Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (30): 4883-4889.doi: 10.12307/2023.801
Previous Articles Next Articles
Application of carbon dots-based materials in bone tissue engineering
Zhang Qingmei1, Zhang Lupeng1, Du Xiujuan1, Li Bing2
- 1School of Applied Science, Taiyuan University of Science and Technology, Taiyuan 030024, Shanxi Province, China; 2School of Stomatology of Shanxi Medical University, Shanxi Key Laboratory of Prevention and Treatment of Oral Diseases and New Materials, Taiyuan 030001, Shanxi Province, China
-
Received:2022-05-05Accepted:2022-07-08Online:2023-10-28Published:2023-04-03 -
Contact:Li Bing, MD, Chief physician, Doctoral supervisor, School of Stomatology of Shanxi Medical University, Shanxi Key Laboratory of Prevention and Treatment of Oral Diseases and New Materials, Taiyuan 030001, Shanxi Province, China -
About author:Zhang Qingmei, MD, Associate professor, Master’s supervisor, School of Applied Science, Taiyuan University of Science and Technology, Taiyuan 030024, Shanxi Province, China -
Supported by:National Natural Science Foundation of China, No. 11704274 (to ZQM)
CLC Number:
Cite this article
Zhang Qingmei, Zhang Lupeng, Du Xiujuan, Li Bing. Application of carbon dots-based materials in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4883-4889.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
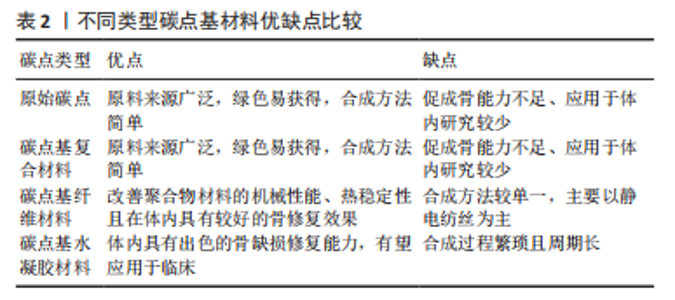
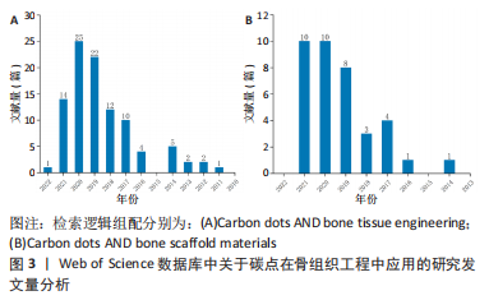
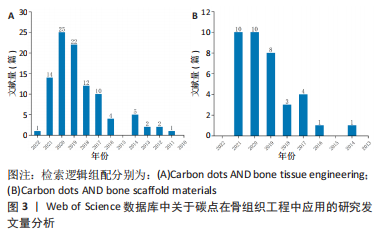
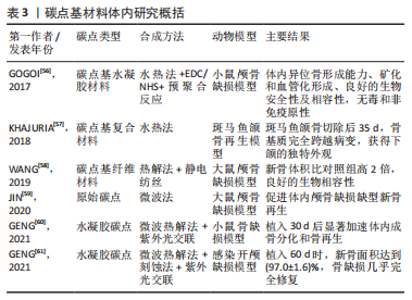
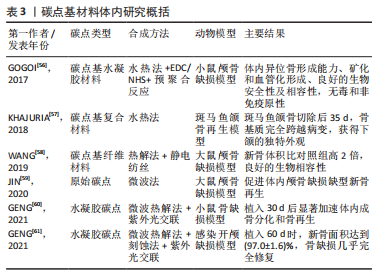
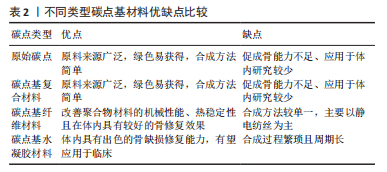
从图3可以看出,无论是碳点在骨组织工程还是骨支架材料中的应用近5年来均出现了上升趋势。这表明碳点在骨组织工程应用中具有很好的发展潜力,同时表明将碳点应用于骨组织工程或者骨支架材料是一种新的发展趋势。 2.2 碳点基材料发展历程 近年来,骨组织工程因组织来源丰富、无继发感染等独特优势从而成为骨缺损治疗的研究热点。鉴于骨组织工程在治疗骨缺损和相关疾病方面的巨大潜力,已经研究了多种生物材料,如金属、陶瓷、聚合物和碳基纳米材料,以构建具有适当结构、机械和生物学特性的骨支架材料。目前世界范围内迫切需要成本较低、生物活性高、可实现体内骨组织重建和初始强度高的高性能骨组织工程支架材料。 在2014年,FAN等[54]通过水热法成功合成了具有与周围组织更高的骨整合能力、生物兼容性以及更好成骨分化能力的碳点和羟基磷灰石复合物,这是碳点基材料应用于骨组织工程研究的首次报道。2015年,GOGOI等[55]报道了碳点基纤维支架材料,该工作首次证明碳点可以促进成骨细胞在可生物降解单宁酸基水性超支化聚氨酯支架上生长。2016年,QIU等[40]率先研究了石墨烯量子点对骨髓间充质干细胞自我更新和分化潜力等的影响,这是原始碳点应用于骨组织工程研究的最早报道。2017年,GOGOI等[56]使用带有肽功能化水性超支化聚氨酯制备了碳点凝胶,用肽功能化的凝胶不仅增强了支架的生物相容性,而且提高了聚合物系统的骨导率和骨分化能力,有望直接应用于骨组织工程研究。该工作不仅是碳点基水凝胶支架材料研究的首次报道,同时也是碳点基支架材料应用于体内研究的首次报道。 在2018年,KHAJURIA等[57]通过静电相互作用和氢键作用将羟基磷灰石纳米颗粒和N掺杂碳点复合,该碳点基复合材料增强了斑马鱼颚骨再生,增加了受伤部位的矿物质密度,该工作是碳点基复合材料应用于体内的首次也是唯一的报道。2019年,WANG等[58]将静电纺丝和热解法相结合得到的Zn-碳点/羟基磷灰石/明胶支架材料应用于大鼠颅骨缺损模型,修复了体内严重的骨缺损,这是碳点基纤维材料应用于体内的首次报道。2020年,JIN等[59]以抗坏血酸和聚乙烯亚胺为原料,通过微波法合成的碳点不仅有效增强了体外基质矿化,而且促进了体外成骨分化及颅骨缺损模型在体内的新骨再生,该工作是原始碳点应用于体内骨修复的最早报道。2021年,GENG等[60]的研究表明,带负电的石墨烯量子点基水凝胶材料在体内具有显著的骨修复效果。同年,该作者将带正电的碳量子点和带负电的碳量子点结合起来构建了一种多功能可生物降解的水凝胶支架材料[61]。体内研究发现,该支架同时具有抗菌和促成骨双重效果,为治疗感染性骨缺损提供了一种有前景的候选材料。 2.3 碳点基材料成骨分化机制 澄清碳点基材料成骨分化机制对于促进该材料的合理设计及在骨组织工程应用中的发展至关重要。2018年,KHAJURIA等[57]的研究认为N掺杂碳点和羟基磷灰石复合材料通过双重机制增强成骨分化:首先通过激活骨形态发生蛋白信号通路,其次是通过激活自噬并以形态依赖性方式释放N掺杂碳点和羟基磷灰石复合纳米颗粒,以促进成骨分化基因如 RUNX2、碱性磷酸酶和骨钙素的表达。该工作首次提出成骨机制是通过激活骨形态发生蛋白/Smad信号通路来实现的,该通路介导成骨相关基因和蛋白质的表达。该工作奠定了碳点基材料成骨机制的基础,随后的研究证实了这一结论[59-60]。在2019年,WANG等[58]提出 Zn-碳点可以作为骨髓间充质干细胞的化学刺激剂,通过活性氧途径激活MAPK信号通路,从而诱导成骨分化。