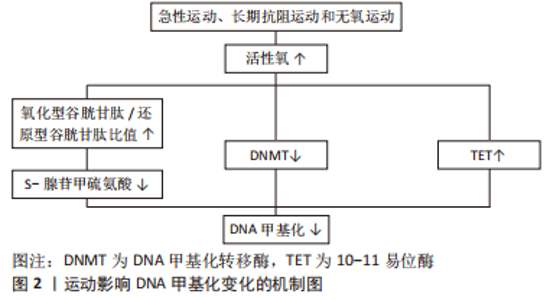
2.2 运动影响DNA损伤、DNA甲基化和端粒长度变化的可能机制 2.2.1 运动影响DNA损伤变化的可能机制 由表1-3总结可得,长期有氧、抗阻和无氧运动均能改善氧化应激状态,减少DNA损伤。有文献指出运动改善氧化应激的机制为抗氧化能力提高,即表现为抗氧化标志物(如超氧化物歧化酶)水平提高,过氧化标志物(如丙二醛)水平降低[55]。另外,一些研究也比较了不同运动方式对氧化应激的调控差异。孙波等[56]比较了高强度无氧间歇运动和有氧运动对2型糖尿病大鼠肾脏氧化应激的影响,结果显示高强度间歇运动更有效。田鹏等[57]研究显示相较于抗阻运动,有氧运动在缓解氧化应激方面的效果更明显。这提示高强度无氧间歇运动可能是改善DNA损伤的最佳运动方式。另外,研究结果还显示急性运动会增加DNA损伤。FISHER-WELL MAN等[58]在其综述中指出,急性有氧运动和无氧运动都会增加活性氧和活性氮氧化物的产生,从而导致人体和动物模型中的氧化应激。 2.2.2 运动影响DNA甲基化变化的可能机制 由表1-3可知,急性运动、长期抗阻运动和无氧运动在降低DNA甲基化方面具有积极作用,究其原因可能与活性氧有关。活性氧水平适当的增长有利于诱导DNA去甲基化[59]。既往研究显示急性运动、长期抗阻运动和无氧运动会促进活性氧增加,导致还原型谷胱甘肽下降,从而改变氧化型谷胱甘肽/还原型谷胱甘肽比值,最终减少S-腺苷甲硫氨酸(SAM)合成并下调DNA甲基化水平[60-62]。STEPHENS等[63]也证实10周的有氧运动可以下调与线粒体功能和还原型谷胱甘肽代谢相关的基因启动子甲基化水平。上述研究表明,运动诱导的活性氧可能通过改变氧化型谷胱甘肽/还原型谷胱甘肽比值影响线粒体DNA去甲基化。另外,考虑到活性氧对DNA甲基化转移酶(DNA methylation transferase,DNMT)的蛋白表达具有调节作用,而DNA甲基化主要依赖于DNMT的催化和维持,因此运动诱导的活性氧也可能通过下调 DNMT 的表达进而改善DNA甲基化。还值得注意的是,LI等[64]研究表明运动诱导的活性氧也可能通过增加10-11易位酶(ten-eleven translocation,TET)蛋白活性,从而下调线粒体相关基因的DNA甲基化水平。基于以上证据,运动可能通过诱导的活性氧介导氧化型谷胱甘肽/还原型谷胱甘肽比值和DNMT和TET的表达,进而对DNA甲基化产生调控作用,见图2。"

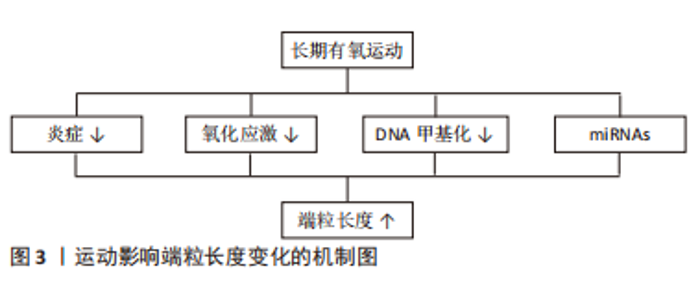
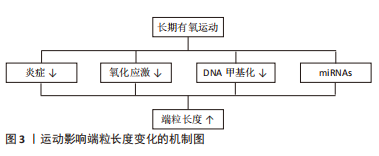
2.2.3 运动影响端粒长度变化的可能机制 由前文可知,仅有长期有氧运动在改善端粒长度方面具有积极效果,其原因可能与端粒动力学改变有关。运动方式是影响端粒动力学的重要因素,既往研究指出相较于抗阻运动等运动方式,有氧运动与更高的平均心率和最大心率相关,从而引起更高的血管剪切力。高血管剪切力会导致血管壁释放更高水平的一氧化氮,使端粒动力学发生改变,进而促进端粒长度的增加[65]。 在生物学机制方面,炎症和氧化应激机制可能是有氧运动改变端粒长度的重要途径,见图3。首先在炎症方面,以往文献表明端粒缩短与白细胞介素6、肿瘤坏死因子α等促炎因子过度增高有关[66],而有氧运动可以有效减少炎症因子水平,故推测有氧运动可能通过抑制炎症进而维持端粒长度;其次在氧化应激方面,自由基引起的氧化应激水平升高被认为是端粒磨损和衰老的重要原因[67]。李俊等[68]和田鹏等[57]研究显示有氧运动可以通过上调总抗氧化能力和超氧化物歧化酶活性,进而使氧化应激水平降低。因此推测有氧运动也可以通过调节氧化应激进而达到增加端粒长度的目的。另外,端粒长度也受DNA甲基化和微小RNA(miRNAs)等表观遗传因素影响,故该因素也可能是有氧运动改变端粒长度的潜在途径。刘波等[69]研究指出有氧运动通过下调DNMT的活性可降低甲基化程度,进而导致同型半胱氨酸生成减少,最终使端粒酶活性得以提高。在miRNAs方面,CHILTON等[70]表明有氧运动可通过下调miR-155的表达并提高端粒重复因子1的翻译效率,进而延缓端粒缩短速度。综上可得,表观遗传在介导运动调节端粒长度中发挥重要作用。"
| [1] AHMAD S, HERACLIDES A, SUN Q, et al. Telomere length in blood and skeletal muscle in relation to measures of glycaemia and insulinaemia. Diabet Med. 2012;29(10):e377-e381. [2] MAYNARD S, KEIJZERS G, HANSEN AM, et al. Associations of subjective vitality with DNA damage, cardiovascular risk factors and physical performance. Acta Physiol (Oxf). 2015;213(1):156-170. [3] 陈璐,司佳卉,孙点剑一,等.中国成年人生活方式和心血管代谢因素与甲基化年龄加速的相关性分析[J].中华流行病学杂志,2022,43(7):1019-1029. [4] ZSURKA G, PEEVA V, KOTLYAR A, et al. Is There Still Any Role for Oxidative Stress in Mitochondrial DNA-Dependent Aging. Genes (Basel). 2018;9(4):175. [5] HUANG E, CHENG SH, DRESSMAN H, et al. Gene expression predictors of breast cancer outcomes. Lancet. 2003;361(9369):1590-1596. [6] HANNUM G, GUINNEY J, ZHAO L, et al. Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol Cell. 2013;49(2):359-367. [7] 胡晓磐,李世昌,孙朋.