Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (34): 5546-5553.doi: 10.12307/2023.560
Previous Articles Next Articles
Natural small molecular compounds in the treatment of bone-related diseases by regulating type H blood vessels and its application in tissue engineering
Xu Zhengyi, Wan Qianbing, Chen Junyu
- Department of Prosthodontics, State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2022-09-22Accepted:2022-10-28Online:2023-12-08Published:2023-04-23 -
Contact:Chen Junyu, Associate professor, Master’s supervisor, Department of Prosthodontics, State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Xu Zhengyi, Master candidate, Department of Prosthodontics, State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:National Natural Science Foundation of China, Nos. 81901060, 82270961 (to CJY); Special Funded Project of China Postdoctoral Science Foundation, No. 2021T140483 (to CJY)
CLC Number:
Cite this article
Xu Zhengyi, Wan Qianbing, Chen Junyu. Natural small molecular compounds in the treatment of bone-related diseases by regulating type H blood vessels and its application in tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5546-5553.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

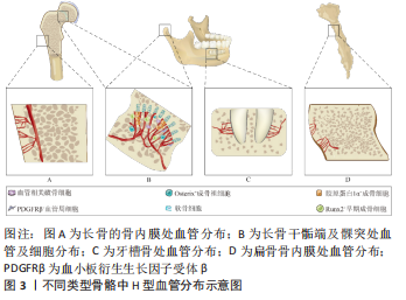
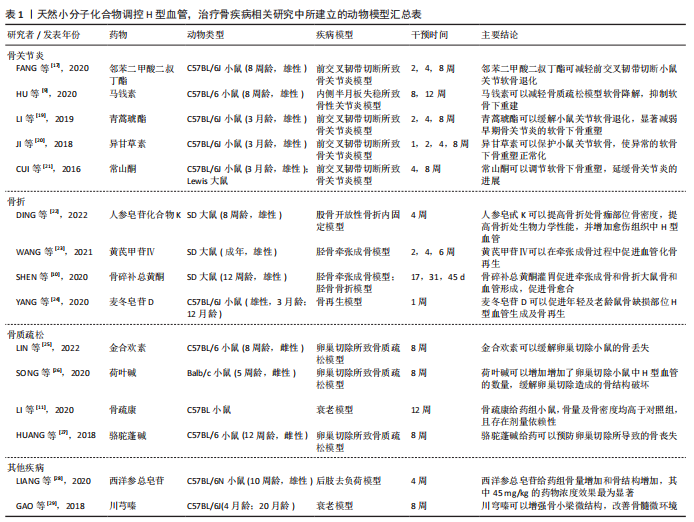
以往研究普遍认为H型血管成线形排列,有独特的柱状结构,在远端由血管弓相互连接[1]。最近一项研究表明,在骨再生过程中产生的H型血管不是柱状排列的,骨折间隙中H型血管的组织结构显得更加杂乱无章,这提示柱状分布这一特征可能不是H型血管所固有的[14],这项研究与先前的发现相符。柱状结构由远端血管弓出芽生成,血管芽带有短的丝状足,并与软骨细胞基质相连[15]。在干骺端软骨细胞高度有序,柱状排列,它们的结构可能在血管芽长入软骨基质时被强加在这些区域的新生H型血管上。 H型血管具有显著的年龄相关性。H型血管在发育中的年轻动物中最为常见,在老年小鼠及人体内数量急剧减少。出生后H型血管的长度和血管芽的丰度随骨骼生长过程而增加。在成年小鼠的长骨中,血管弓仍显著存在,但血管芽的丰度明显减少;老年小鼠的血管弓稀疏,跨度增长,血管芽几乎完全消失,并且CD31hiEMCNhi内皮细胞数量迅速减少,几乎检测不到[1,15]。但在牙槽骨中,虽然H型血管的数量也随年龄的增长而降低,但不同于在胫骨等长骨中,牙槽骨中的H型血管能维持在一个相对恒定的水平,这与牙槽骨的长期重建潜能有关[13]。 这些关于H型血管形态、分布及增龄性变化的研究,有助于更加精准方便地确定出不同年龄段以及不同部位的H型血管。H型血管的时空及形态特征的揭示虽然停留在形态学层面,但是针对不同时空及部位的H型血管,系统性探究其与不同标志物的共定位及与不同血管周细胞的交互作用,对后续有关H型血管的成血管-成骨偶联相关分子机制的研究具有重要意义。 2.1.2 H型血管在偶联成血管成骨中的作用 H型血管在血管生成与成骨两种生理过程的偶联中起重要作用。相比于H型血管,长骨骨干内的窦状血管在骨干的骨髓腔内形成致密的、高度分支的毛细血管网络,CD31和Emcn低表达,称为L型(CD31loEMCNlo)血管。富氧血液从动脉和远端小动脉流入,直接与H型血管相连。然后,血液继续进入干骺端和干骺端交界处的L型血管,最终流入中央静脉。由于与小动脉直接相连,H型血管具有较高的氧气浓度与血液流速[15]。 尽管H型内皮细胞仅占骨骼中全部内皮细胞的1.77%和骨髓内皮细胞总数的0.015%,Osterix+成骨祖细胞、胶原蛋白1α+成骨细胞、大鼠侏儒相关转录因子2+(RUNX family transcription factor 2,Runx2+)早期成骨细胞以及PDGFRβ+血管周细胞选择性地位于H型血管周围,这些细胞在成骨过程中具有重要的调节作用,见图3B。成骨细胞与H型血管空间上的紧密联系,也使其更容易获得氧气及其他营养成分[1]。破骨细胞在干骺端近端主要分布在骨/软骨界面,在远端主要分布在骨小梁表面。近端的破骨细胞直径较小(15-20 μm)只含有一两个细胞核,被定义为血管相关破骨细胞(vessel-associated osteoclasts,VAO)。血管相关破骨细胞对于生长板的吸收必不可少,它通过调节H型远端血管弓的吻合,起到维持骨/软骨界面边界的作用,见图3B。肿瘤坏死因子配体超家族成员11α基因(tnfsf11α),在H型血管内皮细胞中高度表达,参与了H型血管和血管相关破骨细胞之间的RANKL-RANK信号转导。H型血管通过血管相关破骨细胞参与软骨基质的吸收介导骨的定向伸长[16]。 这些关于H型血管调控成骨成血管的机制研究,为以H型血管为靶点治疗骨相关疾病的研究奠定了基础。 2.2 天然小分子化合物通过调控H型血管治疗骨疾病的应用 多种骨骼相关疾病中都可发现H型血管的改变。H型血管已被证明是骨关节炎、骨折及骨质疏松等疾病治疗的潜在治疗位点。通过天然小分子化合物上调或下调H型血管以调控骨生成及骨再生,进而治疗骨骼相关疾病,是一类具有潜力的治疗方法。表1总结了天然小分子化合物调控H型血管,治疗骨疾病相关研究中所建立的动物模型[17-28]。"