锌可以通过增加Runx2/Cbfa1转录因子的表达和调节Ⅰ型胶原蛋白、碱性磷酸酶和骨钙素等蛋白质的合成来刺激成骨细胞分化;同时,锌通过抑制成骨细胞中RANKL的表达而显示出对破骨细胞形成的抑制作用。 在2020年,JIN等[59]在小鼠成骨细胞中使用抗坏血酸碳点,发现该碳点激活了细胞中的内质网应激,导致细胞内钙水平的增加。碳点通过激活PERK-eIF2αATF4途径上调了骨丝裂蛋白和骨钙素的表达,进而促进体内的成骨分化。通常,MAPK或Smad信号通路的激活可上调下游成骨相关基因和蛋白,包括COL-1,OCN和RUNX2。2021年,Geng等[60]的研究表明,带正电的骨形态发生蛋白和带负电的石墨烯量子点之间的相互作用诱导骨形态发生蛋白信号通路表达的增加,然后促进Smad1/5磷酸化。由磷酸化的 Smad1/5和Smad4 形成的复合物随后移位到细胞核中进而促进了人间充质干细胞的上调。这一机制再次证实了KHAJURIA等[57]当初提出的促成骨机制。 2.4 碳点基材料分类 基于合成方法的难易程度,该综述将碳点基材料分为4种类型,分别为原始碳点、碳点基复合材料、碳点基纤维材料和碳点基水凝胶材料。文章按照研究的先后顺序,梳理了每一种碳点基材料应用于骨组织工程的研究现状。 2.4.1 原始碳点 最近,关于单独使用碳点促进骨组织再生能力的研究也浮出水面。在这些研究中,碳点在适当浓度下表现出非常好的生物相容性,并且没有明显的细胞毒性。2016年,QIU等[40]率先研究了石墨烯量子点对骨髓间充质干细胞自我更新和分化潜力等的影响,石墨烯量子点促进碱性磷酸酶活性增强,相关成骨基因(如Runx2、骨桥蛋白和骨钙素)和特异性生物标志物表达上调;进一步的基因微阵列结果表明,石墨烯量子点通过骨形态发生蛋白和转化生长因子β信号通路增强骨髓间充质干细胞的成骨分化。在2019年,YANG等[62]通过一步水热葡萄糖镁盐合成Mg掺杂碳点,并发现该碳点可以通过改善碱性磷酸酶活性和上调成骨相关基因表达来诱导成骨分化,且随着碳点培养浓度的增加,形成的钙结节水平明显升高。同时,掺杂不同金属碳点也被证明有良好的成骨能力,拉开了金属离子掺杂碳点应用于促成骨研究的序幕[63]。 在2020年,JIN等[59]通过一步微波辅助热解法获得了抗坏血酸碳点,该碳点不仅有效增强了体外基质矿化,而且促进了体外成骨分化及颅骨缺损模型在体内的新骨再生。在小鼠成骨细胞中加入抗坏血酸碳点,发现碳点激活了细胞中内质网应激,导致细胞内钙水平的增加。研究表明,成骨机制主要是源于碳点通过激活PERK-eIF2αATF4途径上调了骨丝裂蛋白和骨钙素的表达,该碳点有望应用于修复临床实践中的骨缺损。类似的研究还有HAN等[64]通过腺苷和阿司匹林的一步水热法制备了一种新型生物活性碳点,该碳点具有很好的细胞相容性和生物安全性,能够对骨髓间充质干细胞进行长期荧光追踪;与单独用腺苷或阿司匹林处理的细胞相比,该碳点表现出更有效的成骨分化行为;在没有任何外部骨诱导因子的情况下,基于腺苷和阿司匹林的碳点可以诱导骨髓间充质干细胞的成骨分化。进一步将合成的碳点应用于小鼠动物模型,实验上观察到28 d后未出现任何不良反应,表明它具有良好的生物学安全性。综上所述,原始碳点具有促成骨分化的能力已经得到证实。 2.4.2 碳点基复合材料 碳点基复合材料是应用于骨组织工程研究的最早报道。在2014年,FAN等[54]首次以十六烷基三甲基溴化铵为软模板,柠檬酸三钠作为钙离子螯合剂,氧化石墨烯作为还原剂,通过一锅水热法成功合成了氧化石墨烯/羟基磷灰石纳米棒复合材料,与原始氧化石墨烯和羟基磷灰石相比,含有40% 羟基磷灰石的复合材料具有与周围组织更高的骨整合能力、生物兼容性以及更好的成骨分化能力。在2018年, KHAJURIA等[57]通过静电相互作用和氢键作用将羟基磷灰石纳米颗粒和N掺杂碳点复合,他们的研究发现该碳点复合材料显著增加了大鼠成骨细胞的增殖、矿化和碱性磷酸酶活性;在体内研究中,该复合材料还增强了斑马鱼颚骨再生,增加了受伤部位的矿物质密度,表明该复合材料在骨再生和骨折愈合中的治疗潜力;同时,该研究在分子水平上证实了这种现象是通过激活骨形态发生蛋白信号通路来实现的,该通路介导了成骨相关基因和蛋白质的表达。最近,SAMADIAN等[65]为了提高碳量子点-藻酸盐复合材料的生物降解性,设计了微胶囊结构的新型碳量子点-藻酸盐-明胶纳米复合材料,采用DAPI染色和MTT检测微胶囊中骨髓间充质干细胞结合和增殖情况,通过评估碱性磷酸酶的活性来研究骨髓间充质干细胞的成骨分化。研究发现,14 d后碳量子点-海藻酸盐-明胶的成骨率高于海藻酸盐明胶(不含碳量子点的复合材料)。作为复合材料的一部分,碳点的添加被证明可增加支架的机械强度,细胞附着力和增殖。 2.4.3 碳点基纤维材料 在2015年,印度科学家GOGOI等[55]首次报道由Colocasia esculenta球茎合成碳点,并与单宁酸基水性超支化聚氨酯复合,在脂肪酸基聚酰胺存在下,通过加入不同质量百分比(0.5%,1.0%和1.5%)碳点,并利用甘油基超支化环氧树脂固化水性纳米复合材料得到所需的热固性物质。与热塑性原始材料相比,热固性碳点基复合材料表现出机械和热稳定性的剂量依赖性改善;同时,该复合薄膜对MG63成骨细胞具有良好的体外黏附、增殖和分化能力。该工作首次证明碳点可以促进成骨细胞在可生物降解支架上的生长。类似的工作表明,碳点的加入一方面调控了聚合物薄膜的形貌且诱导了薄膜表面的矿化[66],另一方面纤维支架的热稳定性以及细胞相容性均得到了改进,同时保留了生物降解性和成骨的生物活性[67-68]。为了进一步改善聚已内酯基质的机械性能,YADAV等[69]在静电纺丝过程中将纳米氧化石墨烯掺入聚乳酸或聚已内酯中,制备的纳米纤维支架具有增强的力学性能、增加的亲水性、良好的细胞相容性和成骨生物活性。在碳点低负载(0.5%-2.5%)以下,与相应的纯纤维相比,纤维的强度和刚度均得到了有效改善。 在2018年,LU等[42]制备了一种新型的碳点掺杂壳聚糖/羟基磷灰石支架,该支架通过在体外上调和成骨相关的基因来增强大鼠骨髓间充质干细胞黏附和骨诱导能力。与纯支架相比,4周时显著改善了血管化新骨组织的形成;同时受碳点优异的光热效应启发,该支架被应用于近红外照射(808 nm,1 W/cm2)下肿瘤的光热治疗,该支架在体外显著抑制骨肉瘤细胞增殖,在体内有效抑制肿瘤生长,且在近红外照射下对临床采集的金黄色葡萄球菌和大肠杆菌具有显著的抗菌特性。该工作表明碳点基支架材料应用于杀死肿瘤细胞,尤其是难以触及的骨癌细胞以及用于预防/治疗骨损伤或手术后常见感染的潜力。在2019年,WANG 等[58]通过一步水热法合成了Zn2+钝化的碳点,研究发现,Zn2+作为表面钝化剂可以稳定碳点,同时在体外表现出有效的荧光成像和诱导MC3T3-E1 细胞成骨分化的能力;体内实验中,作者构建了大鼠颅骨缺损模型,并应用Zn掺杂碳点修复了严重的骨缺损。同年,SHAFIEI等[43]评估了碳点在聚己内酯/聚乙烯醇纳米纤维支架中的骨传导性、生物相容性和体外成骨,结果表明,碳点/磷酸钙的协同效应导致最高的成骨分化和增殖率。此外,2020年,GHORGHI 等[70]通过静电纺丝方法制备了卡托普利/碳量子点/聚己内酯纳米复合材料支架,碳量子点浓度为0.5%的支架由于纤维直径减小和表面亲水性增加而显著增加了 MG-63 细胞的黏附、增殖和碱性磷酸酶活性。可以看出碳点基纤维材料有望成为未来应用于骨组织工程中的一种替代材料。 