运动对骨质影响的表观遗传机制研究进展[J].体育科学,2020,40(4):59-66. [8] BARRÓN-CABRERA E, RAMOS-LOPEZ O, GONZÁLEZ-BECERRA K, et al. Epigenetic Modifications as Outcomes of Exercise Interventions Related to Specific Metabolic Alterations: A Systematic Review. Lifestyle Genom. 2019;12(1-6):25-44. [9] 杨园,叶啟发,杨翼.运动调节衰老的表观遗传机制研究进展[J].中国运动医学杂志,2019,38(8):712-716. [10] DENHAM J, O’BRIEN BJ, PRESTES PR, et al. Increased expression of telomere-regulating genes in endurance athletes with long leukocyte telomeres. J Appl Physiol (1985). 2016;120(2):148-158. [11] DENHAM J, NELSON CP, O’BRIEN BJ, et al. Longer leukocyte telomeres are associated with ultra-endurance exercise independent of cardiovascular risk factors. PLoS One. 2013;8(7):e69377. [12] CASSIDY S, THOMA C, HOUGHTON D, et al. High-intensity interval training: a review of its impact on glucose control and cardiometabolic health. Diabetologia. 2017;60(1):7-23. [13] DENHAM J, MARQUES FZ, BRUNS EL, et al. Epigenetic changes in leukocytes after 8 weeks of resistance exercise training. Eur J Appl Physiol. 2016;116(6):1245-1253. [14] BARRÈS R, YAN J, EGAN B, et al. Acute exercise remodels promoter methylation in human skeletal muscle. Cell Metab. 2012;15(3):405-411. [15] 刘辰东,杨露,蒲红州,等.运动对骨骼肌基因表达的表观遗传调控作用[J].遗传,2017,39(10):888-896. [16] ÇAKIR-ATABEK H, ÖZDEMIR F, ÇOLAK R. Oxidative stress and antioxidant responses to progressive resistance exercise intensity in trained and untrained males. Biol Sport. 2015;32(4):321-328. [17] 王冬冬.补充虾青素对人体急性大强度运动恢复期代谢影响的1H-NMR研究[D].太原:山西大学,2017. [18] BELVIRANLI M, GÖKBEL H. Acute exercise induced oxidative stress and antioxidant. Front Physiol. 2006;3(3):126-131. [19] REICHHOLD S, NEUBAUER O, EHRLICH V, et al. No acute and persistent DNA damage after an Ironman triathlon. Cancer Epidemiol Biomarkers Prev. 2008; 17(8):1913-1919. [20] TRYFIDOU DV, MCCLEAN C, NIKOLAIDIS MG, et al. DNA Damage Following Acute Aerobic Exercise: A Systematic Review and Meta-analysis. Sports Med. 2020; 50(1):103-127. [21] MORENO-VILLANUEVA M, KRAMER A, HAMMES T, et al. Influence of Acute Exercise on DNA Repair and PARP Activity before and after Irradiation in Lymphocytes from Trained and Untrained Individuals. Int J Mol Sci. 2019;20(12): 2999. [22] MCGEE SL, HARGREAVES M. Epigenetics and Exercise. Trends Endocrinol Metab. 2019;30(9):636-645. [23] FABRE O, INGERSLEV LR, GARDE C, et al. Exercise training alters the genomic response to acute exercise in human adipose tissue. Epigenomics. 