Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (7): 1117-1125.doi: 10.12307/2023.044
Previous Articles Next Articles
Application and design of piezoelectric materials for bone defect repair
Tang Haotian, Liao Rongdong, Tian Jing
- Department of Orthopedics, Zhujiang Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China
-
Received:2022-02-10Accepted:2022-03-02Online:2023-03-08Published:2022-07-20 -
Contact:Tian Jing, Master, Professor, Doctoral supervisor, Department of Orthopedics, Zhujiang Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China -
About author:Tang Haotian, Master candidate, Department of Orthopedics, Zhujiang Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China
CLC Number:
Cite this article
Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

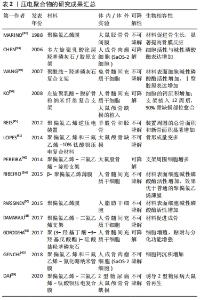
2.1 压电材料的分类 压电材料是受到压力作用时会在两端面间出现电压的晶体材料。1880年法国科学家居里兄弟观察到石英晶体在受压后表面会产生电荷,并且电荷量与所受压力成正比,因此将这一现象命名为压电效应[9]。压电材料在受到外力后发生形变,材料内部晶体中正负离子因发生位移导致中心不对称,从而出现大量由负电荷中心指向正电荷中心的电偶极矩(也称为电偶极子),使得表面出现电荷。随后于1892年,Julius Wolff提出了著名的Wolff定律:骨骼受到适当强度的力能促进骨组织的再生,而持续缺乏受力将引起骨吸收,导致骨密度降低[10]。该定律在当时解释了为何长期卧床患者容易患骨质疏松,鼓励患有骨病患者通过康复训练来加速骨修复,但其原因本质——骨的压电效应在当时尚未被发现。1957年,日本科学家首次发现骨具有压电效应,这也揭示了骨的表面生物电位的来源,为从生物电机制探究骨修复打下了基础[11]。1964年,BASSETT等[12]研究表明,骨组织在受到应力时,带负电位的一面成骨细胞增殖分化能力加快,骨重建能力增强,带正电位一面的破骨细胞活性增强,诱发骨吸收,电信号可能是骨重建的原因,此研究发现为骨的电刺激治疗打下理论基础。1969年,有研究开始了对聚偏氟乙烯压电效应的研究,而聚偏氟乙烯成为了目前研究最广泛的压电聚合物,具有应用于骨缺损修复的潜力[13]。1981年,PARK等[14]首次将压电材料应用于骨修复,首次将钛酸钡材料植入狗的股骨皮质中,发现钛酸钡压电陶瓷能和骨组织形成良好的骨结合,这种不借助于外加电源而依靠压电材料自发电给予骨断端电刺激治疗的方法首次得到验证。然而,单一的压电材料具有局限性,难以胜任骨植入物的任务。2013年CIOFANI等[15]成功制造了符合人体生理环境的稳定的水溶性钛酸钡纳米分散颗粒,是纳米材料应用于压电复合材料研发的典型。通过纳米技术,使得材料的填充、涂层修饰变得更加容易,并随着压电材料研发技术的不断更新,两种或多种压电材料相结合的压电复合材料的研发逐渐增多,其力学性能与生物活性能更加满足骨植入物的需求,成为了当前压电材料的研究热点[16],压电材料的发展见图3。"

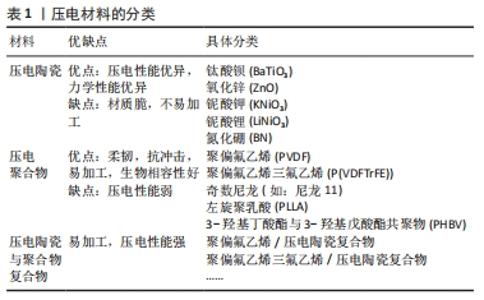
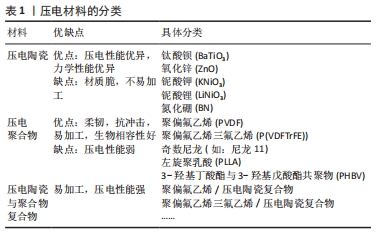
压电聚合物又分为天然压电聚合物和人工合成压电聚合物,天然压电聚合物包括纤维素、胶原和甲壳素,而人工合成压电聚合物包含聚偏氟乙烯以及它的共聚物聚偏氟乙烯三氟乙烯[poly(vinylidene fluorideand-trifluoroethylene)P(VDF-TrFE)]、奇数尼龙(如:尼龙11)、左旋聚乳酸(poly-L-lactic acid,PLLA)、3-羟基丁酸酯与 3-羟基戊酸酯共聚物(poly-3-hydroxybutyrate-3-hydroxy valerate,PHBV)等。压电聚合物具有良好的柔韧性、易加工性、抗冲击性和生物相容性,在组织工程领域得到广泛的应用。压电陶瓷是一种多晶体,如钛酸钡(barium titanate,BaTiO3)、铌酸钾钠(potassium sodium niobate,KNN)和铌酸锂钠钾(lithium sodium potassium niobate,LKNN)、氧化锌(Zinc oxide,ZnO),氮化硼纳米管(boron nitride nanotubes,BNNT)等材料,这一类压电材料相较于压电聚合物具有更强的压电性能。压电复合材料则是将压电陶瓷与聚合物通过复合工艺而制成的新型材料。压电陶瓷压电性能优异,但其脆性和不易加工性限制了其单独在骨组织工程中的应用[17]。压电聚合物具有良好的柔韧性、抗冲击性、易加工性和生物相容性,但其压电性能较弱,难以达到刺激细胞的阈值。开发聚合物-陶瓷复合材料有望结合它们的优点,克服各自的限制,研发出性能更加优异的压电材料[18]。 2.2 骨的压电效应 骨组织是一种具有再生能力的组织,骨修复也是一个复杂的过程,依赖于机械、化学、生物电等生物环境[19]。人体的骨组织是一种天然的压电材料。日本科学家于1954年首次报道了骨具有压电性[20]。人体骨骼由细胞和细胞外基质组成,其中细胞包含了成骨细胞、破骨细胞、骨细胞和骨髓细胞(包括造血干细胞),而细胞外基质由65%的矿物质(羟基磷灰石为主)与35%胶原组成,Ⅰ型胶原约占胶原的90%,具有三重螺旋结构,为骨骼提供主要的拉伸强度。而无机物则以羟基磷灰石钙的形式为骨骼提供主要的抗压强度。最早的研究认为,骨的压电效应主要来源于胶原分子。胶原是一种纤维性蛋白,FUKADA[21]的研究提出剪切力使胶原纤维相互错动进而发生极化,使骨骼体现出压电性能。骨的压电效应使骨胶原在受力发生形变时,其压缩侧呈负电位,张力侧呈正电位。天然骨骼具有固有的Zeta电位为-5 mV,而由于骨胶原的压缩而产生的负电荷增加了自然活骨中的总Zeta电位和流动电位[22]。除了胶原纤维,骨的压电性能和羟基磷灰石也有关。LANG等[23]通过对应压电力显微镜观察发现了羟基磷灰石具有压电性,该实验结果表明,羟基磷灰石对骨的压电性具有一定贡献。根据沃尔夫定律,骨可以对机械负载做出反应,在应力较大的区域骨的生长和重塑能力增强[24],其原因在于机械应力增加了骨压电电位的负电荷 [25]。在一定范围内,骨所受压力负荷越高,骨表面负电位越高,促进成骨细胞增殖分化的效果越强,成骨作用越强,这也很好地解释了为什么长期卧床的患者和太空旅行的宇航员往往骨密度低,易患骨质疏松。有研究表明,骨组织在受到应力时,带负电位的一面成骨细胞增殖分化能力加快,该面呈骨修复状态,带正电位一面的破骨细胞活性增强,该面呈骨吸收状态[26]。因此,骨的压电效应在骨修复中起重要作用。 2.3 骨修复中常用的压电材料 压电材料是电活性材料,具有成为新型骨替代材料的巨大潜力。压电材料将自身受到的压力转化为合适的电刺激,具有成为骨替代材料的潜力。骨修复研究中的压电材料除了应具备优异的电学性能以外,还应有良好的生物相容性和机械性。 2.3.1 压电聚合物 压电聚合物质地与无机晶体相比,其柔韧性好,易于加工。当前,应用于组织工程学的压电聚合物满足以下条件:①存在永久的分子偶极子;②拥有校准或对齐分子偶极子的能力;③具有维持偶极子对齐状态的能力;④聚合物在受到机械应力时抗牵张的能力[27]。压电聚合物大多生物相容性良好,但机械强度常不足,难以作为骨植入物承受生理负荷。