2.4.4 碳点基水凝胶材料 水凝胶支架具有优良的生物相容性、可降解性和结构可控性,使得它能为细胞增殖分化提供一个良好的生物活性环境。但是水凝胶自身不能有效地对抗细菌、促进成骨分化,为此人们想到将具有促成骨分化的碳点搭载于水凝胶支架上可能有效促进骨再生。2017年,GOGOI 等[56]使用带有肽功能化水性超支化聚氨酯制备两种碳点凝胶,所开发的凝胶复合物具有高度的特异性,并刺激成骨细胞黏附、增殖、分化以及诱导血管形成。植入2 周后,凝胶被成熟的成骨细胞覆盖,并且在载有成骨细胞的支架上血管化和矿化结节更为明显。用肽的功能化不仅增强了生物相容性,而且提高了聚合物系统的骨导率和骨分化能力,有望直接应用于骨组织工程。2019年,LU等[71]通过天然产物交联剂(京尼平)将生物相容性碳点缀合到胶原蛋白上,以制备可注射胶原蛋白-京尼平-碳点水凝胶,与纯胶原蛋白水凝胶相比,该碳点基水凝胶的压缩模量高出21倍,降解率低39.3%,同时使骨髓间充质干细胞增殖增加了50.3%。碳点通过光动力疗法诱导产生少量活性氧改善了骨髓间充质干细胞的软骨形成分化,并增强了小鼠模型异位软骨形成和软骨缺损大鼠模型的软骨再生。该研究表明,碳点修饰的水凝胶注射和光动力学治疗的结合代表了一种微创修复软骨缺损的新策略。 在2021年,上海大学GENG等[60]通过调控石墨烯量子点的表面电荷,发现相较于电中性与正电荷石墨烯量子点,用带负电荷的石墨烯量子点处理后,人间充质干细胞的碱性磷酸酶活性和矿化基质显著增加;在构建的小鼠颅骨缺损模型植入该碳点基水凝胶材料4周后,带负电荷的石墨烯量子点处理小鼠的新骨量明显高于对照组,表明掺杂负电荷石墨烯量子点的水凝胶具有更好的促进颅骨缺损部位愈合的潜力。同年,该课题组又构建了一种多功能可生物降解带正电碳量子点/二硫化钨和带负电碳量子点共封装的明胶甲基丙烯酰胺水凝胶支架,在耐甲氧西林金黄色葡萄球菌感染的开颅缺损模型中植入该支架,发现感染的骨缺损几乎完全修复。该支架将带正电碳量子点带负电碳量子点结合起来实现了抗菌和促成骨双重效果,为治疗感染性骨缺损提供了一种有前景的候选材料[61]。毫无疑问,碳点基水凝胶支架材料在未来应用于体内骨缺损修复方面展现出了巨大的潜力。 此外,吉林大学BU等[72]以抗坏血酸和聚乙烯亚胺为原料,采用微波辅助热解合成表面带正电荷碳点,该作者将该碳点与具有促进成骨能力且表面带负电荷的miR-2861基因通过静电相互作用结合,再通过细胞膜的内吞途径内化到骨髓间充质干细胞,结果表明碳点和miR-2861基因协同作用,显著促进体外成骨分化和新骨再生(缺损骨面积恢复71%),在未来临床骨缺损修复中展现出巨大的应用潜力。 通过上述研究可见,碳点基材料应用于骨组织工程已经取得了重要进展,由最初的原始碳点、碳点基复合材料及碳点基纤维材料再发展到兼具抗菌和促成骨的碳点基水凝胶材料,表明碳点基材料在未来应用于临床上骨缺损修复的巨大潜力。同时,文章注意到了不同类型碳点基材料在促成骨方面的应用各有优缺点,见表2。"

在2017年,GOGOI等[56]通过水热法合成碳点,然后结合EDC-NHS生物偶联和水性超支化聚氨酯缀合获得碳点基聚合物凝胶,并将两种聚合物凝胶皮下注射到小鼠的异位区域。结果表明,该碳点基凝胶复合材料表现出体内异位骨形成能力,体内矿化和血管化的研究揭示了钙化和血管化形成的发生;生化分析、血液学参数以及淋巴细胞水平证实了碳点基聚合物凝胶在体内的生物安全性以及相容性;此外,在整个体内实验期间,植入部位没有观察到任何植入后综合征,如发烧、基本体征、植入物排斥等迹象,表明其无毒和非免疫原性行为。该工作首次通过简便的途径成功开发了独特的碳点/肽/聚氨酯/明胶体系,有望直接应用于骨组织再生。在2018年,KHAJURIA等[57]采用水热法合成了碳点和羟基磷灰石复合材料,选择斑马鱼颚骨再生模型研究了该碳点基复合材料在体内的治疗潜力,对斑马鱼颌骨切除术后第0天,第14,28,35天的显微CT图像(下上颌和全身)进行定量测量骨体积和骨矿物质密度。与对照组相比,N掺杂碳点和羟基磷灰石复合材料处理组在切除后14 d修复了大量的下颌骨;对照组颚骨再生分析显示,在切除后35 d,骨基质完全跨越了所有实验动物的病变,获得了下颌的独特外观。2019年,WANG等[58]以葡萄糖酸锌、明胶和羟基磷灰石为原料,结合热解法和静电纺丝技术制备了碳点/羟基磷灰石/明胶支架并构建大鼠颅骨缺损模型,研究发现Zn-碳点组的新骨形成体积比对照组高2倍,进一步的组织学评估证实,Zn-碳点组在8 周时明显出现新骨形成。 在2020年,JIN等[59]以抗坏血酸和聚乙烯亚胺为前驱体,采用简单快速的微波法合成了碳点,并通过建立大鼠颅骨缺损模型研究了原始碳点在体内的促成骨能力。研究发现,对照组的缺损区域新骨的形成非常有限,而碳点治疗组的新骨形成显着增加;定量分析数据显示,碳点治疗组6周和12周时被新骨覆盖的面积分别达到(23.9±3.12)%和(36.2±2.17)%。2021年,GENG等[60]通过紫外光交联将带负电荷的石墨烯量子点封装在明胶甲基丙烯酰胺中,从而获得3D可注射碳点基水凝胶支架,将该水凝胶支架注入小鼠颅骨缺损模型的缺陷部位来评估其骨修复效果。定量分析结果表明,植入带负电的石墨烯量子点/明胶甲基丙烯酰胺水凝胶支架4周后新骨面积达到(94.6±3.5)%,远高于明胶甲基丙烯酰胺水凝胶组(8.4±2.1)%,证实增强的骨再生是通过掺入带负电荷的石墨烯量子点来实现的。同年,该作者构建了一种多功能可生物降解的带正电碳量子点/二硫化钨和带负电碳量子点共封装的明胶甲基丙烯酰胺水凝胶支架[61],在感染的开颅手术缺陷模型中,植入该水凝胶支架可使感染骨缺损几乎完全修复。 总之,碳点基材料在体内的研究还较少,目前仅占到全部研究的1/3(8/24),因此这方面的研究工作仍然有待进一步深入。作者注意到,原始碳点、碳点基复合材料和碳点基纤维材料在体内的研究非常少,目前都只有1篇文献。而对于碳点基水凝胶材料,报道的所有工作均研究了体内,且是开展体内研究工作最早的一类碳点,提示碳点基水凝胶材料未来应用于临床上骨缺损修复的潜力。同时,碳点基纤维材料的研究工作最多(文章引用了9篇),说明碳点基纤维材料也是有望应用于临床上骨缺损修复的一种很有前途的材料。此外,骨支架材料研究的一个重要趋势是开发基于复合材料而不是单一材料的支架。目前的研究表明,具有不同纳米结构和化学成分的纳米材料可以影响不同的成骨分化途径,如转化生长因子β、骨形态发生蛋白和Wnt信号通路等[73-75]。然而,实际的成骨分化过程总是取决于多种途径的协同作用,而不是单一途径。因此,采用碳点复合材料制造骨支架是有意义的,其中可以激活多个成骨分化途径。碳点基纳米材料因其在促进聚合物和生物陶瓷等各种材料的成骨性能和机械性能的提升方面显示出巨大潜力,因此是未来非常有前途的候选材料之一。"

