Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (30): 4876-4882.doi: 10.12307/2023.503
Previous Articles Next Articles
Functional characteristics of three-dimensional biological scaffolds for repairing injured spinal cord
Jiu Jingwei1, 2, Liu Haifeng1, 2, Wang Guishan2, Li Dijun2, Yan Lei1, 2, Zhao Bin1, 2, Wang Bin2, 3
- 1Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China; 2Shanxi Key Laboratory of Bone and Soft Tissue Repair, Taiyuan 030000, Shanxi Province, China; 3Department of Orthopedics, First Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou 310006, Zhejiang Province, China
-
Received:2022-06-06Accepted:2022-08-24Online:2023-10-28Published:2023-04-03 -
Contact:Zhao Bin, MD, Chief physician, Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China; Shanxi Key Laboratory of Bone and Soft Tissue Repair, Taiyuan 030000, Shanxi Province, China Wang Bin, PhD, Associate chief physician, Shanxi Key Laboratory of Bone and Soft Tissue Repair, Taiyuan 030000, Shanxi Province, China; Department of Orthopedics, First Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou 310006, Zhejiang Province, China -
About author:Jiu Jingwei, Master candidate, Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China; Shanxi Key Laboratory of Bone and Soft Tissue Repair, Taiyuan 030000, Shanxi Province, China Liu Haifeng, Doctoral candidate, Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China; Shanxi Key Laboratory of Bone and Soft Tissue Repair, Taiyuan 030000, Shanxi Province, China -
Supported by:the Central Government Guides Local Science and Technology Development Funds, No. YDZX20201400001738 (to WB); National Natural Science Foundation of China, No. 81802204 (to WB); Major Scientific and Technological Research Special Project of Shanxi Provincial Health Commission, No. 2020XM11 (to ZB)
CLC Number:
Cite this article
Jiu Jingwei, Liu Haifeng, Wang Guishan, Li Dijun, Yan Lei, Zhao Bin, Wang Bin. Functional characteristics of three-dimensional biological scaffolds for repairing injured spinal cord[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4876-4882.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