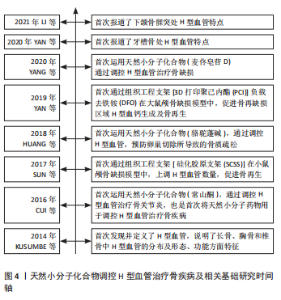
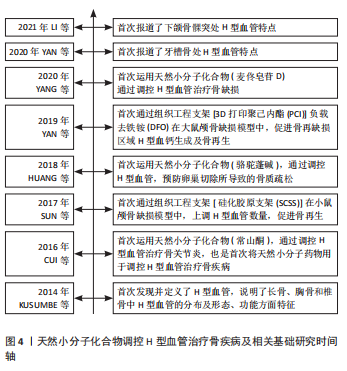
2.2.1 骨关节炎 骨关节炎是世界范围内最常见的多因素退行性关节疾病之一[29-30],目前的干预措施只能暂时缓解临床症状,直到疾病晚期需要关节置换时,才能有效地治疗骨关节炎,因此,迫切需要更多的治疗手段[31-33]。近年来研究表明H型血管在骨关节炎的进展中起着至关重要的作用。来自H型血管的基质金属蛋白酶(matrix metalloproteinase,MMP)可能消化软骨基质且H型血管异常生成受基质金属蛋白酶调控;破骨前细胞异常分泌的血小板衍生生长因子BB(PDGF-BB)通过诱导病理性软骨下骨的形成,促进骨关节炎的发展;转化生长因子β也能促进H型血管的形成[2]。因此,通过天然小分子药物降低H型血管的异常生成,改善关节软骨退化及软骨下骨硬化,进而延缓骨关节炎的发生,是一种有前景的治疗方式。 多数药物以调控MMP生成为主,马钱素(Loganin)是从山茱萸(Corni Fructus,CF)中提取的一种环烯醚萜苷类化合物,具有抗炎镇痛、保护神经、抗骨质疏松等多种药理活性[34]。研究表明,马钱素可增加软骨中Ⅱ型胶原(COL2)的表达,降低基质金属蛋白酶3、基质金属蛋白酶13、Ⅹ型胶原(COL10)、低温蛋白和半胱氨酸的天冬氨酸蛋白水解酶1(Caspase-1)的表达;核转录因子κB信号通路参与了基质金属蛋白酶 和 Ⅹ型胶原的调节[9],基质金属蛋白酶等因子的表达降低,可能与H型血管生成遭到抑制有关。马钱素抑制软骨下骨中H型血管的生成并通过增加透明软骨厚度,减少钙化软骨厚度,并减少骨赘形成,还能减轻软骨基质的分解代谢和关节软骨松弛的发生,从而起到保护软骨的作用。 草本植物的根茎是常用的药物来源,异甘草素(Isiquiritigenin,ISL)与常山酮(Halofuginone,HF)都是从植物的根中提取出的具有抗血管生成作用的药物活性成分。在骨性关节炎早期的异甘草素用药,对前交叉韧带切除小鼠有相当的疗效。异甘草素通过使血管内皮生长因子受体2和基质金属蛋白酶2的过高表达恢复正常,间接抑制转化生长因子β信号通路,抑制软骨下骨中H型血管的形成[20]。常山酮通过间接抑制依赖于基质金属蛋白酶2的血管网络的形成来抑制H型血管生成,减少软骨下的异常充血并延缓前交叉韧带切除大鼠关节软骨退变和软骨下骨硬化,从而延缓骨关节炎的进展[21]。 此外,天然小分子药物还能通过其他途径减少H型血管的异常生成。三七(Panax notoginseng,PN)长期以来一直用于治疗骨质疏松症、骨折和骨关节炎等骨病[35-37]。有研究采用了二维/骨髓单核细胞/细胞膜色谱/飞行时间质谱技术筛选出三七中的活性成分邻苯二甲酸二叔丁酯(diterbutyl phthalate,DP)。在具有前交叉韧带切除 (anterior cruciate ligament transection,ACLT) 的骨关节炎小鼠模型中,邻苯二甲酸二叔丁酯给药阻碍了软骨下骨中过度活化的破骨细胞生成并改善了关节软骨的退化,减弱了软骨下骨髓中异常 H 型血管的形成,并通过抑制软骨下骨中破骨细胞介导的感觉神经支配来减轻关节疼痛相关行为。体外实验表明,邻苯二甲酸二叔丁酯 (20 μmol/L) 通过抑制核转录因子κB受体活化因子配基(receptor activator of nuclear factor-κB ligand,RANKL) 介导的细胞外调节蛋白激酶/原癌基因c-fos/活化T细胞核转录因子1蛋白(ERK/c-fos/NFATc1) 通路,降低破骨细胞融合的两个关键调节剂树突状细胞-特异性跨膜蛋白(DC-STAMP)和 囊泡ATP水解酶V0区d2亚基(Atp6v0d2)来抑制破骨细胞融合来延缓破骨细胞生成[17]。 青蒿琥酯是青蒿素的半合成衍生物之一,近年来,青蒿琥酯除了作为抗疟疾药物外,还因其成骨及抗血管作用而受到越来越多的关注[38-39]。LI等[19]向前交叉韧带切除小鼠每日腹腔注射青蒿琥酯,结果表明青蒿琥酯通过使前交叉韧带切除小鼠体内高水平的血管内皮生长因子A和血管生成素1(ANG-1)恢复正常,并激活血管内皮生长因子β抑制软骨下骨H型血管生成,改善了关节软骨退变和软骨下骨硬化。 2.2.2 骨折 骨折愈合是一个多阶段的复杂修复过程,该修复过程由即刻炎症反应启动,间充质干细胞募集,随后分化为软骨细胞,产生软骨和成骨细胞,软骨基质产生后,矿化的软骨转化为骨[40]。H型血管在软骨基质替换为骨的过程中充当重要角色。已有研究表明,提高骨折小鼠H型血管丰度,可以有效促进骨折愈合[41]。 人参皂甙化合物K (Ginsenoside Compound K,CK)是人参皂甙Rb1,Rb2和Rc通过体内肠道细菌代谢产生的代谢产物,已被证明具有促进成骨细胞分化等多种药理作用[42-43]。在大鼠开放式股骨骨折模型中,人参皂甙化合物K可以促进愈伤组织中H型血管生成,加速大鼠骨折愈合。体外实验表明,人参皂甙化合物K可以激活大鼠骨髓间充质干细胞中无翼集成蛋白 /β-连环蛋白(Wnt/β-catenin)信号通路,增强大鼠骨髓间充质干细胞中成骨标志物mRNA的表达。此外当人脐静脉内皮细胞和骨髓间充质干细胞共培养时,人参皂甙化合物K 增加了血管的形成[22]。麦冬皂苷D(ophiopogonin D,OP-D)是一种潜在的抗氧化剂,可被用于治疗骨质疏松[44]。最近研究表明,麦冬皂苷D也可以增加H型血管丰度,促进骨愈合。体外实验证明麦冬皂苷D可以影响Krüppel样因子3(KLF3)与JUNB启动子的结合,促进H型血管的形成[45]。进一步研究发现,在手术切除松质骨诱导的小鼠骨再生模型中,给予麦冬皂苷D可以增加H型血管数量,促进骨损伤后的骨再生,这为加速骨骼愈合提供了一种潜在的新策略[24]。 牵张成骨是临床上一种独特有效的骨再生技术。骨碎补(Drynaria)的根茎常被用于治疗骨质疏松症和骨折[46]。对骨碎补有效成分的研究主要集中在骨碎补总黄酮(total flavonoids of Rhizoma Drynariae,TFRD)上[47]。骨碎补总黄酮可以通过血小板衍生生长因子BB/血小板衍生生长因子受体β(PDGF-BB/PDGFFR-β)通路促进H型血管形成,从而增强牵张成骨过程中成骨-成血管偶联。此外,在大鼠牵张成骨模型中H型血管多于骨折模型,这提示规则的拉应力可以刺激H型血管的生成[10]。