| [1] GONCALVES EM, OLIVEIRA FJ, SILVA RF, et al. Three-dimensional printed PCL-hydroxyapatite scaffolds filled with cnts for bone cell growth stimulation. J Biomed Mater Res B Appl Biomater. 2016;104(6):1210-1219. [2] 李欣. 聚己内酯基骨组织工程支架的制备和性能[J].兰州:兰州大学,2017. [3] HUANG B, VYAS C, BYUN JJ, et al. Aligned multi-walled carbon nanotubes with nanohydroxyapatite in a 3d printed polycaprolactone scaffold stimulates osteogenic differentiation. Mater Sci Eng C Mater Biol Appl. 2020;108:110374. [4] 余和东,丽张,夏凌云,等.3D打印成型纳米羟基磷灰石/壳聚糖/聚己内酯三元复合支架材料的构建及表征[J].中国组织工程研究,2020,24(10):1496-1501. [5] WANG Q, ZHANG Y, LI B, et al. Controlled dual delivery of low doses of bmp-2 and vegf in a silk fibroin-nanohydroxyapatite scaffold for vascularized bone regeneration. J Mater Chem B. 2017;5(33):6963-6972. [6] QI Q, YAO Y, JIA X, et al. Effects of polyethylene glycol content on the properties of a silk fibroin/nano-hydroxyapatite/polyethylene glycol electrospun scaffold. RSC Advances. 2019;9(58):33941-33948. [7] WANG K, CHENG W, DING Z, et al. Injectable silk/hydroxyapatite nanocomposite hydrogels with vascularization capacity for bone regeneration. J Mater Sci Technol. 2021;63:172-181. [8] SUN J, ZHANG Y, LI B, et al. Controlled release of BMP-2 from a collagen-mimetic peptide-modified silk fibroin-nanohydroxyapatite scaffold for bone regeneration. J Mater Chem B. 2017;5(44):8770-8779. [9] TSAI SW, HUANG SS, YU WX, et al. Collagen scaffolds containing hydroxyapatite-CaO fiber fragments for bone tissue engineering. Polymers. 2020;12(5):1174. [10] LIU H, DU Y, YANG G, et al. Delivering proangiogenic factors from 3D-printed polycaprolactone scaffolds for vascularized bone regeneration. Adv Healthc Mater. 2020;9(23):2000727. [11] GODOY-GALLARDO M, PORTOLES-GIL N, LOPEZ-PERIAGO AM, et al. Multi-layered polydopamine coatings for the immobilization of growth factors onto highly-interconnected and bimodal PCL/HA-based scaffolds. Mat Sci Eng C-Mater. 2020; 117:111245. [12] 杨湘俊,陈俊宇,朱舟,等.PCL基复合骨组织工程支架研究现状及发展[J].中国生物医学工程学报,2021,4(4):485-492. [13] WANG W, OMORI M, WATARI F, et al. Novel bulk carbon nanotube materials for implant by spark plasma sintering. Dent Mater J. 2005;24(4):478-486. [14] GEORGIA, PAGONA, NIKOS, et al. Carbon nanotubes: Materials for medicinal chemistry and biotechnological applications. Curr Med Chem. 2006;13(15):1789-1798. [15] MARRS B, ANDREWS R, RANTELL T, et al. Augmentation of acrylic bone cement with multiwall carbon nanotubes. J Biomed Mater Res A. 2006; 77(2):269-276. [16] LI X, LIU H, NIU X, et al. The use of carbon nanotubes to induce osteogenic differentiation of human adipose-derived mscs in vitro and ectopic bone formation in vivo. Biomaterials. 2012;33(19):4818-4827. [17] LA WG, PARK S, YOON HH, et al. Delivery of a therapeutic protein for bone regeneration from a substrate coated with graphene oxide. Small. 2013;9(23):4051-4060. [18] CAO J, LU Y, CHEN H, et al. Bioactive poly (etheretherketone) composite containing calcium polyphosphate and multi-walled carbon nanotubes for bone repair: Mechanical property and in vitro biocompatibility. J Bioact Compat Pol. 2018;33(5):543-557. [19] SARAVANAN S, CHAWLA A, VAIRAMANI M, et al. Scaffolds containing chitosan, gelatin and graphene oxide for bone tissue regeneration in vitro and in vivo. Int J Biol Macromol. 