并且聚合物压电性能不如压电陶瓷,因此单独采用压电聚合物可能难以达到促进骨修复的电刺激阈值。目前多推荐聚合物与无机陶瓷结合形成压电复合材料,一方面增强自身的机械强度,另一方面复合材料本身会使聚合物生物相容性增加,这可能是材料压电性能增强从而促进了细胞的贴附、增殖与分化功能。 组织工程研究中常见的压电聚合物如下: 聚偏氟乙烯:是当前研究最广泛的压电共聚物,其压电常数为34 pC/N。由于聚偏氟乙烯柔韧性高,无细胞毒性,因此广泛应用于生物医学领域并被研制出多种产品,包括了组织工程支架和可植入的自充电设备。当前已有研究证实压电聚合物聚偏氟乙烯基底与小鼠颅顶前成骨细胞 (MC3T3-E1)、人间充质干细胞、人脂肪干细胞和山羊骨髓干细胞的生物相容性[28-31]。极化的聚偏氟乙烯压电薄膜能产生足够的电势,诱导前成骨细胞的增殖分化。MARINO等[32]报告,当极化与未极化的聚偏氟乙烯聚合物基质分别植入大鼠胫骨骨间膜6周时,极化的聚偏氟乙烯成骨性能显著高于未极化聚偏氟乙烯。有学者报道聚偏氟乙烯共聚物和钛酸钡压电复合物相结合时能显著提高聚偏氟乙烯的压电性能,同时也能解决钛酸钡质脆、难加工的问题[33]。RIBEIRO等[34]制作的表面极化的β-聚偏氟乙烯薄膜同普通聚偏氟乙烯薄膜相比,能够提高人骨髓间充质干细胞碱性磷酸酶碱性磷酸酶的活性,同时薄膜在受到动态的机械振动时,其促成骨的效果会进一步增强。REIS等[35]报道了一种基于聚偏氟乙烯的新型压电装置能够利用逆压电效应有效地刺激骨-种植体界面处的骨生长。 当前普遍认为聚偏氟乙烯是具有良好生物相容性的热塑性聚合物,具有极高的耐腐蚀性,虽然其在极端碱性环境会降解,但在正常的生物环境下是不可降解的材料,这也因此限制了其在组织工程学的广泛应用[36]。 聚偏氟乙烯-三氟乙烯:是偏氟乙烯和三氟乙烯的共聚物,是当前压电常数最高的压电聚合物(38 pC/N)。偏氟乙烯和三氟乙烯共聚物具有良好的细胞生物相容性并对细胞的贴附和分化有促进作用[37],可促进骨、皮肤、软骨和肌腱等多种组织的再生。DAMARAJU等[38]通过静电纺丝技术制造了偏氟乙烯和三氟乙烯共聚物的3D支架,并证明了与非压电材料相比,压电性和机械应力的联合作用更能刺激人骨髓间充质干细胞的增殖、分化、细胞外基质矿化和基因表达。DAI等[39]制备的聚偏氟乙烯-三氟乙烯-钛酸钡压电复合膜可模拟骨组织生物电微环境,通过炎症介导的机制,促进糖尿病大鼠的骨再生。聚合物混合物在骨和软骨组织工程的应用的正变得越来越重要,而聚偏氟乙烯和聚偏氟乙烯-三氟乙烯与淀粉等天然聚合物结合,其形成的聚偏氟乙烯-淀粉和聚偏氟乙烯和三氟乙烯-淀粉植入物异物反应少,弹性模量与松质骨相当,可作为骨修复支架,具有应用于骨组织工程的潜力[40]。LOPES等[41]比较了聚偏氟乙烯和三氟乙烯-10%钛酸钡压电复合材料和聚四氟乙烯分别植入大鼠颅骨缺损4周和8周后的新骨生成量,与聚四氟乙烯相比,偏氟乙烯和三氟乙烯共聚物-10%钛酸钡压电复合材料的表面成骨显著增加,这清楚地表明了压电基底的促进骨生长的效果,这可能是因为聚偏氟乙烯和三氟乙烯-10%钛酸钡压电复合材料通过其压电性、亲水性从而增强蛋白结合能力,促进骨生成。而聚偏氟乙烯和三氟乙烯与氮化硼纳米管形成的偏氟乙烯和三氟乙烯共聚物- 氮化硼纳米管压电复合材料相比偏氟乙烯和三氟乙烯共聚物其压电性能提高近2倍,因此偏氟乙烯和三氟乙烯共聚物- 氮化硼纳米管压电复合材料的骨生成量会增加[42]。 聚-3-羟基丁酸-3-羟基戊酸酯:是聚羟基脂肪酸家族的一员,于1986年问世,由于其良好的生物相容性、可生物降解性和热塑性,在生物医学领域的地位逐步提高[43]。聚-3-羟基丁酸-3-羟基戊酸酯,其通过酶降解机制水解并释放二氧化碳,与其他生物聚合物相比,其生物相容性良好,植入动物体内引起的炎症反应发生率更低,并且其压电常数(1.3 pC/N)与人骨相似,以上特性使其具有模拟自然骨及作为骨植入物的潜力[44]。 聚3-羟基丁酸-3-羟基戊酸酯由于生物活性以及亲水性差,因此需要通过研发与其他材料相结合的复合压电材料来克服聚(3-羟基丁酸-3-羟基戊酸酯)的缺点。可生物降解的聚-3-羟基丁酸-3-羟基戊酸酯-羟基磷灰石复合材料已被证明具有促进成骨的功能,因此 GORODZHA等[45]成功制备了由聚(3-羟基丁酸-3-羟基戊酸酯)与含硅酸盐的羟基磷灰石组成的复合材料骨支架,该支架促进了人骨髓间充质干细胞的增殖、贴附与分化功能。有研究制备的聚-3-羟基丁酸-3-羟基戊酸酯-壳聚糖-羟基磷灰石支架相较于普通聚-3-羟基丁酸-3-羟基戊酸酯支架其生物相容性显著提高,并有效促进成骨细胞贴附、增殖与分化[46]。 总之,聚羟基脂肪酸家族可与无机材料、聚合物材料和生物活性材料相结合形成复合材料从而达到改善自身生物活性与亲水性差的问题[47]。当前对于聚(3-羟基丁酸-3-羟基戊酸酯)混合材料研发有助于不断完善单一材料自身的缺陷,以实现材料之间促进成骨效应的协同作用。通过在未来混合材料制作的统一标准出台,聚(3-羟基丁酸-3-羟基戊酸酯)有望制成可生物降解的复合支架,用来治疗骨缺损。 聚酰胺:又称为尼龙,作为最早工业化的高分子材料已有50多年的历史,由于密度小、耐磨、耐油、易加工、特别好的力学性能而广泛应用于机械、交通、电气、化工、纺织及日用品方面。而奇术尼龙(尼龙-5,尼龙-7),其每个单体单元上含有偶数个亚甲基和一个酰胺基,因为自身的一个酰胺键可以产生净偶极矩(3.7 D)从而具有压电性。尼龙的压电常数(d31)在25 ℃时为3 pC/N,在107 ℃时为14 pC/N[48]。WANG等[49]报道,聚酰胺-羟基磷灰石复合材料在植入12周后可促进成骨。聚酰胺可作为聚合物基质用于软骨修复或再生,但由于其细胞相容性一般,所以需要适当的修饰以促进软骨细胞的细胞附着和增殖。由于聚酰胺缺乏降解模式使得其在组织工程中的应用受到限制。 左旋聚乳酸:半晶态左旋聚乳酸可以在未极化的状态下产生压电性,其原因在于左旋聚乳酸受到机械应力时,其分子中的中C=O键的会发生机械移位,导致净偶极矩和电荷的出现[50]。IKADA等[51]使用左旋聚乳酸作为骨替代物,并证明了左旋聚乳酸的极化压电效应能促进骨的生成。薄膜和杆状的压电左旋聚乳酸基板已被证实具有骨组织潜在替代物的适用性[52]。含有磷灰石和胶原的左旋聚乳酸支架在细胞外培养基中能促进人成骨肉瘤细胞(SaOS-2细胞)和人胎儿成骨细胞的代谢[53]。KO等[54]通过静电纺丝技术制备了左旋聚乳酸-脱矿骨粉纳米纤维复合支架,并在大鼠模型中观察到左旋聚乳酸-脱矿骨粉纳米纤维复合支架能有效促进人骨髓间充质干细胞的成骨分化,有效修复颅骨缺损。左旋聚乳酸由于具有可生物降解的特点,因此材料加工会受到限制,而左旋聚乳酸与淀粉形成淀粉共混物能增加左旋聚乳酸的延展性和压缩模量从而改善左旋聚乳酸加工性能的问题[55]。左旋聚乳酸具有无细胞毒性、可生物降解、材质适合骨科手术植入等优势,有望制备成可降解螺钉、固定钉与缝合锚钉从而避免植入物需行二次手术取出的问题[56]。 当前左旋聚乳酸生物相容性良好,可生物降解,植入体内不良反应少,机械强度高,但因其具有疏水性不利于细胞贴附,降解产生酸性产物能引起机体局部炎症反应,以及降解时间过长,往往超过骨愈合时间,因此左旋聚乳酸需要与其他材料如纳米陶瓷材料、碱金属材料相结合,改善自身的疏水性,减弱降解产生不良反应,并将生物降解时间控制在有利于骨与血管长入支架的合理区间,这样才有助于将左旋聚乳酸应用于骨缺损修复。 2.3.2 压电陶瓷 目前常见的压电陶瓷包括了锆钛酸铅(lead zirconate titanate,PZT)、钛酸钡、氧化锌(ZnO)、铌酸钾钠(KNN)、铌酸锂钠钾(lithium sodium potassium niobate,LNPN)和氮化硼纳米管(boron nitride nanotubes),其相较于压电聚合物的特点是压电常数高。评估压电陶瓷能否应用于骨组织工程的重要指标就是材料的细胞毒性。总体而言,含铅的压电陶瓷如锆钛酸铅,具有细胞毒性,因此在组织工程的材料研发与应用中较为少见。其他无铅的压电陶瓷的细胞毒性与材料的含量相关,相较于含铅压电陶瓷,在组织工程的研究中更受关注。总体而言,钛酸钡中所含钛与钡具有细胞毒性,氧化锌分解产物活性氧具有细胞毒性,而氮化硼、铌酸钾钠等材料所含元素无细胞毒性。但当前压电陶瓷往往与其他材料结合形成压电复合材料,通过改良与修饰可以克服陶瓷自身的不足[57-79]。 目前压电聚合物研究成果见表2,压电陶瓷研究成果见表3。 "