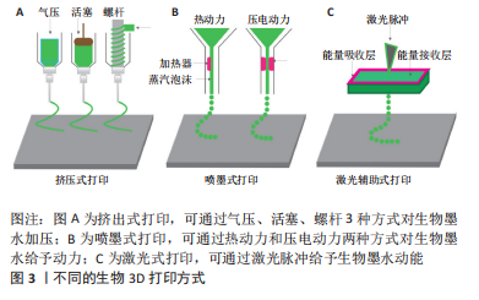
2.1 生物3D打印 2.1.1 生物3D打印的概念及优势 生物3D打印技术由3D打印技术衍生而出,该技术使用人工生物材料、生长因子、活细胞、生物复合细胞材料等活性成分构建出具有一定结构和生物功能的人工组织和器官,其主要目的是用于患者组织或器官的修复与替代[8]。 生物3D打印技术可以针对组织工程应用以生物材料模拟细胞外基质,与特定细胞混合制成生物墨水打印成3D生物支架,经体外培养后植入体内,诱导组织再生与修复[9-10]。生物3D打印技术拥有可定制性和高度灵活性,可以更好地模拟受损脊髓,复现组织的原有功能和结构,在组织工程支架构建上体现出无可比拟的优势[7]。 2.1.2 生物3D打印的主要技术方式 生物3D打印技术主要有微挤压式、喷墨式、激光辅助式以及光固化成型法[11],见图3。根据生物3D打印的细胞活性、生物材料的特性及打印精度等以及这些技术的特点,可以考虑选择更合适的打印方式。"
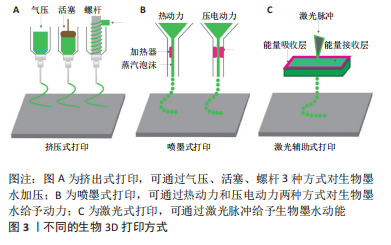
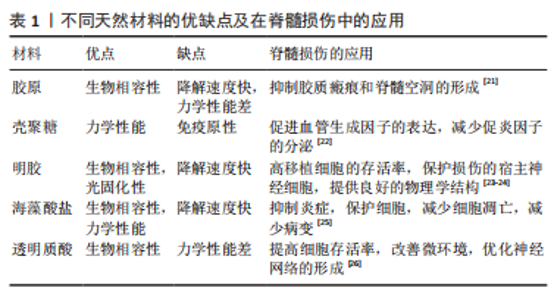
挤出式生物打印是最常用的打印方式,相比于其他3D打印技术,它具有较高的准确性、更快的打印速度、更多的生物材料选择和更低的成本等优势,但是由于挤压更容易对细胞造成损害、导致细胞的存活率下降是其需要解决的主要问题。喷墨3D打印技术的特点是滴落成型,其生物材料需在液态下形成滴落,黏度过低时支架不易成型,黏度过高会堵塞喷头。激光辅助生物打印的优势在于精度较高,可以对高密度细胞样品中细胞的位置进行精准打印,但是成本相对较高,难以原位印刷。光固化成型法则是利用紫外线、红外线或其他光使光敏材料光固化,通过光固化使支架成型而不塌陷,常用于辅助其他3种打印方法[12]。 2.2 不同材料对脊髓损伤后组织修复的影响 目前应用于组织修复及再生领域的材料主要分为两类:天然聚合物(明胶、胶原等)和合成聚合物(聚乙二醇及聚乳酸等)[13-14]。天然生物材料的优势在于它们拥有良好的生物活性且与人类细胞外基质相似,缺点在于机械结构难以控制而使支架容易塌陷。合成材料的优势在于能够根据不同组织与器官的需求来选择特定性能的材料,例如加入导电材料模拟脊髓组织的电生理信号[15],但是在使用合成聚合物的过程中会面临生物相容性差、在降解过程中机械性能下降以及毒性降解产物多等问题。 在组织再生领域,理想的生物打印材料应具备可打印性、生物相容性、降解性、结构与机械性能和仿生学特性等[16-20]。每种材料都有优缺点,根据损伤组织的结构和功能,选择更合适的材料以替代原有组织是3D生物支架应用于脊髓损伤治疗的一大难题。 2.2.1 天然材料 在脊髓损伤的修复中,较常使用的生物墨水材料有明胶、胶原、海藻酸盐、透明质酸和壳聚糖等[21-26],见表1。它们在不同方面拥有着不同的特性,实验中常使用其中一种或多种天然聚合物相互搭配,有时也会根据特定功能的需要添加合成聚合物。"
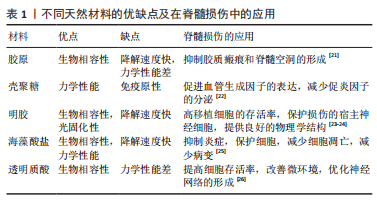
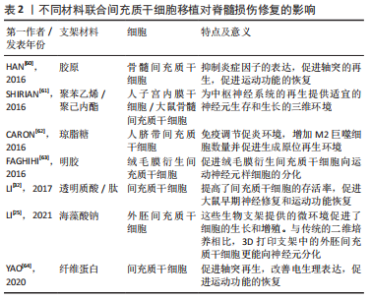
胶原:是一种常用的天然组织工程材料,是细胞外基质的重要组成成分[27-28]。胶原具有良好的生物相容性,并且免疫原性较低,在体内可以降解,不会引起不良炎症反应。但是在传统生物支架移植中对运动功能的恢复并不明显,并且支架的力学结构较为不稳定,容易塌陷,致使引导轴突的腔管结构难以维持[21,29]。SUN等[22]使用挤压式打印制备了用于脊髓损伤后组织修复的3D生物支架,他们的研究使用了一种由胶原和壳聚糖组成的负载有神经干细胞的生物墨水,该墨水利用壳聚糖弥补胶原力学性能的不足,具有比单纯胶原支架更好的机械强度,形成的支架不易塌陷。他们利用大鼠第8胸椎完全横断模型研究了支架的性能,结果表明胶原3D生物支架不仅可以促进神经纤维的再生、减少脊髓空洞和胶质瘢痕的形成,而且可以有效改善运动功能的恢复。 明胶:是胶原降解后的一种产物,来源十分广泛。明胶的免疫原性比胶原更低,具有良好的生物相容性和可降解性。明胶可以使细胞维持较高的活力,保持较高的细胞存活率。明胶的修饰产物——甲基丙烯酸酰化明胶(Gelatin Methacrylamide,GelMA)因具有细胞黏附和基质金属蛋白酶反应肽基的存在而更有利于细胞的增殖和扩散[30]。WANG等[23]采用挤压法制备了一种同时载有施万细胞和间充质干细胞并将其分层打印的GelMA支架,在体外培养14 d后,3D支架组的细胞存活率高达95%,细胞数量相比较于普通三维支架组也有明显增多,结果表明GelMA组成的3D支架相比较于普通三维支架更利于细胞的存活和增殖。 海藻酸盐:具有良好的生物相容性和可吸收性,是一个较为理想的三维支架材料,但是在体内的降解速率较快。脊髓损伤后微环境的改变是造成继发性损害的关键因素,有研究表明海藻酸盐被用于脊髓损伤修复时可以起到较好的抗凋亡和抗炎作用[31]。