黄芪甲苷 (Astragaloside IV,AS-IV) 通过在RANKL诱导下保留破骨前细胞来增加血小板衍生生长因子的产生进而促进H型血管的生成,这种作用归因于蛋白激酶B/激酶3/β-连环蛋白(AKT/GSK-3β/β-catenin) 通路的激活。体内实验表明黄芪甲苷在牵引成骨过程中加速骨巩固,在牵张区通过促进H型血管生成增强血管化骨再生[23]。 2.2.3 骨质疏松 骨质疏松是世界上最复杂、最普遍的骨骼疾病之一,主要由雌激素缺乏和衰老引起[48-49]。在去卵巢小鼠及老龄小鼠模型中,H型血管显著数量减少,同样在骨质疏松患者骨组织中也发现了类似的结果[1,4,50]。骨质疏松症小鼠模型中,H型血管减少主要由血小板衍生生长因子BB的减少导致,可通过抑制组织蛋白酶K增加前破骨细胞的数量,刺激血小板衍生生长因子BB水平升高,从而刺激去卵巢小鼠H型血管生成[51]。 目前多种天然小分子化合物可以通过保护前破骨细胞,抑制多核破骨细胞的成熟,从而促进H型血管生成与骨再生。金合欢素(acacetin)是一种天然黄酮类化合物,先前研究表明它在体外可促进成骨细胞分化、抑制破骨细胞形成[52-53]。金合欢素可以减少多核破骨细胞的数量,并增加 血小板衍生生长因子BB+/抗酒石酸酸性磷酸酶+(PDGF-BB+/TRAP+)前破骨细胞的数量,从而刺激去卵巢小鼠的H血管生成和骨形成。机制层面,金合欢素不仅抑制 RANKL 诱导的破骨细胞形成过程中主要转录因子活化T-细胞核转录因子1(NFATc1) 和核转录因子κB的表达,而且抑制RANKL诱导的蛋白激酶B(Akt)、糖原合成酶激酶3β(GSK3β)、核转录因子κB抑制因子α(IκBα) 和核转录因子κB p65亚基的磷酸化,以此减弱了破骨细胞生成[25]。荷叶碱(Nuciferine,NCF)与骆驼蓬碱(Harmine)也被证明可以通过抑制多核破骨细胞的成熟与活性并促进H型血管生成。荷叶碱通过抑制丝裂原活化蛋白激酶/核转录因子κB/原癌基因c-fos/活化T细胞核转录因子1蛋白(MAPK/NF-κB/c-Fos/NFATc1)信号通路上调血小板衍生生长因子BB,抑制破骨细胞成熟[26-27]。而骆驼蓬碱上调PDGF-BB的机制仍待进一步研究。 骨疏康是一种传统的复方中药,常用于治疗骨质疏松[54]。体内实验结果证明,骨疏康处理后的小鼠H型血管更多;蛋白质印迹分析以及实时荧光定量-聚合酶链式反应(qRT-PCR)都表明,治疗组的缺氧诱导因子1α同样高于对照组。提示骨疏康可能通过增强缺氧诱导因子1α的表达而诱导H型血管的生成与骨生成[11]。 2.2.4 其他疾病 除上述几种常见骨疾病外,天然药物小分子在治疗衰老所引起的退行性骨丧失、治疗失重性骨丢失中也受到了研究者的关注。 H型血管在衰老的小鼠和人类长骨中的丰度大幅下降[1,13]。而衰老是导致人类骨骼退行性疾病和功能缺陷的主要病理因素之一。因此能否通过促进H型血管生成,改善衰老条件下骨疾病的发生与预后引起了研究者的关注。川芎嗪(Tetramethylpyrazine,TMP)是从川穹根茎中提取纯化的主要活性成分[55-56]。局部注射川芎嗪可以诱导CD31hiEmcnhi内皮细胞生成,显著提高小鼠体内血管内皮生长因子和血小板衍生生长因子BB水平。进一步研究证实,川芎嗪通过单磷酸腺苷依赖的蛋白激酶哺乳动物雷帕霉素靶蛋白-缺氧诱导因子1α-血管内皮生长因子(AMPK-mTOR-HIF1α-VEGF)途径诱导CD31hiEmcnhi内皮细胞形成管状结构及血管芽,进而生成H型血管,改善衰老骨髓的微环境[29]。 失重性骨丢失是一种由长期卧床或太空飞行所导致的骨生成与骨吸收失衡,最后可能导致骨质疏松性骨折,是宇航员所面临的主要健康问题之一。尽管在国际空间站上配备有跑步机、高级阻力锻炼装置等器械供宇航员日常使用,但这种刺激并不能有效减少宇航员的骨萎缩[57-58]。鉴于H型血管在血管成骨及血管生成中的耦合作用,H型血管也可能成为治疗失重性骨丢失的潜在靶点。西洋参皂苷是从西洋参茎叶中提取的一种主要成分,已被证明具有促进心肌血管生成等功能[59]。体内实验证明,后肢去负荷小鼠的骨形成与H型血管同时减少,而西洋参皂苷可以增加通过上调血管内皮生长因子和头蛋白(Noggin)的分泌促进H型血管生成,提升成骨细胞功能,进而促进后肢去负荷小鼠的骨生成[28]。 综上所述,天然小分子化合物可以通过多种信号通路调控H型血管,其主要调控对象包括核转录因子κB、β-连环蛋白和血小板衍生生长因子BB等。在体外实验中可以有效地促进人脐静脉内皮细胞和骨髓间充质干细胞增殖与分化,在体内实验部分关于天然小分子药物促进或者抑制H型血管的结果和程度具有差异性,而且不同研究中的对于H型血管的免疫荧光的结果呈现和定性分析存在质量的差异性,此部分值得研究者进一步进行高质量的H型血管免疫荧光检测。此外,H型血管在神经、免疫调节也被证实发挥作用[14,60],许多天然小分子都具有安神及抗炎的功效,能否通过其调控神经免疫系统,调节H型血管生成,进而治疗骨疾病,也需要进一步研究。 2.3 天然小分子与组织工程支架调控H型血管的应用现状与不足 目前,有关应用组织工程支架负载天然小分子化合物调控H型血管治疗骨疾病的研究较少,但应用组织工程支架负载天然小分子化合物治疗骨疾病亦或是搭载其他药物通过调控H型血管治疗骨疾病已经取得较好效果。这些研究为后续组织工程支架与天然小分子药物协同调控H型血管奠定了基础。 2.3.1 天然小分子与组织工程支架治疗骨疾病的应用 天然小分子在治疗骨质疏松等疾病中具有优势,但传统给药方式包括口服给药、局部给药与全身给药,均存在不同程度缺陷。组织工程学可以提高局部药物浓度、维持药物释放等,天然小分子药物也可以调节支架降解速度,改善其力学性能等,故将二者结合在一起提供了一种较好的替代方案。 在不同疾病模型中天然小分子与组织工程支架相结合,可以显著促进缺损处的血管形成与骨再生。大鼠股骨缺损模型证实,在建模后12周的时间里,负载较高浓度天麻素(Gastrodin)的支架可以有效促进大鼠股骨髁状突骨缺损模型中血管骨组织的再生[61]。Zhao等[62]分别将含有与不含有柚皮苷(Naringin)的复合丝素蛋白/羟基磷灰石支架应用于大白兔股骨缺损模型,术后4周,非载药组持续存在骨缺损,而载药组出现骨组织充填及矿化;术后8周载药组骨缺损处可见新生骨,而对照组的骨缺损处仅见少量再生骨。在骨质疏松这种复杂的病理环境中,将续断负载于3D打印支架中,可以加速骨质疏松兔中临界骨缺损处的骨再生[63] 。 以上研究证明了将组织工程支架负载天然小分子化合物,对于治疗不同类型骨疾病具有良好的效果。 