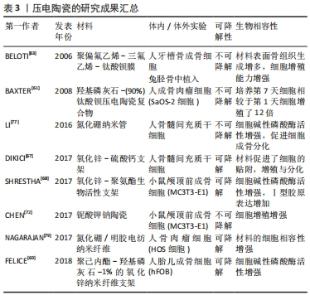
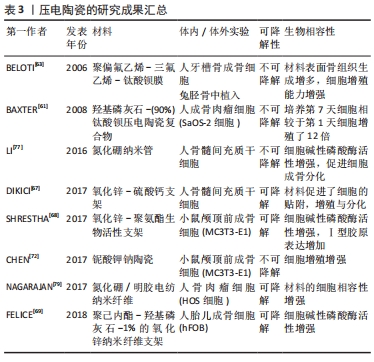
钛酸钡(BaTiO3):钛酸钡由于生物相容性良好、压电性能优异同时具有满足骨骼生理负荷的机械强度,成为了目前研究最多的压电材料[57]。钛酸钡的结构在居里温度(120 ℃)以下其形态会发生从非极性的对称立方到极性的不对称立方的转变,这使得钛酸钡会产生自发的电极化[58]。钛酸钡是最早作为骨植入物治疗骨缺损的压电材料,有研究者于1980年将钛酸钡植入狗的股骨内,最后观察到了骨组织在材料表面矿化结果良好,未见植入物引起的炎症与排斥反应,证明了钛酸钡良好的生物相容性。随着对钛酸钡的性能不断研究,发现钛酸钡压电性能优异,即使少量应用也可满足骨生长所需电微环境,因此目前钛酸钡常常少量地作为填充物或表面涂层应用于压电材料中,例如羟基磷灰石-钛酸钡,聚偏氟乙烯-钛酸钡等。 目前已有研究支持了具有生物相容性的羟基磷灰石-钛酸钡压电陶瓷在骨种植支架开发的巨大潜能。BAXTER等[60]证明人成骨肉瘤细胞(SaOS-2细胞)在羟基磷灰石-(90%)钛酸钡压电陶瓷复合物(d33=57.8 pC/N)上培养,培养第7天细胞相较于第1天细胞增殖了12倍。羟基磷灰石-钛酸钡复合物的压电性能与钛酸钡的含量有关,钛酸钡含量越高,材料压电性能越强,当钛酸钡含量低于70%,复合物便失去压电性[61]。DUBEY等[62]在小鼠膝关节内注射羟基磷灰石-40%钛酸钡颗粒。注射7 d后,小鼠的活动和体质量正常,组织病理学分析表明,注射颗粒既没有转移到任何主要器官,如心脏、肾脏、肝脏、肺等,也没有对这些器官造成任何不良影响。而聚集在膝关节区域的注射颗粒并未造成任何炎症与异物反应,这表明羟基磷灰石-钛酸钡生物相容性良好。钛酸钡诱导的压电聚合物能显著提高聚合物的生物相容性并且能有效促进体内与体外的成骨效应。BELOTI等[63]观察到压电聚合物薄膜[聚偏氟乙烯-三氟乙烯-钛酸钡]促进人成骨细胞增殖分化的能力强于非压电聚合物聚四氟乙烯,这是由于压电聚合物薄膜[聚偏氟乙烯-三氟乙烯-钛酸钡]的亲水性与蛋白结合能力更强。 从上述研究可发现,钛酸钡压电陶瓷能产生压电电位,刺激羟基磷灰石的生成,增强细胞功能,在动物模型中展现出良好的生物相容性。但钛酸钡单独作为骨植入物具有局限性,具体表现在钡离子与钛离子具有细胞毒性,且材料自身的生物惰性阻碍了细胞贴附以及不可降解,导致了钛酸钡不可长期植入体内。因此,研究者应该利用钛酸钡自身压电优异的性能,高机械强度的优势,与其他材料结合,用来改善其他骨植入材料的性能,例如与羟基磷灰石相结合以提高材料的机械强度,与钛合金相结合以提高钛合金的生物相容性和压电性能,与聚合物相结合提高通过提高材料压电效应从而提高材料生物相容性。随着对钛酸钡的研究不断深入,钛酸钡有成为新一代骨植入物的潜能。 氧化锌(ZnO):锌通过调节转录因子、金属蛋白酶和聚合酶等不同酶的活性调节细胞代谢,是细胞增殖和分化中的重要元素。氧化锌生物相容性好,抗菌性能好,是良好的组织工程材料。纳米氧化锌具有压电效应,在受到细胞自身固有机械应力时能产生局部电位,显著改善人SaOS-2成骨样细胞和巨噬细胞的代谢活性[64]。LAURENTI等[65]提出,对于特定的固定纳米尺寸,高浓度(60 μg/mL)的氧化锌纳米颗粒与低浓度(30 μg/mL)相比更容易降低骨髓间充质干细胞的生物活性,这表明氧化锌具有浓度依赖性的细胞毒性。除了浓度,氧化锌的细胞毒性还取决于颗粒直径和孔隙密度。由于纳米氧化锌会生成活性氧,随着颗粒直径的减小,活性氧生成增加,细胞毒性增强。随着孔隙密度的增加,纳米氧化锌供蛋白反应以及细胞贴附的表面积增加,成纤维细胞的功能也随之提升[66]。大量文献研究表明,在生物材料中加入适量的氧化锌可以促进材料与成纤维细胞、成骨细胞、干细胞等的生物相容性[65-67]。例如,DIKICI等[67]报告,随着硫酸钙支架中氧化锌含量增加至0.5%,材料表面的人骨髓间充质干细胞密度也随之升高,该现象可归因于压电氧化锌能增强支架的亲水性,从而利于细胞的贴附。适量的压电氧化锌也能提高聚合物支架(如聚己内酯和聚氨酯)的成骨反应。SHRESTHA等[68]通过引入体积分数0.2%氧化锌纳米颗粒,在聚氨酯聚合物支架上观察到前成骨细胞MC3T3-E1的刺激成骨增殖和分化。据报道,与不含氧化锌的支架相比,通过在聚己内酯-羟基磷灰石中加入体积分数1%的氧化锌纳米纤维支架能增强人胎儿成骨细胞活性 [69]。纳米氧化锌改善生物材料相容性的原因可能与锌离子的可持续释放相关,而释放的锌离子可调节各种细胞代谢活动,如蛋白质合成和mRNA表达等[70]。 氧化锌生物相容性好,展现出良好的压电性,自身生成的活性氧一方面具有抗菌效果,另一方面会产生剂量依赖的细胞毒性,这是不可忽视的。因此一方面,研究者在用氧化锌进行临床试验前,应该在动物模型中寻找氧化锌的最适浓度,另一方面应通过对氧化锌材料进行改进和修饰,从而使氧化锌能够作用于骨组织工程。 铌酸钾钠(KNN)和铌酸锂钠钾(LKNN):作为无铅的压电材料,其中包含的金属铌为人体必需微量元素,具有无细胞毒性、生物相容性好的特点,有学者利用铌酸钾钠的压电性能将其制为纳米药物输送设备,从而达到促进骨与软骨修复与再生的目的[71]。CHEN等[72]报道了表面极化的铌酸钾钠陶瓷材料相较于非极化陶瓷材料均能促进细胞蛋白吸收,无论是正电极化还是负电极化,但该实验中小鼠颅顶前成骨细胞(MC3T3-E1) 在负电位极化表面细胞密度显著高于正电位极化与非极化表面。铌酸锂钠钾具有化学稳定性与亲水的特点,这意味着其具有应用于骨科的价值[73]。当前利用铌酸钾钠诱导压电性的羟基磷灰石-铌酸钾钠梯度功能材料研发受到关注。羟基磷灰石-铌酸钾钠是将铌酸钾钠材料置于羟基磷灰石层之间,这样可以在不影响羟基磷灰石自身生物相容性的同时将羟基磷灰石的极化率提高3倍以上,而铌酸锂钠钾置于羟基磷灰石层之间可以使羟基磷灰石极化率提高6倍[73]。 正如上述研究所述,材料的生物相容性与自身的细胞毒性和表面极化有很大关系,而羟基磷灰石极化率的增加可能会促进细胞的增殖和分化。铌酸钾钠在受到生理负荷的情况下产生表面电荷从而达到极化的效果,能促进细胞的贴附与增殖,同时铌酸钾钠与铌酸锂钾钠自身无细胞毒性,两者相互作用使材料具有良好的生物相容性,有助于骨的再生。 氮化硼纳米管(BNNTs):硼,在分子水平上通过调节骨形态发生蛋白、骨钙素、RunX2(转录因子)的水平,在调节小鼠颅顶前成骨细胞(MC3T3-E1)和人骨髓间充质干细胞的成骨功能中发挥重要作用[74-75]。例如,NAKHMANSON等[76]研究了氮化硼纳米管(氮化硼纳米管)的压电性和自发极化的特点。氮化硼纳米管通过在培养基中释放的微量硼和氮化硼纳米管纤维对人骨髓间充质干细胞施加的使细胞受到拉伸,激活肌动蛋白丝的应力,促进人骨髓间充质干细胞向成骨细胞分化[77]。促进氮化硼纳米管成骨的另一个因素是其对细胞蛋白的亲和力强[78]。通过在明胶聚合物上添加质量分数1%–5%的氮化硼制备的静电纺纳米纤维,能使聚合物具备良好的促生物矿化作用和与人骨肉瘤细胞的相容性[79]。 随着纳米技术的发展,纳米结构的陶瓷材料开始出现。当前的压电聚合物大多生物相容性优于压电陶瓷且易于加工处理,但由于其自身机械强度与压电性能不足导致了其难以单独作为骨植入物。氮化硼纳米管仅需少量的加入便可提升压电聚合物的机械强度和压电性能,从而使复合材料能够更加符合骨植入物的力学要求。总的来说,上述研究清楚地表明了氮化硼在骨组织工程应用中的潜力。 2.4 压电材料优缺点总结 单一的压电材料在应用于骨修复缺损时都存在自身的限制与不足。应用于骨缺损修复的骨植入物需要在力学性质上满足承重要求,在细胞生物学上需要具备良好的生物活性,以利于细胞的贴附生长与分化。当前压电复合材料是组织工程的研究热点,其优势在于将不同压电材料相结合从而弥补单一材料的缺陷,整合不同材料的优点,使得制备理想的骨修复材料成为可能。为了实现此目的,一方面需要利用先进的技术手段改进材料的理化性质,通过多次实验以使材料压电电位达到可增强细胞生物相容性的临界值;另一方面便是了解不同压电材料的优缺点、不良反应、生物相容性,方可在材料的研发阶段达到优势互补的效果,从而制备出促进骨缺损修复的新型骨植入物。压电材料的优缺点总结见表4。 "