2017;104:1975-1985. [20] HERMENEAN A, CODREANU A, HERMAN H, et al. Chitosan-graphene oxide 3d scaffolds as promising tools for bone regeneration in critical-size mouse calvarial defects. Sci Rep. 2017;7(1):16641. [21] Xu X, Ray R,GU Y, et al. Electrophoretic analysis and purification of fluorescent single- walled carbon nanotube fragments. J Am Chem Soc. 2004;126(40):12736-12737. [22] SUN YP, ZHOU B, LIN Y, et al. Quantum-sized carbon dots for bright and colorful photoluminescence. J Am Chem Soc. 2006;128(24):7756-7757. [23] XIA C, ZHU S, FENG T, et al. Evolution and synthesis of carbon dots: from carbon dots to carbonized polymer dots. Adv Sci. 2019;6(23):1901316. [24] MIAO H, WANG L, ZHUO Y, et al. Label-free fluorimetric detection of cea using carbon dots derived from tomato juice. Biosens Bioelectron. 2016;86:83-89. [25] WANG Y, MA T, MA S, et al. Fluorometric determination of the antibiotic kanamycin by aptamer-induced fret quenching and recovery between moS2 nanosheets and carbon dots. Microchim Acta. 2016;184(1):203-210. [26] WANG X, JIANG X, ZHANG Z, et al. A fluorescence and resonance rayleigh scattering di-model probe was developed for trace k+ coupled n-doped carbon dot and aptamer. J lumin. 2019;214(C):116559-116559. [27] SU Y, LIU S, GUAN Y, et al. Renal clearable hafnium-doped carbon dots for ct/fluorescence imaging of orthotopic liver cancer. Biomaterials. 2020;255:120110. [28] JIANG Q, LIU L, LI Q, et al. Nir-laser-triggered gadolinium-doped carbon dots for magnetic resonance imaging, drug delivery and combined photothermal chemotherapy for triple negative breast cancer. J Nanobiotechnology. 2021;19(1):64. [29] CHEN BB, LIU ML, HUANG CZ,et al. Recent advances of carbon dots in imaging-guided theranostics. TrAC-Trend Anal Chem. 2021;134:116116. [30] ZHENG M, LIU S, LI J, et al. Integrating oxaliplatin with highly luminescent carbon dots: An unprecedented theranostic agent for personalized medicine. Adv Mater. 2014;26(21):3554-3560. [31] SUN T, ZHENG M, XIE Z, et al. Supramolecular hybrids of carbon dots with doxorubicin: synthesis, stability and cellular trafficking. Mater Chem Front. 2017;1(2):354-360. [32] CHEN S, JIA Q, ZHENG X, et al. Pegylated carbon dot/mno2 nanohybrid: a new ph/h2o2-driven, turn-on cancer nanotheranostics. Sci China Mater. 2018;61(10):1325-1338. [33] HUA XW, BAO YW, ZENG J, et al. Ultrasmall all-in-one nanodots formed via carbon dot-mediated and albumin-based synthesis: Multimodal imaging-guided and mild laser-enhanced cancer therapy. ACS Appl Mater Inter. 2018;10(49):42077-42087. [34] SUN Q, WANG Z, LIU B, et al. Self-generation of oxygen and simultaneously enhancing photodynamic therapy and mri effect: an intelligent nanoplatform to conquer tumor hypoxia for enhanced phototherapy. Chem Eng J. 2020;390:124624. [35] PAL P, DAS B, DADHICH P, et al. Carbon nanodot impregnated fluorescent nanofibers for in vivo monitoring and accelerating full-thickness wound healing. J Mater Chem B. 2017;5(32):6645-6656. [36] OMIDI M, YADEGARI A, TAYEBI L, et al. Wound dressing application of ph-sensitive carbon dots/chitosan hydrogel. RSC Adv. 2017;7(18):10638-10649. [37] WANG Z, LIU L, BU W, et al. Carbon dots induce epithelial-mesenchymal transition for promoting cutaneous wound healing via activation of tgf‐β/p38/snail pathway. Adv Funct Mater. 