2018;10(8): 1033-1050. [24] WERNER CM, HECKSTEDEN A, MORSCH A, et al. Differential effects of endurance, interval, and resistance training on telomerase activity and telomere length in a randomized, controlled study. Eur Heart J. 2019;40(1):34-46. [25] SIMPSON RJ, COSGROVE C, CHEE MM, et al. Senescent phenotypes and telomere lengths of peripheral blood T-cells mobilized by acute exercise in humans. Exerc Immunol Rev. 2010;16:40-55. [26] LUDLOW AT, GRATIDÃO L, LUDLOW LW, et al. Acute exercise activates p38 MAPK and increases the expression of telomere-protective genes in cardiac muscle. Exp Physiol. 2017;102(4):397-410. [27] MASTALOUDIS A, LEONARD SW, TRABER MG. Oxidative stress in athletes during extreme endurance exercise. Free Radic Biol Med. 2001;31(7):911-922. [28] BORGHINI A, GIARDINI G, TONACCI A, et al. Chronic and acute effects of endurance training on telomere length. Mutagenesis. 2015;30(5):711-716. [29] HAJIZADEH MALEKI B, TARTIBIAN B, MOOREN FC, et al. Low-to-moderate intensity aerobic exercise training modulates irritable bowel syndrome through antioxidative and inflammatory mechanisms in women: Results of a randomized controlled trial. Cytokine. 2018;102:18-25. [30] ALGHADIR AH, GABR SA, AL-EISA ES. Effects of Moderate Aerobic Exercise on Cognitive Abilities and Redox State Biomarkers in Older Adults. Oxid Med Cell Longev. 2016;2016:2545168. [31] RAHNAMA N, GAEINI AA, HAMEDINIA MR. Oxidative stress responses in physical education students during 8 weeks aerobic training. J Sports Med Phys Fitness. 2007;47(1):119-123. [32] 柏茜茜,韩承刚,徐莹,等.肠易激综合征饮食干预策略研究进展[J].食品工业科技,2022,43(16):421-431. [33] 李可欣,吕静,于冰,等.老年衰弱新兴标志物的研究进展及展望[J].中国全科医学,2021,24(36):4580-4586. [34] ROWLANDS DS, PAGE RA, SUKALA WR, et al. Multi-omic integrated networks connect DNA methylation and miRNA with skeletal muscle plasticity to chronic exercise in Type 2 diabetic obesity. Physiol Genomics. 2014;46(20):747-765. [35] BOYNE DJ, KING WD, BRENNER DR, et al. Aerobic exercise and DNA methylation in postmenopausal women: An ancillary analysis of the Alberta Physical Activity and Breast Cancer Prevention (ALPHA) Trial. PLoS One. 2018;13(6):e0198641. [36] HAGMAN M, FRISTRUP B, MICHELIN R, et al. Football and team handball training postpone cellular aging in women. Sci Rep. 2021;11(1):11733. [37] LAROCCA TJ, SEALS DR, PIERCE GL. Leukocyte telomere length is preserved with aging in endurance exercise-trained adults and related to maximal aerobic capacity. Mech Ageing Dev. 2010;131(2):165-167. [38] FRIEDENREICH CM, WANG Q, TING NS, et al. Effect of a 12-month exercise intervention on leukocyte telomere length: Results from the ALPHA Trial. Cancer Epidemiol. 2018;56:67-74. [39] MASON C, RISQUES RA, XIAO L, et al. Independent and combined effects of dietary weight loss and exercise on leukocyte telomere length in postmenopausal women. Obesity (Silver Spring). 