| [1] CHEN R, WANG J, LIU C. Biomaterials act as enhancers of growth factors in bone regeneration. Adv. Funct. Mater. 2016;26(48):8810-8823. [2] 熊莹,许燕,周建平,等.组织工程研究中的电活性生物材料[J].中国组织工程研究,2019,23(34):5223-5330. [3] VASQUEZ-SANCHO F, ABDOLLAHI A, DAMJANOVIC D, et al. Flexoelectricity in bones. Adv Mater. 2018;30(9):1705316.1-1705316.5. [4] KIM D, HAN SA, KIM JH, et al. Biomolecular piezoelectric materials:from amino acids to living tissues. Adv Mater. 2020;32(14):1906989.1-1906989.16. [5] CHERNOZEM RV, SURMENEVA MA, ABALYMOV AA, et al. Piezoelectric hybrid scaffolds mineralized with calcium carbonate for tissue engineering: analysis of local enzyme and small-molecule drug delivery, cell response and antibacterial performance. Mater Sci Eng C Mater Biol Appl. 2021;122:111909.1-111909.10. [6] TANDON B, BLAKER JJ, CARTMELL SH, et al. Piezoelectric materials as stimulatory biomedical materials and scaffolds for bone repair. Acta Biomater. 2018;73(4):1-20. [7] RIBEIRO C, CORREIA DM, RODRIGUES I, et al. In-vivo demonstration of the suitability of piezoelectric stimuli for bone reparation. Materials Letters. 2017; 209(7):118-121. [8] 刘国峰.自供电刺激骨支架的激光增材制造及其性能研究[D].南昌:江西理工大学,2021. [9] BUI TT, SHIN MK, JEE SY, et al. Ferroelectric PVDF nanofiber membrane for high-efficiency PM0.3 air filtration with low air flow resistance. Colloids Surf A Physicochem Eng Asp. 2022;640:128418. [10] RAHMAN T, MARTIN NP, JENKINS JK, et al. Nb2O5, LiNbO3, and (Na, K)NbO3 Thin Films from High-Concentration Aqueous Nb-Polyoxometalates. Inorg Chem. 2022;61(8):3586-3597. [11] WEI H, GENG W, BI K, et al. High-performance piezoelectric-type MEMS vibration sensor based on LiNbO3 single-crystal cantilever beams. Micromachines (Basel). 2022;13(2):329. [12] BASSETT CA, PAWLUK RJ, BECKER RO. Effects of electric currents on bone in vivo. Nature. 1964;204(4959):652-654. [13] CARTER A, POPOWSKI K, CHENG K, et al. Enhancement of bone regeneration through the converse piezoelectric effect, a novel approach for applying mechanical stimulation. Bioelectricity. 2021;3(4):255-271. [14] PARK JB, KELLY BJ, KENNER GH, et al. Piezoelectric ceramic implants:in vivo results. J Biomed Mater Res. 1981;15(1):103-110. [15] CIOFANI G, RICOTTI L, CANALE C, et al. Effects of barium titanate nanoparticles on proliferation and differentiation of rat mesenchymal stem cells. Colloids Surf B Biointerfaces. 2013;102:312-320. [16] LI J, YANG F, LONG Y, et al. Bulk ferroelectric metamaterial with enhanced piezoelectric and biomimetic mechanical properties from additive manufacturing. ACS Nano. 2021;15(9):14903-14914. [17] MAIA FR, BASTOS AR, OLIVEIRA JM. Recent approaches towards bone tissue engineering. Bone. 2021;154:116256. [18] KAO FC, CHIU PY, TSAI TT, et al. The application of nanogenerators and piezoelectricity in osteogenesis. Sci Technol Adv Mater. 2019;20(1):1103-1117. [19] BRADY MA, WALDMAN SD, ETHIER CR. The application of multiple biophysical cues to engineer functional neocartilage for treatment of osteoarthritis. Part I: cellular response. Tissue Eng Part B Rev. 2015;21(1):1-19. [20] JARKOV L, ALLAN SJ, BOWEN C, et al. Piezoelectric materials and systems for tissue engineering and implantable energy harvesting devices for biomedical applications. Int Mater Rev. 2021. doi:10.1080/09506608.2021.1988194. [21] FUKADA E. Piezoelectric properties of biological polymers. Q Rev Biophys. 