2020;30(43):2004886. [38] LI P, LIU S, YANG X, et al. Low-drug resistance carbon quantum dots decorated injectable self-healing hydrogel with potent antibiofilm property and cutaneous wound healing. Chem Eng J. 2021;403:126387. [39] BELZA J, OPLETALOVA A, POLAKOVA K, et al. Carbon dots for virus detection and therapy. Mikrochim Acta. 2021;188(12):430. [40] QIU J, LI D, MOU X, et al. Effects of graphene quantum dots on the self-renewal and differentiation of mesenchymal stem cells. Adv Healthc Mater. 2016;5(6):702-710. [41] DEHGHANI A, ARDEKANI SM, HASSAN M, et al. Collagen derived carbon quantum dots for cell imaging in 3d scaffolds via two-photon spectroscopy. Carbon. 2018; 131:238-245. [42] LU Y, LI L, LI M, et al. Zero-dimensional carbon dots enhance bone regeneration, osteosarcoma ablation, and clinical bacterial eradication. Bioconjugate Chem. 2018;29(9):2982-2993. [43] SHAFIEI S, OMIDI M, NASEHI F, et al. Egg shell-derived calcium phosphate/carbon dot nanofibrous scaffolds for bone tissue engineering: fabrication and characterization. Mat Sci Eng C-Mater. 2019;100:564-575. [44] PENG Z, MIYANJI E H, ZHOU Y, et al. Carbon dots: Promising biomaterials for bone-specific imaging and drug delivery. Nanoscale. 2017;9(44):17533-17543. [45] MOLAEI MJ. Carbon quantum dots and their biomedical and therapeutic applications: a review. RSC Adv. 2019;9(12):6460-6481. [46] MANISHA H, PRIYA SWETHA PD, SHIM YB, et al. Revisiting fluorescent carbon nanodots for environmental, biomedical applications and puzzle about fluorophore impurities. Nano-Structures Nano-Objects. 2019;20(8):100391. [47] ANSARI L, HALLAJ S, HALLAJ T, et al. Doped-carbon dots: recent advances in their biosensing, bioimaging and therapy applications. Colloid Surface B. 2021;203:111743. [48] 郑敏,刘坤梅,苏雅,等.碳点在生物医学领域中的应用[J].发光学报,2021, 42(8):1233-1244. [49] PENG Z, ZHAO T, ZHOU Y, et al. Bone tissue engineering via carbon-based nanomaterials. Adv Healthc Mater. 2020;9(5):e1901495. [50] FENG Z, ADOLFSSON KH, XU Y, et al. Carbon dot/polymer nanocomposites: From green synthesis to energy, environmental and biomedical applications. Sustain Mater Tech. 2021;29(206):00304. [51] LIU H, CHEN J, QIAO S, et al. Carbon-based nanomaterials for bone and cartilage regeneration: a review. ACS Biomater Sci Eng. 2021;7(10):4718-4735. [52] VEDHANAYAGAM M, RAJA IS, MOLKENOVA A, et al. Carbon dots-mediated fluorescent scaffolds: recent trends in image-guided tissue engineering applications. Int J Mol Sci. 2021;22(10):5378. [53] 李泽华,谢元栋,詹骆宁,等.碳点在组织再生支架中的应用研究进展[J].化学与生物工程,2022,39(2):6-9. [54] FAN Z, WANG J, WANG Z, et al. One-pot synthesis of graphene/hydroxyapatite nanorod composite for tissue engineering. Carbon. 2014;66:407-416. [55] GOGOI S, KUMAR M, MANDAL BB, et al. High performance luminescent thermosetting waterborne hyperbranched polyurethane/carbon quantum dot nanocomposite with in vitro cytocompatibility. Compos Sci Technol. 2015;118:39-46. [56] GOGOI S, MAJI S, MISHRA D, et al. Nano-bio engineered carbon dot-peptide functionalized water dispersible hyperbranched polyurethane for bone tissue regeneration. Macromol Biosci. 