2013;21(12):E549-E554. [40] PUTERMAN E, WEISS J, LIN J, et al. Aerobic exercise lengthens telomeres and reduces stress in family caregivers: A randomized controlled trial - Curt Richter Award Paper 2018. Psychoneuroendocrinology. 2018;98:245-252. [41] SCHELLNEGGER M, LIN AC, HAMMER N, et al. Physical Activity on Telomere Length as a Biomarker for Aging: A Systematic Review. Sports Med Open. 2022; 8(1):111. [42] FLYNN MG, MCFARLIN BK, PHILLIPS MD, et al. Toll-like receptor 4 and CD14 mRNA expression are lower in resistive exercise-trained elderly women. J Appl Physiol (1985). 2003;95(5):1833-1842. [43] FRANZKE B, HALPER B, HOFMANN M, et al. The impact of six months strength training, nutritional supplementation or cognitive training on DNA damage in institutionalised elderly. Mutagenesis. 2015;30(1):147-153. [44] SOARES JP, SILVA AM, OLIVEIRA MM, et al. Effects of combined physical exercise training on DNA damage and repair capacity: role of oxidative stress changes. Age (Dordr). 2015;37(3):9799. [45] DIMAURO I, SCALABRIN M, FANTINI C, et al. Resistance training and redox homeostasis: Correlation with age-associated genomic changes. Redox Biol. 2016;10:34-44. [46] GARGALLO P, COLADO JC, JUESAS A, et al. The Effect of Moderate- Versus High-Intensity Resistance Training on Systemic Redox State and DNA Damage in Healthy Older Women. Biol Res Nurs. 2018;20(2):205-217. [47] KADI F, PONSOT E, PIEHL-AULIN K, et al. The effects of regular strength training on telomere length in human skeletal muscle. Med Sci Sports Exerc. 2008;40(1):82-87. [48] HAGSTROM AD, DENHAM J. The Effect of Resistance Training on Telomere Length in Women Recovering from Breast Cancer. J Funct Morphol Kinesiol. 2018;3(1):9. [49] HAJIZADEH MALEKI B, TARTIBIAN B. High-intensity interval training modulates male factor infertility through anti-inflammatory and antioxidative mechanisms in infertile men: A randomized controlled trial. Cytokine. 2020;125:154861. [50] HAJIZADEH MALEKI B, TARTIBIAN B, CHEHRAZI M. The effects of three different exercise modalities on markers of male reproduction in healthy subjects: a randomized controlled trial. Reproduction. 2017;153(2):157-174. [51] HELLSTEN Y, APPLE FS, SJÖDIN B. Effect of sprint cycle training on activities of antioxidant enzymes in human skeletal muscle. J Appl Physiol (1985). 1996;81(4):1484-1487. [52] STREESE L, KHAN AW, DEISEROTH A, et al. High-intensity interval training modulates retinal microvascular phenotype and DNA methylation of p66Shc gene: a randomized controlled trial (EXAMIN AGE). Eur Heart J. 2020;41(15):1514-1519. [53] DENHAM J, O’BRIEN BJ, MARQUES FZ, et al. Changes in the leukocyte methylome and its effect on cardiovascular-related genes after exercise. J Appl Physiol (1985). 