1983; 16(1):59-87. [22] AHN AC, GRODZINSKY AJ. Relevance of collagen piezoelectricity to “Wolff’s Law”: a critical review. Med Eng Phys. 2009;31(7):733-741. [23] LANG S, TOFAIL S, KHOLKIN A, et al. Ferroelectric polarization in nanocrystalline hydroxyapatite thin films on silicon. Sci Rep. 2013;3(1):1-6. [24] WANG Q, YANG J, ZHANG W, et al. Manufacture and cytotoxicity of a lead-free piezoelectric ceramic as a bone substitutc consolidation of porous lithium sodium potassium niobate by cold isostatic pressing. Int J Oral Sci. 2009;5(2):99-104. [25] HASTINGS GW, MAHMUD FA, Electrical effects in bone. J Biomed Eng. 1988;10(6): 515-521. [26] ZHENG T, HUANG Y, ZHANG X, et al. Mimicking the electrophysiological microenvironment of bone tissue using electroactive materials to promote its regeneration. J Mater Chem B. 2020;8(45):10221-10256. [27] GUO W, TAN C, SHI K, et al. Wireless piezoelectric devices based on electrospun PVDF/BaTiO3 NW nanocomposite fibers for human motion monitoring. Nanoscale. 2018;10(37):17751-17760. [28] RIBEIRO C, SENCADAS V, CORREIA DM, et al. Piezoelectric polymers as biomaterials for tissue engineering applications. Colloids Surf B Biointerfaces. 2015;136(8):46-55. [29] DAMARAJU SM, WU S, JAFFE M, et al. Structural changes in PVDF fibers due to electrospinning and its effect on biological function. Biomed Mater. 2013; 8(4):045007. [30] PARSSINEN J, HAMMAREN H, RAHIKAINEN R, et al. Enhancement of adhesion and promotion of osteogenic differentiation of human adipose stem cells by poled electroactive poly (vinylidene fluoride). J Biomed Mater Res. 2015;103(3):919-928. [31] RODRIGUES MT, GOMES ME, MANO JF, et al . β- membranes induce cellular proliferation and differentiation in static and dynamic conditions. Mater Sci Forum. 2008;587-588:72-76. [32] MARINO A, ROSSON J, GONZALEZ E, et al. Quasi static charge interactions in bone. J Electrost. 1988;21:347-360. [33] ZHANG CG, LIU WW, CAO C, et al. Modulating surface potential by controlling the β phase content in poly (vinylidene fluoridetrifluoroethylene) membranes enhances bone regeneration. Adv Healthc Mater. 2018;7(11):e1701466. [34] RIBEIRO C, PÄRSSINEN J, SENCADAS V, et al. Dynamic piezoelectric stimulation enhances osteogenic differentiation of human adipose stem cells. J Biomed Mater Res A. 2015;103(6):2172-2175. [35] REIS J, FRIAS C, CANTO E CASTRO C, et al. A new piezoelectric actuator induces bone formation in vivo:a preliminary study. Biomed Res Int. 2012;2012:613403. [36] NEUSS S, APEL C, BUTTLER P, et al. Assessment of stem cell/biomaterial combinations for stem cell-based tissue engineering. J Biomater. 2008;29(3):302-313. [37] WEBER N, LEE YS, SHANMUGASUNDARAM S, et al. Characterization and in vitro cytocompatibility of piezoelectric electrospun scaffolds. Acta Biomater. 2010; 6(9):3550-3556. [38] DAMARAJU SM, SHEN Y, ELELE E, et al. Three-dimensional piezoelectric fibrous scaffolds selectively promote mesenchymal stem cell differentiation. J Biomater. 2017;149(9):51-62. [39] DAI XH, HENG BC, BAI YY, et al. Restoration of electrical microenvironment enhances bone regeneration under diabetic conditions by modulating macrophage polarization. Bioact Mater. 2020;6(7):2029-2038. [40] PEREIRA JD, CAMARGO RC, FILHO CJ, et al. Biomaterials from blends of fluoropolymers and corn starchimplant and structural aspects. Mater Sci Eng. 2014;36(12):226-236. [41] LOPES HB, SANTOS TDS, OLIVEIRA FSD, et al. Poly (vinylidene-trifluoroethylene)/barium titanate composite for in vivo support of bone formation. J Biomater Appl. 2014;29(1):104-112. [42] GENCHI GG, SINIBALDI E, CESERACCIU L, et al. Ultrasound-activated piezoelectric P (VDFTrFE)/boron nitride nanotube composite films promote differentiation of human SaOS-2 osteoblast-like cells. Nanomed Nanotechnol Biol Med. 2018;14(7): 2421-2432. [43] PRYADKO A, SURMENEVA MA, SURMENEV RA, et al. Review of hybrid materials based on polyhydroxyalkanoates for tissue engineering applications. Polymers (Basel). 