LI等[25]开发了含有海藻酸盐的复合3D生物支架,通过体外研究发现来自SD大鼠的外间充质干细胞不仅没有不良的炎症反应,而且拥有比传统支架更高的神经分化效率。这表明含有海藻酸盐的3D生物支架有可能通过抑制脊髓损伤后炎性微环境的改变而促进神经的再生,修复受损的脊髓。 透明质酸:是生物3D打印的重要生物材料,是一种天然的细胞外基质成分。有研究表明负载有间充质干细胞的透明质酸生物支架可以在脊髓损伤节段形成有序的神经纤维序列[32]。但是将透明质酸用于生物3D打印制作出的3D支架时[12],不仅可以促进神经纤维的生成和干细胞向神经分化,更可以通过3D支架中稳固的腔管通道促进轴突的再生,形成具有一定三维结构的神经网络。 壳聚糖:具有良好的力学性能,可以弥补胶原、明胶等机械性能差的材料的不足[33-34]。壳聚糖支架用于脊髓损伤移植时常可以促进血管的重建,这对于脊髓损伤后缺血而引发的继发损伤具有明显的疗效[35]。LIU等[36]将一种包含脑源性神经营养因子的壳聚糖胶原3D生物支架移植入大鼠脊髓损伤模型中,发现支架可以维持脑源性神经营养因子的缓慢释放,而且减少了炎症因子的分泌,在损伤节段形成了类似于血管的管腔样结构,实现了促进神经再生、改善功能恢复的效果。 上述单一材料虽各具优势,但作为支架仍难以满足脊髓组织结构的所有要求,常需要通过将两种或两种以上生物材料联合使用,取长补短,提高材料性能,以期得到一种理想的脊髓修复材料。例如,虽然胶原蛋白是一种很有前途的生物材料,具有良好的相容性,但其力学性能较差,降解速度太快。而壳聚糖已被成功地用于改善大鼠脊髓横断组织间隙以弥补胶原的不足。同时壳聚糖的降解时间较长,可以通过调整两种材料的比例调节降解速度[22]。因此,两种或多种材料的结合可以克服单一材料的局限性。 2.2.2 合成材料 天然材料在生物相容性方面具有更大的优势,但是很多天然材料如明胶和胶原的物理性能较差,通过生物3D打印技术制作出的支架难以维持其结构,容易塌陷,从而失去了3D生物支架的优势,导致治疗效果欠佳。因此,在支架中添加具备特定功能的合成材料成为了很多研究者的选择。例如,聚乳酸3D支架具有一定机械性能的同时,还可以通过调节比例而调整降解的周期,可以针对不同组织实现对应的支架降解速度[37]。加入聚苯乙,聚吡咯等导电材料对于恢复神经的电生理特性和达到健康神经范围内的神经传导速率非常重要[15,38],这是实现更好的功能恢复所必需的。其他常用的合成聚合物还有聚乙二醇、聚己内酯和聚乳酸等[37,39-40],合成聚合物的使用往往是为了满足支架特定的功能需求,根据不同的功能需要添加合适的合成聚合物也是3D生物支架在脊髓损伤修复领域的研究方向之一。 2.3 不同干细胞对脊髓损伤后组织修复的影响 脊髓损伤的治疗是一个相当大的挑战,由于其脊髓损伤所引发的组织损伤和持续的感觉运动损伤[41],一直受到临床科学家的极大关注。干细胞是一种具有可以产生多种类型细胞的自然能力的原始细胞,可以在脊髓损伤处提供一种修复系统[42-43]。干细胞结合3D生物打印支架可以更好地改善损伤组织的微环境,可以为组织和细胞提供营养支持,从而增强血管化、改变炎症反应并防止囊性改变[44]。 在支架制造过程中,可将活细胞嵌入支架或置于生物打印墨水中[45]。但是干细胞的选择是一个重要的因素。被选择用于打印的细胞需要在生物支架中模仿在体内的生理状态,并维持其功能[46]。不同类型的干细胞已被用于促进神经再生。用于治疗脊髓损伤的常见干细胞包括间充质干细胞、神经干细胞、胚胎干细胞、诱导多能干细胞、造血干细胞和牙髓干细胞,其中,最常应用的是间充质干细胞和神经干细胞[47]。 有研究发现,在将人脊髓神经干细胞和人脐带间充质干细胞分别装载到纵向胶原海绵支架上移植到T9完全横断的大鼠脊髓中,人脐带间充质干细胞和人脊髓神经干细胞在减少病变间隙周围的胶质瘢痕形成方面具有相同的作用[28]。然而,两者相比较时,植入人神经干细胞更能显著改善运动功能,植入人脐带间充质干细胞更能减轻炎症、促进细胞存活。 大量的研究报道,3D生物支架可以减少移植细胞与宿主细胞之间的距离,并提供一个细胞在体内近似的生理环境。有充分的文献表明,干细胞增殖细胞的形成、黏附、迁移和分化受到干细胞所处基质周围的生化、机械和物理表面特性的影响,而且它们对基质的微环境极其敏感并有反应[48-49]。 2.3.1 间充质干细胞 间充质干细胞是治疗脊髓损伤时最好的候选细胞,它们由于具有耐用性、多效性、免疫调节和自我更新能力,并且容易分离和培养等特点,已经广泛应用于再生医学[50]。间充质干细胞具有分化为不同类型细胞的能力,间充质干细胞根据来源不同分为骨髓间充质干细胞、脐带间充质干细胞和胎盘衍生间充质干细胞 [51-54]。 间充质干细胞可通过巨噬细胞极化机制抑制炎症反应来修复脊髓损伤。间充质干细胞可以促进巨噬细胞向M2型巨噬细胞转化,抑制M1型活化,M2巨噬细胞可以促进组织再生,提供抵抗机体炎症的能力,进而通过控制继发性损伤的影响以达到修复脊髓的目的[55]。间充质干细胞亦可以通过释放脑源性神经营养因子、神经生长因子等因子促进神经组织的再生[56-58],引导细胞的迁移。然而,单纯细胞移植仍存在细胞存活率低的问题。