2015;118(4):475-488. [54] LAINE MK, ERIKSSON JG, KUJALA UM, et al. Effect of intensive exercise in early adult life on telomere length in later life in men. J Sports Sci Med. 2015;14(2):239-245. [55] 耿雪,申晋波.不同运动方式干预2型糖尿病机制的研究进展[J].体育科研, 2018,39(2):91-99. [56] 孙波,曾桂芳,林森,等.不同运动方式对T2DM大鼠形成过程中肾脏氧化应激及细胞凋亡表达的影响[J].华南国防医学杂志,2022,36(1):1-6. [57] 田鹏,师捷璇,马娇,等.不同运动方式对青年期和老年期SAMP8小鼠胫骨前肌氧化应激影响的研究[J].中国康复医学杂志,2020,35(12):1416-1421. [58] FISHER-WELLMAN K, BLOOMER RJ. Acute exercise and oxidative stress: a 30 year history. Dyn Med. 2009;8:1. [59] TOMASETTI M, GAETANI S, MONACO F, et al. Epigenetic Regulation of miRNA Expression in Malignant Mesothelioma: miRNAs as Biomarkers of Early Diagnosis and Therapy. Front Oncol. 2019;9:1293. [60] 袁乾坤.抗阻训练调控Mitofusin-2表达对老龄大鼠骨骼肌线粒体功能的影响[D].大连:辽宁师范大学,2015. [61] 潘子君.一次性递增负荷运动激活骨骼肌UPRmt和线粒体自噬的研究[D].天津:天津体育学院,2021. [62] 王蒙,张海峰.高强度间歇运动通过调控Nrf2/ARE和NF-κB信号通路调节高脂饮食诱导肥胖大鼠的氧化应激和炎症反应[J].基因组学与应用生物学, 2020,39(5):2324-2331. [63] STEPHENS NA, BROUWERS B, EROSHKIN AM, et al. Exercise Response Variations in Skeletal Muscle PCr Recovery Rate and Insulin Sensitivity Relate to Muscle Epigenomic Profiles in Individuals With Type 2 Diabetes. Diabetes Care. 2018;41(10):2245-2254. [64] LI J, WANG Z, LI C, et al. Impact of Exercise and Aging on Mitochondrial Homeostasis in Skeletal Muscle: Roles of ROS and Epigenetics. Cells. 2022;11(13):2086. [65] 王艳霞.运动引起的血流剪切力信号调控内皮细胞功能的体外研究[D].大连:大连理工大学,2019. [66] ZHANG J, RANE G, DAI X, et al. Ageing and the telomere connection: An intimate relationship with inflammation. Ageing Res Rev. 2016;25:55-69. [67] KHAN S, CHUTURGOON AA, NAIDOO DP. Telomeres and atherosclerosis. Cardiovasc J Afr. 2012;23(10):563-571. [68] 李俊,王少兵,许桂清,等.有氧运动和二甲双胍对2型糖尿病大鼠血管炎症的影响及机制[J].上海体育学院学报,2019,43(2):69-76,96. [69] 刘波,陈祥和,杨康,等.骨代谢紊乱的表观遗传重编程与运动调控[J].中国组织工程研究,2021,25(20):3210-3218. [70] CHILTON W, O’BRIEN B, CHARCHAR F. Telomeres, Aging and Exercise: Guilty by Association. Int J Mol Sci. 2017;18(12):2573. |
| [1] | Liu Jinyu, Zhang Hanshuo, Cui Hongpeng, Pan Lingzhi, Zhao Boran, Li Fei, Ding Yu. Finite element biomechanical analysis of minimally invasive treatment of cervical spondylotic myelopathy and accurate exercise rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1359-1364. |
| [2] | Zhao Lu, Zhao Yifei, Gao Da, Liu Yanfang, Fu Tingting, Xu Jiangyan. Expression of suppressor of Zeste 12 in kidney tissues of rats with diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1179-1186. |
| [3] | Guo Tingting, Xie Hong, Xu Guanghua. Finite element analysis of elastic ankle brace performance [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1031-1037. |