2.3.2 搭载药物的组织工程支架调控H型血管治疗骨疾病的应用 组织工程在血管化骨再生方面具有巨大的潜力。由于H型血管在偶联成骨和成血管方面的功能特性,研究者们设计了多种支架负载药物或细胞因子通过调控H型血管治疗骨相关疾病。缺氧诱导因子1α是调控成血管-成骨两生理过程的重要刺激因子。负载去铁铵3D打印的聚己内酯支架通过缺氧诱导因子1α信号通路促进血管内皮生长因子的表达,促进H型血管生成且具有类似于天然松质骨的力学性能,体内实验也证明了该支架可促进大鼠骨缺损模型中血管的生长并促进缺损区的骨再生[64]。 有研究通过将负载伐他汀的β-环糊精接枝到壳聚糖和明胶上,在纯钛表面构建了具有成骨和血管生成功能的多层膜。体外实验表明,局部应用匹伐他汀可显著增强间充质干细胞的成骨能力和内皮细胞的血管生成能力。大鼠股骨圆柱形缺损模型证明,经过表面改性的载药种植体可以将内源性干细胞以及血管内皮细胞动员到种植体-骨界面进而促进H型血管生成及骨再生,最终促进种植体周围骨整合[7]。 以上研究证明了组织工程支架在调控H型血管治疗骨疾病的应用具有良好前景。上文中所提及的人参皂苷化合物K、黄芪甲苷、骨碎补总黄酮和麦冬皂苷D均可以通过调控H型血管促进骨折处血管生成与骨再生;金合欢素、荷叶碱、骨疏康及骆驼蓬碱对骨质疏松都具有较好的调节作用。将这些药物负载于组织工程支架上,用以调控H型血管,治疗特定情境下的骨缺损等骨疾病,将成为一种具有良好前景的新方法。 文章总结了有关H型血管、天然小分子药物调控H型血管治疗骨疾病以及组织工程学支架载药调控H型血管的相关研究进展时间线,见图4。"
| [1] KUSUMBE AP, RAMASAMY SK, ADAMS RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014;507(7492):323-328. [2] CHEN JY, HENDRIKS M, CHATZIS A, et al. Bone vasculature and bone marrow vascular niches in health and disease. J Bone Miner Res. 2020;35(11):2103-2120. [3] WANG J, GAO Y, CHENG P, et al. CD31hiEmcnhi vessels support new trabecular bone formation at the frontier growth area in the bone defect repair process. Sci Rep. 2017;7(1):4990. [4] YIN H, HUANG J, CAO X, et al. Inhibition of src homology 2 domain-containing protein tyrosine phosphatase-2 facilitates CD31hiEndomucinhi blood vessel and bone formation in ovariectomized mice. Cell Physiol Biochem. 2018;50(3):1068-1083. [5] YIN S, ZHANG W, ZHANG Z, et al. Recent advances in scaffold design and material for vascularized tissue-engineered bone regeneration. Adv Healthc Mater. 2019; 8(10):e1801433. [6] DAI K, SHEN T, YU Y, et al. Generation of rhBMP-2-induced juvenile ossicles in aged mice. Biomaterials. 2020;258:120284. [7] CHEN W, XIE G, LU Y, et al. An improved osseointegration of metal implants by pitavastatin loaded multilayer films with osteogenic and angiogenic properties. Biomaterials. 2022;280:121260. [8] SUN J, JIAO K, NIU L, et al. Intrafibrillar silicified collagen scaffold modulates monocyte to promote cell homing, angiogenesis and bone regeneration. Biomaterials. 2017;113:203-216. [9] HU J, ZHOU J, WU J, et al. Loganin ameliorates cartilage degeneration and osteoarthritis development in an osteoarthritis mouse model through inhibition of NF-κB activity and pyroptosis in chondrocytes. J Ethnopharmacol. 2020;247:112261. [10] SHEN Z, CHEN Z, LI Z, et al. Total flavonoids of rhizoma drynariae enhances angiogenic-osteogenic coupling during distraction osteogenesis by promoting type H vessel formation through PDGF-BB/PDGFR-β instead of HIF-1α/VEGF axis. Front Pharmacol. 