研究发现将明胶3D支架与间充质干细胞一起植入到损伤部位,联合支架移植的细胞比单独移植的细胞存活率更高[40,59], 并且胶原联合骨髓间充质干细胞形成3D支架植入到损伤节段后,受损部位的轴突得到明显改善[60-64]。此外,联合海藻酸盐和间充质干细胞到损伤部位,明显抑制了损伤部位的炎症反应,减少了神经细胞的凋亡[62]。有研究发现,通过将骨髓间充质干细胞和施万细胞联合移植到损伤部位,骨髓间充质干细胞可以生成类似神经鞘膜包裹的髓鞘和轴突,通过调节炎症细胞的募集,减少了损伤区域附近的小胶质细胞和T淋巴细胞,从而减少了神经细胞凋亡和继发炎症,促进脊髓损伤的功能恢复[23]。 文章总结了不同材料联合间充质干细胞移植对脊髓损伤修复的影响相关研究成果,见表2。"
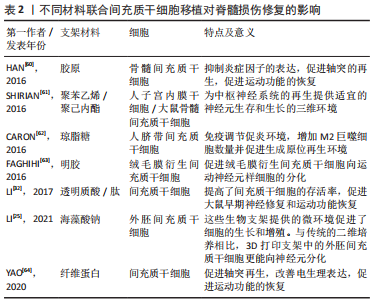

总之,联合3D生物支架移植间充质干细胞在脊髓损伤修复中发挥了很重要的作用,所选用的支架材料不同,支架相应的特性也会不同,间充质干细胞的存活和分化情况也不同,发挥的治疗机制也不相同。根据脊髓损伤的具体发病情况和机制的不同,探索合适的生物材料以及搭配方式可能是进一步的研究重点。 2.3.2 神经干细胞 神经干细胞是一种拥有自我更新能力且容易诱导分化为多种类型的多能祖细胞亚型,如神经元细胞、少突胶质细胞和星形胶质细胞[47]。脊髓损伤后,因为脊髓受到的外力撕裂、氧化应激、继发炎症和缺血缺氧等造成神经元损伤、轴突脱髓鞘、胶质瘢痕的形成及脊髓空洞的产生等改变。研究发现,联合神经干细胞制成的生物支架可以通过减少活化巨噬细胞和小胶质细胞来减少炎症、抑制瘢痕形成[65]。在生物3D打印制成的支架中,结合支架内部留有的空心通道,神经干细胞可以具有更高的存活率和增殖速度,提高向神经方向分化的能力,促进轴突的生成,形成神经网络[65-66]。 文章总结了不同材料联合神经干细胞移植对脊髓损伤修复的影响的相关研究进展,见表3。"


总之,3D仿脊髓脊髓支架保障和促进了神经干细胞的存活和分化,抑制了瘢痕的形成,有效地促进了轴突的再生以及改善了运动功能的恢复。但是,3D生物打印支架如何促进神经干细胞分化为神经元而抑制向星形胶质细胞的分化以及抑制损伤节段胶质的产生仍有待研究。 2.3.3 其他细胞 多种细胞都可以和3D生物打印支架联合进行移植,从而达到促进轴突再生、进一步分化为神经细胞或分泌相关因子、减轻局部炎症反应的作用,从而修复受损的脊髓组织。研究发现,人子宫内膜干细胞、嗅鞘细胞、牙髓干细胞、脂肪来源干细胞和胚胎干细胞等都能和3D生物支架联合移植治疗脊髓损伤,从而促进神经营养因子等的释放或促进细胞向神经分化[47]。 KATOH等[67]利用脂肪组织制成三维支架并将其移植至脊髓半切模型的大鼠体内,发现基因激活的同基因脂肪组织可在一段较长的时间内将治疗分子输送到脊髓损伤部位,同时脂肪细胞可以减轻损伤区域的炎症反应,从而改善微环境,利于脊髓的再生。HEO等[15]将背根神经节细胞装载至含有聚苯乙烯的3D支架中,发现其支架可以通过增强导电性而促进细胞向神经方向分化。 因此,探索其他更多可用于联合3D生物打印支架治疗脊髓损伤的细胞联合治疗可能是未来发展的一个方向。"

| [1] RAMER LM, RAMER MS, BRADBURY EJ. Restoring function after spinal cord injury: towards clinical translation of experimental strategies. Lancet Neurol. 2014;13(12):1241-1256. [2] MCDONALD JW, SADOWSKY C. Spinal-cord injury. Lancet. 2002;359(9304):417-425. [3] CUNHA C, PANSERI S, ANTONINI S. Emerging nanotechnology approaches in tissue engineering for peripheral nerve regeneration. Nanomedicine. 2011;7(1):50-59. [4] SUBRAMANIAN A, KRISHNAN UM, SETHURAMAN S. Development of biomaterial scaffold for nerve tissue engineering: Biomaterial mediated neural regeneration. J Biomed Sci. 2009;16(1):108. [5] QASIM M, CHAE DS, LEE NY. Advancements and frontiers in nano-based 3D and 4D scaffolds for bone and cartilage tissue engineering. Int J Nanomedicine. 2019;14:4333-4351. [6] 赵瑾,袁晓燕,姚康德.