2021;13(11):1738. [44] CHERNOZEM RV, SURMENEVA MA, SHKARINA SN, et al. Piezoelectric 3-D fibrous poly (3-hydroxybutyrate)-based scaffolds ultrasound-mineralized with calcium carbonate for bone tissue engineering: inorganic phase formation, Osteoblast Cell Adhesion, and Proliferation. ACS Appl Mater Interfaces. 2019;11(21):19522-19533. [45] GORODZHA SN, MUSLIMOV, AR, SYROMOTINA, DS, et al. A comparison study between electrospun polycaprolactone and piezoelectric poly (3-hydroxybutyrate-co-3-hydroxyvalerate) scaffolds for bone tissue engineering. Colloids Surf B Biointerfaces. 2017;160(9):48-59. [46] ZHANG S, PRABHAKARAN MP, QIN X, et al. Biocomposite scaffolds for bone regeneration:Role of chitosan and hydroxyapatite within poly-3-hydroxybutyrate-co-3-hydroxyvalerate on mechanical properties and in vitro evaluation. J Mech Behav Biomed Mater. 2015;51(6):88-98. [47] CHERNOZEM RV, GUSELNIKOVA O, SURMENEVA MA, et al. Diazonium chemistry surface treatment of piezoelectric polyhydroxybutyrate scaffolds for enhanced osteoblastic cell growth. Applied Materials Today. 2020;20:100758. [48] NEWMAN B, CHEN P, PAE K, et al. Piezoelectricity in nylon 11. J Appl Phys. 1980; 51(10):5161-5164. [49] WANG H, LI Y, ZUO Y, et al. Biocompatibility and osteogenesis of biomimetic nano-hydroxyapatite/polyamide composite scaffolds for bone tissue engineering. Biomaterials. 2007;28(22):3338-3348. [50] SAWANO M, TAHARA K, ORITA Y, et al. New design of actuator using shear piezoelectricity of a chiral polymer, and prototype device. Poly Int. 2010;59(3): 365-370. [51] IKADA Y, SHIKINAMI Y, HARA Y, et al. Enhancement of bone formation by drawn poly(L-lactide). J Biomed Mater Res. 1996;30(4):553-558. [52] SHIMONO T, MATSUNAGA S, FUKADA E, et al. The effects of piezoelectric poly L lactic acid films in promoting ossification in vivo. In Vivo. 1996;10(5):471-476. [53] CHEN Y, MAK AFT, WANG M, et al. PLLA scaffolds with biomimetic apatite coating and biomimetic apatite/collagen composite coating to enhance osteoblast-like cells attachment and activity. Sur Coat Techn. 2006;201(12):575-580. [54] KO EK, JEONG SI, RIM NG, et al. In Vitro osteogenic differentiation of human mesenchymal stem cells and in vivo bone formation in composite nanofiber meshes. Tissue Eng Part A. 2008;14(12):2105-2119. [55] NEVES NM, KOUYUMDZHIEV A, REIS RL. The morphology, mechanical properties and ageing behavior of porous injection molded starch-based blends for tissue engineering scaffolding, Mater Sci Eng C. 2005:25(1):195-200. [56] BUCHOLZ RW, HENRY S, HENLEY MB. Fixation with bioabsorbable screws for the treatment of fractures of the ankle. J Bone Joint Surg Am Vol. 1994;76(3):319-324. [57] 李丹,许文君,周恒为,等.钙含量对铪钛酸钡陶瓷压电性能的影响[J].伊犁师范学院学报(自然科学版),2020,14(1):28-33. [58] HWANG KS, SONG JE, YANG HS, et al. Effect of poling conditions on growth of calcium phosphate crystal in ferroelectric BaTiO3 ceramics. J Mater Sci Mater Med. 2002;13(1):133-138. [59] YANG C, SONG S, CHEN F, et al. Fabrication of PVDF/BaTiO3/CNT Piezoelectric Energy Harvesters with Bionic Balsa Wood Structures through 3D printing and supercritical carbon dioxide foaming. ACS Appl Mater Interfaces. 