2020;11:503524. [11] LI W, ZHOU X, JIANG T, et al. Positive effect of gushukang on type-H vessel and bone formation. Front Cell Dev Biol. 2020;8:265. [12] LI H, LIAO L, Hu Y, et al. Identification of type H vessels in mice mandibular condyle. J Dent Res. 2021;100(9):983-992. [13] YAN Z, WANG X, ZHOU Y, et al. H-type blood vessels participate in alveolar bone remodeling during murine tooth extraction healing. Oral Dis. 2020;26(5):998-1009. [14] STEFANOWSKI J, LANG A, RAUCH A, et al. Spatial distribution of macrophages during callus formation and maturation reveals close crosstalk between macrophages and newly forming vessels. Front Immunol. 2019;10(2588):2588-2588. [15] RAMASAMY SK, KUSUMBE AP, Schiller M, et al. Blood flow controls bone vascular function and osteogenesis. Nat Commun. 2016;7:13601-13601. [16] ROMEO S, ALAWI K, RODRIGUES J, et al. Endothelial proteolytic activity and interaction with non-resorbing osteoclasts mediate bone elongation. Nat Cell Biol. 2019;21(4):430-441. [17] PENG Y, WU S, LI Y, et al. Type H blood vessels in bone modeling and remodeling. Theranostics. 2020;10(1):426-436. [18] FANG C, GUO JW, WANG YJ, et al. Diterbutyl phthalate attenuates osteoarthritis in ACLT mice via suppressing ERK/c-fos/NFATc1 pathway, and subsequently inhibiting subchondral osteoclast fusion. Acta Pharmacologica Sinica. 2022;43(5):1299-1310. [19] LI Y, MU W, XU B, et al. Artesunate, an anti-malaria agent, attenuates experimental osteoarthritis by inhibiting bone resorption and CD31hiEmcnhi vessel formation in subchondral bone. Front Pharmacol. 2019;10:685. [20] JI B, ZHANG Z, GUO W, et al. Isoliquiritigenin blunts osteoarthritis by inhibition of bone resorption and angiogenesis in subchondral bone. Sci Rep. 2018;8(1):1721. [21] CUI Z, CRANE J, XIE H, et al. Halofuginone attenuates osteoarthritis by inhibition of TGF-β activity and H-type vessel formation in subchondral bone. Ann Rheum Dis. 2016;75(9):1714-1721. [22] DING L, GU S, ZHOU B, et al. Ginsenoside compound K enhances fracture healing via promoting osteogenesis and angiogenesis. Front Pharmacol. 2022;13: 855393-855393. [23] WANG F, QIAN H, KONG L, et al. Accelerated bone regeneration by astragaloside iv through stimulating the coupling of osteogenesis and angiogenesis. Int J Biol Sci. 2021;17(7):1821-1836. [24] YANG M, LI C, XIAO Y, et al. Ophiopogonin D promotes bone regeneration by stimulating CD31 EMCN vessel formation. Cell Prolif. 2020;53(3):e12784. [25] LIN X, XU F, ZHANG KW, et al. Acacetin prevents bone loss by disrupting osteoclast formation and promoting type H vessel formation in ovariectomy-induced osteoporosis. Front Cell Dev Biol. 2022;10:796227. [26] SONG C, CAO J, LEI Y, et al. Nuciferine prevents bone loss by disrupting multinucleated osteoclast formation and promoting type H vessel formation. FASEB J. 2020;34(3):4798-4811. [27] HUANG J, YIN H, RAO S, et al. Harmine enhances type H vessel formation and prevents bone loss in ovariectomized mice. Theranostics. 