组织工程多孔支架制备技术进展[J].化工进展, 2002,21(9):644-648. [7] RICHARDS DJ, TAN Y, JIA J, et al. 3D printing for tissue engineering. Isr J Chem. 2013;53(9-10):805-814. [8] FARZADI A, SOLATI-HASHJIN M, ASADI-EYDIVAND M, et al. Effect of layer thickness and printing orientation on mechanical properties and dimensional accuracy of 3D printed porous samples for bone tissue engineering. PLoS One. 2014;9(9):e108252. [9] NYBERG E, RINDONE A, DORAFSHAR A, et al. Comparison of 3D-printed poly-ɛ-caprolactone scaffolds functionalized with tricalcium phosphate, hydroxyapatite, bio-oss, or decellularized bone matrix. Tissue Eng Part A. 2017;23(11-12):503-514. [10] PEI P, QI X ,DU X , et al. Three-dimensional printing of tricalcium silicate/mesoporous bioactive glass cement scaffolds for bone regeneration. J Mater Chem B. 2016;4(46):7452-7463. [11] BEDIR T, ULAG S, USTUNDAG CB, et al. 3D bioprinting applications in neural tissue engineering for spinal cord injury repair. Mater Sci Eng C Mater Biol Appl. 2020;110:110741. [12] LIU X, HAO M, CHEN Z, et al. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials. 2021;272:120771. [13] HUNT NC, GROVER LM. Cell encapsulation using biopolymer gels for regenerative medicine. Biotechnol Lett. 2010;32(6):733-742. [14] ZHU W, GEORGE JK, SORGER VJ, et al. 3D printing scaffold coupled with low level light therapy for neural tissue regeneration. Biofabrication. 2017; 9(2):025002. [15] HEO DN, LEE SJ, TIMSINA R, et al. Development of 3D printable conductive hydrogel with crystallized PEDOT: PSS for neural tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;99:582-590. [16] JOSE RR, BROWN JE, POLIDO KE, et al. Polyol-silk bioink formulations as two-part room-temperature curable materials for 3D printing. ACS Biomater Sci Eng. 2015;1(9):780-788. [17] NI S, LI X, YANG P, et al. Enhanced apatite-forming ability and antibacterial activity of porous anodic alumina embedded with CaO-SiO2-Ag2O bioactive materials. Mater Sci Eng C Mater Biol Appl. 2016;58:700-708. [18] SHAO HF, HE Y, FU JZ, et al. 3D printing magnesium-doped wolastonite/β-TCP bioceramics scaffolds with high strength and adjustable degradation. J Eur Ceram Soc. 2016;36(6):1495-1503. [19] PEI P, WEI DX, ZHU M, et al. The effect of calcium sulfate incorporation on physiochemical and biological roperties of 3D-printed mesoporous calcium silicatecement scaffolds. Microporous Mesoporous Mater. 