2017;17(3):1600271. [57] KHAJURIA DK, KUMAR VB, GIGI D, et al. Accelerated bone regeneration by nitrogen-doped carbon dots functionalized with hydroxyapatite nanoparticles. ACS Appl Mater Inter. 2018;10(23):19373-19385. [58] WANG B, YANG M, LIU L, et al. Osteogenic potential of zn(2+)-passivated carbon dots for bone regeneration in vivo. Biomater Sci. 2019;7(12):5414-5423. [59] JIN N, JIN N, WANG Z, et al. Osteopromotive carbon dots promote bone regeneration through the PERK-eIF2α-ATF4 pathway. Biomater Sci. 2020; 8(10):2840-2852. [60] GENG B, FANG F, LI P, et al. Surface charge-dependent osteogenic behaviors of edge-functionalized graphene quantum dots. Chem Eng J. 2021;417:128125. [61] GENG B, LI P, FANG F, et al. Antibacterial and osteogenic carbon quantum dots for regeneration of bone defects infected with multidrug-resistant bacteria. Carbon. 2021;184:375-385. [62] YANG M, MENG Y, LIU J, et al. Facile synthesis of mg2+-doped carbon dots as novel biomaterial inducing cell osteoblastic differentiation. Part Part Syst Char. 2019; 36(1):1800315. [63] MENG Y, YANG M, LIU X, et al. Zn2+-doped carbon dots, a good biocompatibility nanomaterial applied for bio-imaging and inducing osteoblastic differentiation in vitro. NANO. 2019;14(3):1950029. [64] HAN Y, ZHANG F, ZHANG J, et al. Bioactive carbon dots direct the osteogenic differentiation of human bone marrow mesenchymal stem cells. Colloids Surf B Biointerfaces. 2019;179:1-8. [65] SAMADIAN S, KARBALAEI A, POURMADADI M, et al. A novel alginate-gelatin microcapsule to enhance bone differentiation of mesenchymal stem cells. Int J Polym Mater. 2021;4:1-8. [66] HASSANZADEH S, ADOLFSSON KH, WU D, et al. Supramolecular assembly of biobased graphene oxide quantum dots controls the morphology of and induces mineralization on poly (epsilon-caprolactone) films. Biomacromolecules. 2016;17(1):256-261. [67] WU D, SAMANTA A, SRIVASTAVA RK, et al. Starch-derived nanographene oxide paves the way for electrospinnable and bioactive starch scaffolds for bone tissue engineering. Biomacromolecules. 2017;18(5):1582-1591. [68] WU D, SAMANTA A, SRIVASTAVA RK, et al. Nano-graphene oxide functionalized bioactive poly(lactic acid) and poly(epsilon-caprolactone) nanofibrous scaffolds. Materials. 2018;11(4):566. [69] YADAV A, ERDAL NB, HAKKARAINEN M, et al. Cellulose-derived nanographene oxide reinforced macroporous scaffolds of high internal phase emulsion-templated cross-linked poly(epsilon-caprolactone). Biomacromolecules. 2020;21(2):589-596. [70] GHORGHI M, RAFIENIA M, NASIRIAN V, et al. Electrospun captopril-loaded pcl-carbon quantum dots nanocomposite scaffold: fabrication, characterization, and in vitro studies. Polymers for Advanced Technologies. 2020;31(12):3302-3315. [71] LU Z, LIU S, LE Y, et al. An injectable collagen-genipin-carbon dot hydrogel combined with photodynamic therapy to enhance chondrogenesis. Biomaterials. 2019;218:119190. [72] BU W, XU X, WANG Z, et al. Ascorbic Acid-PEI carbon dots with osteogenic effects as miR-2861 carriers to effectively enhance bone regeneration. ACS Appl Mater Inter. 