2018;8(9):2435-2446. [28] LIANG S, LING S, DU R, et al. The coupling of reduced type H vessels with unloading-induced bone loss and the protection role of Panax quinquefolium saponin in the male mice. Bone. 2021;143:115712. [29] GAO B, LIN X, JING H, et al. Local delivery of tetramethylpyrazine eliminates the senescent phenotype of bone marrow mesenchymal stromal cells and creates an anti-inflammatory and angiogenic environment in aging mice. Aging cell. 2018; 17(3):e12741. [30] HOOTMAN J, HELMICK C. Projections of US prevalence of arthritis and associated activity limitations. Arthritis Rheum. 2006;54(1):226-229. [31] LAWRENCE R, FELSON D, HELMICK C, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35. [32] LE GRAVERAND-GASTINEAU M. Disease modifying osteoarthritis drugs: facing development challenges and choosing molecular targets. Current drug targets, 2010;11(5):528-535. [33] HAWKER G, MIAN S, BEDNIS K, et al. Osteoarthritis year 2010 in review: non-pharmacologic therapy. Osteoarthritis Cartilage. 2011;19(4):366-374. [34] BERENBAUM F. Osteoarthritis year 2010 in review: non-pharmacological therapies. Osteoarthritis Cartilage. 2011;19(4):361-365. [35] HUANG J, ZHANG Y, DONG L, et al. Ethnopharmacology, phytochemistry, and pharmacology of Cornus officinalis Sieb. et Zucc. J Ethnopharmacol. 2018;213: 280-301. [36] WEI CC, YUE LF, YOU FT, et al. Panax notoginseng saponins alleviate osteoporosis and joint destruction in rabbits with antigen‑induced arthritis. Exp Ther Med. 2021;22(5):1302. [37] 李世杰,马立琼,熊贤梅,等.三七总皂对富血小板血浆促进兔骨缺损愈合的影响[J].中国组织工程研究,2022,26(14):2155-2160. [38] ZHANG Y, CAI W, HAN G, et al. Panax notoginseng saponins prevent senescence and inhibit apoptosis by regulating the PI3K‑AKT‑mTOR pathway in osteoarthritic chondrocytes. Int J Mol Med. 2020;45(4):1225-1236. [39] VERMA S, DAS P, KUMAR V L. Chemoprevention by artesunate in a preclinical model of colorectal cancer involves down regulation of β-catenin, suppression of angiogenesis, cellular proliferation and induction of apoptosis. Chem Biol Interact. 2017;278:84-91. [40] ZHAO C, LIU Q, WANG K. Artesunate attenuates ACLT-induced osteoarthritis by suppressing osteoclastogenesis and aberrant angiogenesis. Biomed Pharmacother. 2017;96:410-416. [41] FAZZALARI NL. Bone fracture and bone fracture repair. Osteoporos Int. 2011; 22(6):2003-2006. [42] YANG XD, YANG YY, OUYANG DS, et al. A review of biotransformation and pharmacology of ginsenoside compound K. Fitoterapia. 2015;100:208-220. [43] YANG N, LIU D, ZHANG X, et al. Effects of ginsenosides on bone remodelling for novel drug applications: a review. Chin Med. 2020;15(1):42. [44] HUANG Q, GAO B, WANG L, et al. Ophiopogonin D: a new herbal agent against osteoporosis. Bone. 2015;74:18-28. [45] YANG M, GUO Q, PENG H, et al. Krüppel-like factor 3 inhibition by mutated lncRNA Reg1cp results in human high bone mass syndrome. J Exp Med. 2019; 216(8):1944-1964. [46] FANG Y, QINGNA L, ZHIHONG T, et al. Effect of total flavonoids