2017;241:11-20. [20] PAGE H, FLOOD P, REYNAUD EG. Three-dimensional tissue cultures: current trends and beyond. Cell Tissue Res. 2013;352(1):123-131. [21] LI XH, ZHU X, LIU XY, et al. The corticospinal tract structure of collagen/silk fibroin scaffold implants using 3D printing promotes functional recovery after complete spinal cord transection in rats. J Mater Sci Mater Med. 2021; 32(4):31. [22] SUN Y, YANG C, ZHU X, et al. 3D printing collagen/chitosan scaffold ameliorated axon regeneration and neurological recovery after spinal cord injury. J Biomed Mater Res A. 2019;107(9):1898-1908. [23] WANG J, KONG X, LI Q, et al. The spatial arrangement of cells in a 3D-printed biomimetic spinal cord promotes directional differentiation and repairs the motor function after spinal cord injury. Biofabrication. 2021. doi: 10.1088/1758-5090/ac0c5f. [24] JOUNG D, TRUONG V, NEITZKE CC, et al. 3D printed stem-cell derived neural progenitors generate spinal cord scaffolds. Adv Funct Mater. 2018; 28(39):1801850. [25] LI Y, CAO X, DENG W, et al. 3D printable Sodium alginate-Matrigel (SA-MA) hydrogel facilitated ectomesenchymal stem cells (EMSCs) neuron differentiation. J Biomater Appl. 2021;35(6):709-719. [26] KIYOTAKE EA, THOMAS EE, HOMBURG HB, et al. Conductive and injectable hyaluronic acid/gelatin/gold nanorod hydrogels for enhanced surgical translation and bioprinting. J Biomed Mater Res A. 2022;110(2):365-382. [27] GU Q, TOMASKOVIC-CROOK E, LOZANO R, et al. Functional 3D neural mini-tissues from printed gel-based bioink and human neural stem cells. Adv Healthc Mater. 2016;5(12):1429-1438. [28] ZOU Y, ZHAO Y, XIAO Z, et al. Comparison of regenerative effects of transplanting three-dimensional longitudinal scaffold loaded-human mesenchymal stem cells and human neural stem cells on spinal cord completely transected rats. ACS Biomater Sci Eng. 2020;6(3):1671-1680. [29] LIU S, XIE YY, WANG LD, et al. A multi-channel collagen