| [4] | Zhang Qing, Gao Chunlan, Yu Feifei, Zhang Zhenghao, Ma Fang, Gao Yuan, Li Guizhong, Jiang Yideng, Ma Shengchao. Ephrin A receptor 2 DNA methylation increases in pancreatic beta cell apoptosis induced by homocysteine [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 714-719. |
| [5] | Zeng Lulu, Xie Hong. Finite element method for predicting the effect of sports knee brace on knee ligaments under different sports conditions [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(36): 5771-5777. |
| [6] | Chen Peng, Wang Ling, Dong Shiyu, Ding Yue, Jia Shaohui, Kou Xianjuan, Zheng Cheng. Effects of whole-body vibration training on anterior cruciate ligament reconstruction: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(36): 5875-5883. |
| [7] | Han Weiyu, Chen Yuanxing, Huang Youyang, Liu Weiwei, Zhao Yongchao, Zhao Ranzun. Regulation of cardiovascular diseases by histone deacetylation modification [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5707-5713. |
| [8] | Zhang Liumei, Liu Jingjing, Lin Xiaoye, Liu Lin, Lu Jiao. Mechanism by which high-intensity intermittent exercise improves skeletal muscle injury and enhances exercise capacity in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5603-5609. |
| [9] | Xiong Bo, Zeng Ping, Liu Jinfu, Lu Guanyu, Chen Cai, Huang Yue, Chen Lihua. Roles of N6-methyladenosine methyltransferase-like 3 in regulating bone metabolism and related diseases [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(28): 4566-4570. |
| [10] | Jiang Yifan, Geng Yu, Zhang Xin, Zuo Zhongfu. Mechanism of dexamethasone against high glucose-induced oxidative damage to glomerular podocytes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3852-3857. |
| [11] | Lin Shukai, Liu Zhonghai, Liu Zhen, Cai Jincheng, Wei Ruting. Role of m6A RNA methylation modification in necrotic apoptosis in a rat model of brain injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3167-3172. |
| [12] | Yuan Shuyue, Liu Chunyan, Liu Bing, Zhao Fei. Macrophage polarization and periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2699-2707. |
| [13] | Liu Kun, Yin Lulu, Ma Zheng, Huang Lihua, Zhu Ran, Fang Ping, Zhang Ye, Ma Yanhong. Kinesiology tape for treating shoulder pain [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(14): 2214-2221. |
| [14] | Chen Chunxiao, Jiang Letao, Shen Yue, Yan Jiakai, Li Ke, Shi Yu, Lin Wenzheng, Li Hanwen, Chen Hao. Memory effect of bone marrow mesenchymal stem cells on osteogenesis after mechanical stimulation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1501-1506. |
| [15] | Han Weiyu, Zhao Yongchao, Zhao Ranzun. Progress in studies on biological properties of stem cells modified by mRNA m6A methylation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1584-1592. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||