from Drynaria rhizome on bone loss in ovariectomized rats. Trop J Pharm Res. 2019;18(6):1285-1289. [47] ZHANG Y, JIANG J, SHEN H, et al. Total flavonoids from Rhizoma Drynariae (Gusuibu) for treating osteoporotic fractures: implication in clinical practice. Drug Des Devel Ther. 2017;11:1881-1890. [48] MAO L, XIA L, CHANG J, et al. The synergistic effects of Sr and Si bioactive ions on osteogenesis, osteoclastogenesis and angiogenesis for osteoporotic bone regeneration. Acta Biomater. 2017;61:217-232. [49] SUN X, GUO Q, WEI W, et al. Current progress on microRNA-based gene delivery in the treatment of osteoporosis and osteoporotic fracture. Int J Endocrinol. 2019;2019:6782653. [50] DING W, XU C, ZHANG Y, et al. Advances in the understanding of the role of type-H vessels in the pathogenesis of osteoporosis. Arch Osteoporos. 2020;15(1):5. [51] XIE H, CUI Z, WANG L, et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat Med. 2014;20(11):1270-1278. [52] LI J, LIN X, ZHANG Y, et al. Preparative purification of bioactive compounds from flos chrysanthemi indici and evaluation of its antiosteoporosis effect. Evid Based Complement Alternat Med. 2016;2016:2587201. [53] KIM SI, KIM YH, KANG BG, et al. Linarin and its aglycone acacetin abrogate actin ring formation and focal contact to bone matrix of bone-resorbing osteoclasts through inhibition of αvβ3 integrin and core-linked CD44. Phytomedicine. 2020; 79:153351. [54] CHAI S, WAN L, WANG JL, et al. Gushukang inhibits osteocyte apoptosis and enhances BMP-2/Smads signaling pathway in ovariectomized rats. Phytomedicine. 2019;64:153063. [55] GONG P, ZHANG Z, ZOU Y, et al. Tetramethylpyrazine attenuates blood-brain barrier disruption in ischemia/reperfusion injury through the JAK/STAT signaling pathway. Eur J Pharmacol. 2019;854:289-297. [56] ZHANG X, DONG H, LIU Y, et al. Tetramethylpyrazine partially relieves hypoxia-caused damage of cardiomyocytes H9c2 by downregulation of miR-449a. J Cell Physiol. 2019. doi: 10.1002/jcp.28151. [57] GABEL L, LIPHARDT A, HULME P, et al. Pre-flight exercise and bone metabolism predict unloading-induced bone loss due to spaceflight. Br J Sports Med. 2021; 56(4):196-203. [58] WANG K, WANG Y, HU Z, et al. Bone-targeted lncRNA OGRU alleviates unloading-induced bone loss via miR-320-3p/Hoxa10 axis. Cell Death Dis. 2020;11(5):382. [59] LI D, LIU M, TAO T Q, et al. Panax quinquefolium saponin attenuates cardiomyocyte apoptosis and opening of the mitochondrial permeability transition pore in a rat model of ischemia/reperfusion. Cell Physiol Biochem. 2014;34(4):1413-1426. [60] CHEN H, HU B, LV X, et al. Prostaglandin E2 mediates sensory nerve regulation of bone homeostasis. N Nat Commun. 2019;10(1):181. [61] LI L, LI Q, GUI L, et al. Sequential gastrodin release PU/n-HA composite scaffolds reprogram macrophages for improved osteogenesis and angiogenesis. Bioact Mater. 