scaffold loaded with neural stem cells for the repair of spinal cord injury. Neural Regen Res. 2021;16(11):2284-2292. [30] YUE K, TRUJILLO-DE SANTIAGO G, ALVAREZ MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. [31] HOSSEINI SM, SHARAFKHAH A, KOOHI-HOSSEINABADI O, et al. Transplantation of neural stem cells cultured in alginate scaffold for spinal cord injury in rats. Asian Spine J. 2016;10(4):611-618. [32] LI LM, HAN M, JIANG XC, et al. Peptide-tethered hydrogel scaffold promotes recovery from spinal cord transection via synergism with mesenchymal stem cells. ACS Appl Mater Interfaces. 2017;9(4):3330-3342. [33] VENKATESAN J, KIM SK. Chitosan composites for bone tissue engineering--an overview. Mar Drugs. 2010;8(8):2252-2266. [34] AAM BB, HEGGSET EB, NORBERG AL, et al. Production of chitooligosaccharides and their potential applications in medicine. Mar Drugs. 2010;8(5):1482-1517. [35] GAO S, ZHAO P, LIN C, et al. Differentiation of human adipose-derived stem cells into neuron-like cells which are compatible with photocurable three-dimensional scaffolds. Tissue Eng Part A. 2014;20(7-8):1271-1284. [36] LIU XY, CHEN C, XU HH, et al. Integrated printed BDNF/collagen/chitosan scaffolds with low temperature extrusion 3D printer accelerated neural regeneration after spinal cord injury. Regen Biomater. 2021;8(6):rbab047. [37] KAPLAN B, MERDLER U, SZKLANNY AA, et al. Rapid prototyping fabrication of soft and oriented polyester scaffolds for axonal guidance. Biomaterials. 2020;251:120062. [38] HUANG J, YAP N, WALTER M, et al. 3D-printed polypyrrole microneedle arrays for electronically controlled transdural drug release. ACS Biomater Sci Eng. 2022;8(4):1544-1553. [39] KOFFLER J, ZHU W, QU X, et al. Biomimetic 3D-printed scaffolds for spinal cord injury repair. Nat Med. 2019;25(2):263-269. [40] HSIEH FY, LIN HH, HSU SH. 3D bioprinting of neural stem cell-laden thermoresponsive biodegradable polyurethane hydrogel and potential in central nervous system repair. Biomaterials. 2015;71:48-57. [41] GUO S, REDENSKI I, LEVENBERG S. Spinal cord repair: from cells and tissue engineering to extracellular vesicles. Cells. 