2021;13(35): 41723-41734. [60] BAXTER FR, TURNER IG, BOWEN CR, et al. An in vitro study of electrically active hydroxyapatite-barium titanate ceramics using Saos-2 cells. J Mater Sci Mater Med. 2009;20(8):1697-708. [61] BAXTER FR, TURNER IG, BOWENCR et al. The structure and properties of electroceramics for bone graft substitution, Key Eng. Mater. 2008;361-363:99-102. [62] DUBEY AK, THRIVIKRAMAN G, BASU B. Absence of systemic toxicity in mouse model towards BaTiO3 nanoparticulate based eluate treatment. J Mater Sci Mater Med. 2015;26(2):103. [63] BELOTI MM, OLIVEIRA PTD, GIMENES R, et al. In vitro biocompatibility of a novel membrane of the composite poly (vinylidenetrifluoroethylene)/barium titanate. J Biomed Mater Res A. 2006;79(2):282-288. [64] MOHAMMADI M, MOUSAVI SHAEGH SA, ALIBOLANDI M, et al. Micro and nanotechnologies for bone regeneration:Recent advances and emerging designs. J Control Release. 2018;274(1):35-55. [65] LAURENTI M, CAUDA V. ZnO nanostructures for tissue engineering applications. nanomaterials (Basel). 2017;7(11):374. [66] HANLEY C, THURBER A, HANNA C, et al. The influences of cell type and ZnO nanoparticle size on immune cell cytotoxicity and cytokine induction. Nanoscale Res Lett. 2009;4(12):1409-1420. [67] DIKICI BA, DIKICI S, KARAMAN O, et al. The effect of zinc oxide doping on mechanical and biological properties of 3D printed calcium sulfate based scaffolds, Biocybern Biomed Eng. 2017;37(8):733-741. [68] SHRESTHA BK, SHRESTHA S, TIWARI AP. et al. Bio-inspired hybrid scaffold of zinc oxide-functionalized multi-wall carbon nanotubes reinforced polyurethane nanofibers for bone tissue engineering. Mater Des. 2017;133(7):69-81. [69] FELICE B, SANCHEZ MA, SOCCI SC, et al. Controlled degradability of PCL-ZnO nanofibrous scaffolds for bone tissue engineering and their antibacterial activity. Mater Sci Eng C Mater Biol Appl. 2018;93:724-738. [70] MA ZJ, YAMAGUCHI M. Role of endogenous zinc in the enhancement of bone protein synthesis associated with bone growth of newborn rats. J Bone Miner Metab. 2001;19(1):38-44. [71] DU YY, JASON L. GUO, et al. Hierarchically designed bone scaffolds: from internal cues to external stimuli. Biomaterials. 2019;218:119334. [72] CHEN W, YU Z, PANG J, et al. Fabrication of biocompatible potassium sodium niobate piezoelectric ceramic as an electroactive implant. Materials (Basel). 2017;10(4):345. [73] WANG Q, CHEN X, ZHU J, et al. Porous Li-Na-K niobate bonesubstitute ceramics: microstructure and piezoelectric properties. Mater Lett. 2008;62(3):3506-3508. [74] HAKKI SS, BOZKURT BS, HAKKI EE, Boron regulates mineralized tissue-associated proteins in osteoblasts (MC3T3-E1). J Trace Elem Med Biol. 2010;24(4):243-250. [75] YING X, CHENG S, WANG W, et al. Effect of boron on osteogenic differentiation of human bone marrow stromal cells. Biol Trace Elem. Res. 2011;144(1-3):306-315. [76] NAKHMANSON SM, CALZOLARI A, MEUNIER V, et al. Spontaneous polarization and piezoelectricity in boron nitride nanotubes. Phys Rev B. 2003;67(23):235406. [77] LI X, WANG XP, JIANG XF, et al. Boron nitride nanotube-enhanced osteogenic differentiation of mesenchymal stem cells. J Biomed Mater Res B Appl Biomater. 2016;104(2):323-329. [78] ZHI C, BANDO Y, TANG C, et al. Immobilization of proteins on boron nitride nanotubes. J Am Chem Soc. 2005;127(49):17144-17145. [79] NAGARAJAN S, BELAID H, POCHAT-BOHATIER C, et al. Design of boron nitride/gelatin electrospun nanofibers for bone tissue engineering. ACS Appl Mater. Interfaces. 