2022;19:24-37. [62] ZHAO ZH, MA XL, ZHAO B, et al. Naringin-inlaid silk fibroin/hydroxyapatite scaffold enhances human umbilical cord-derived mesenchymal stem cell-based bone regeneration. Cell Prolif. 2021; 54(7):e13043. [63] KAO CT, CHIU YC, LEE AK, et al. The synergistic effects of Xu Duan combined Sr-contained calcium silicate/poly-ε-caprolactone scaffolds for the promotion of osteogenesis marker expression and the induction of bone regeneration in osteoporosis. Mater Sci Eng C Mater Biol Appl. 2021;119:111629. [64] YAN Y, CHEN H, ZHANG H, et al. Vascularized 3D printed scaffolds for promoting bone regeneration. Biomaterials. 2019;190-191:97-110. [65] 李高志,石菲,张舒,等.血管新生与骨形成耦联、骨骼疾病发生及治疗中H型血管的作用机制研究进展[J].山东医药,2021,61(3):91-94. |
| [1] | Jiang Xiaocheng, Shi Lu, Wang Yinbin, Li Qiujiang, Xi Chuangzhen, Ma Zefeng, Cai Lijun. Systematical evaluation of bone fusion rate after interbody fusion in patients with osteoporosis and lumbar degenerative disease treated with teriparatide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1427-1433. |
| [2] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [5] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [6] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [7] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [8] | Long Yanming, Xie Mengsheng, Huang Jiajie, Xue Wenli, Rong Hui, Li Xiaojie. Casein kinase 2-interaction protein-1 regulates the osteogenic ability of bone marrow mesenchymal stem cells in osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 878-882. |
| [9] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [10] | Wang Jinling, Huang Xiarong, Qu Mengjian, Huang Fujin, Yin Lingwei, Zhong Peirui, Liu Jin, Sun Guanghua, Liao Yang, Zhou Jun. Effects of exercise training on bone mass and bone microstructure in aged osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 676-682. |
| [11] | Yuan Changshen, Guan Yanbing, Li Zhe, Rong Weiming, Liao Shuning, Chen Lewei, Mei Qijie, Duan Kan. Screening and verification of key genes of necroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 695-700. |
| [12] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [13] | Shao Zichen, Li Huanan, Gu Bing, Zhang Xiaoyun, Sun Weikang, Liu Yongqian, Gan Bin. MicroRNA, long non-coding RNA and circular RNA mediate the mechanism of decreasing uric acid, anti-inflammation and regulating bone metabolism in gout [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 765-771. |
| [14] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| [15] | Wan Guoli, Shi Chenhui, Wang Weishan, Li Ang, Shi Xunda, Cai Yi. Retrospective analysis of the influencing factors of chronic pain after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 558-564. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||