2020;12(45):50287-50302. [73] MU Q, DU G, CHEN T, et al. Suppression of human bone morphogenetic protein signaling by carboxylated single-walled carbon nanotubes. Acs Nano. 2009;3(5):1139-1144. [74] KILIAN KA, BUGARIJA B, LAHN BT, et al. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc Natl Acad Sci U S A. 2010; 107(11):4872-4877. [75] LIU DD, ZHANG JC, ZHANG Q, et al. TGF-β/BMP signaling pathway is involved in cerium-promoted osteogenic differentiation of mesenchymal stem cells. J Cell Biochem. 2013;114(5):1105-1114. |
| [1] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [2] | Yang Zhishan, Tang Zhenglong. YAP/TAZ, a core factor of the Hippo signaling pathway, is involved in bone formation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1264-1271. |
| [3] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [4] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [5] | Wang Jinling, Huang Xiarong, Qu Mengjian, Huang Fujin, Yin Lingwei, Zhong Peirui, Liu Jin, Sun Guanghua, Liao Yang, Zhou Jun. Effects of exercise training on bone mass and bone microstructure in aged osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 676-682. |
| [6] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| [7] | Liu Zixuan, Li Yan, Ji Lin, Xia Delin. Biological properties of nano-hydroxyapatite-zinc oxide composite scaffolds and their effects on the behavior of MC3T3-E1 osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5441-5447. |
| [8] | You Yan, Chen Jiawen, Lin Binbin, Wu Jingyi, Liu Peng, Wu Buling, Sun Tianyu. Mechanism and application of glycosaminoglycan in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5538-5545. |
| [9] | Xu Zhengyi, Wan Qianbing, Chen Junyu. Natural small molecular compounds in the treatment of bone-related diseases by regulating type H blood vessels and its application in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5546-5553. |
| [10] | Wang Xinjie, Wang Guodong, Zheng Zhongren, Shao Yiming, Wang Jialiang, Ma Hui, Zhao Xiaowei. Manganese-containing bioceramic materials in the field of bone repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5570-5576. |
| [11] | Xu Rongwei, Wang Hao, Fu Qiuyue, Lan Xingming, Yang Kun. Bidirectional interaction between inflammatory factors and dental pulp stem cells during bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5385-5393. |
| [12] | Xie Yingchun, Xu Wenjuan, Li Yuwan. Schnurri3 regulates osteogenic differentiation of C3H10T1/2 cells induced by bone morphogenetic protein 2 [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5270-5276. |
| [13] | Zhang Xiaoyu, Chen Qi, Yang Xing, Hao Yuefeng. Application of poly(lactic-co-glycolic acid) copolymer microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4896-4903. |
| [14] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [15] | Yu Aimin, Xu Ting, Zhu Yunying, Liang Jianqiang, Wu Donghui. Physicochemical properties of chitosan/bone powder/cellulose nanocrystals scaffold loaded with antimicrobial peptides for jaw repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3999-4005. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||