2017;9(39):33695-33706. [80] ZHOU Z, QIAN D, MINARY JM. Molecular mechanism of polarization and piezoelectric effect in super-twisted collagen. ACS Biomater Sci Eng. 2016;102 (10):6-21. [81] EHTERAMI A, KAZEMI M, NAZARI B, et al. Fabrication and characterization of highly porous barium titanate based scaffold coated by Gel/HA nanocomposite with high piezoelectric coefficient for bone tissue engineering applications. J Mech Behav Biomed Mater. 2018;79:195-202. [82] JACOB J, MORE N, KALIA K, et al. Piezoelectric smart biomaterials for bone and cartilage tissue engineering. Inflamm Regen. 2018;38:2. [83] MORE N, KAPUSETTI G. Piezoelectric material - a promising approach for bone and cartilage regeneration. Med Hypotheses. 2017;108(7):10-16. [84] GRIFFIN M, BAYAT A. Electrical stimulation in bone healing:critical analysis by evaluating levels of evidence. Eplasty. 2011;11(11):e34. [85] ZHUANG H, WANG W, SELDES RM, et al. Electrical stimulation induces the level of TGF-β1 mRNA in osteoblastic cells by a mechanism involving calcium/calmodulin pathway. Biochem Biophys Res Commun. 1997;237(2):225-229. [86] WANG Z. Up-regulation of bone morphogenetic proteins in cultured murine bone cells with use of specific electric fields. J Bone Joint Surg Am. 2006;88(5):1053-1066. [87] LEE HS, SADLER SJM, WRIGHT MO, et al. Activation of Integrin-RACK1/PKCα signalling in human articular chondrocyte mechanotransduction. Osteoarthritis Cartilage. 2002;10(11):890-897. [88] SRIRUSSAMEE K, MOBINI S, CASSIDY NJ, et al. Direct electrical stimulation enhances osteogenesis by inducing Bmp2 and Spp1 expressions from macrophages and preosteoblasts. Biotechnol Bioeng. 2019;116(12):3421-3432. |
| [1] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [2] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [3] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [4] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [5] | Zhang Lichen, Chen Liang, Gu Yong. Inorganic ion bionic periosteum regulates immune microenvironment to promote bone repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 346-353. |
| [6] | Dai Xianglin, Zhang Wenfeng, Yao Xijun, Shang Jiaqi, Huang Qiujin, Ren Yifan, Deng Jiupeng. Barium titanate/polylactic acid piezoelectric composite film affects adhesion, proliferation, and osteogenic differentiation of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 367-373. |
| [7] | Chen Feng, Ren Guowu, Zhang Xiaoyun, Chen Yueping, Shi Rusheng. Receptor activator of nuclear factor-kappa B ligand signal transduction mechanism and osteoclast activation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 293-299. |
| [8] | Zhang Jie, Tian Ai. Advances in the signaling pathway of M2 macrophages involved in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 314-321. |
| [9] | Luo Di, Liang Xuezhen, Yan Bozhao, Li Jiacheng, Xu Bo, Li Gang. Mechanism of Bushen Huoxue Capsule in repair of bone defects due to steroid-induced osteonecrosis of the femoral head in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 184-191. |
| [10] | Wei Tengfei, He Xiaoming, Wei Yurou, Zhan Zhiwei, He Mincong, He Wei, Wei Qiushi. Differential expression of Piezo1 in osseous tissue of steroid- and alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 270-275. |
| [11] | Liu Yunling, He Ruya, Nie Minhai, Li Tengyan, Liu Xuqian. Application of concentrated growth factor and epidermal growth factor in the field of oral and maxillofacial soft and hard tissue injury repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 105-113. |
| [12] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [13] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [14] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [15] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||