2021;10(8):1872. [42] ZAKRZEWSKI W, DOBRZYŃSKI M, SZYMONOWICZ M, et al. Stem cells: past, present, and future. Stem Cell Res Ther. 2019;10(1):68. [43] HUANG L, FU C, XIONG F, et al. Stem cell therapy for spinal cord injury. Cell Transplant. 2021;30:963689721989266. [44] JOHNSON LDV, PICKARD MR, JOHNSON WEB. The comparative effects of mesenchymal stem cell transplantation therapy for spinal cord injury in humans and animal models: a systematic review and meta-analysis. Biology (Basel). 2021;10(3):230. [45] LEE SJ, ESWORTHY T, STAKE S, et al. Advances in 3D bioprinting for neural tissue engineering. Adv Biosys. 2018;2:1700213. [46] AMBESI-IMPIOMBATO FS, PARKS LA, COON HG. Culture of hormone-dependent functional epithelial cells from rat thyroids. Proc Natl Acad Sci U S A. 1980;77(6):3455-3459. [47] ZAREPOUR A, HOOSHMAND S, GÖKMEN A, et al. Spinal cord injury management through the combination of stem cells and implantable 3D bioprinted platforms. Cells. 2021;10(11):3189. [48] CHEN G, LV Y, GUO P, et al. Matrix mechanics and fluid shear stress control stem cells fate in three dimensional microenvironment. Curr Stem Cell Res Ther. 2013;8(4):313-323. [49] PEK YS, WAN AC, YING JY. The effect of matrix stiffness on mesenchymal stem cell differentiation in a 3D thixotropic gel. Biomaterials. 2010;31(3): 385-391. [50] OCANSEY DKW, PEI B, YAN Y, et al. Improved therapeutics of modified mesenchymal stem cells: an update. J Transl Med. 2020;18(1):42. [51] BARA JJ, RICHARDS RG, ALINI M, et al. Concise review: bone marrow-derived mesenchymal stemcells change phenotype following in vitro culture: implications for basic research and the clinic. Stem cells. 2014;32(7): 1713-1723. [52] OLIVEIRA MS, BARRETO-FILHO JB. Placental-derivedstem cells: culture, differentiation and challenges. World J Stem Cells. 2015;7(4):769-775. [53] TOKIKO NI, HE HP. Umbilical cordderived mesenchymal stem cells: Their advantages andpotential clinical utility. World J Stem Cells. 2014;6(2): 195-202. [54] HAN S, XIAO Z, LI X, et al. Human placenta-derived mesenchymal stem cells loaded on linear ordered collagen scaffold improves functional recovery after completely transected spinal cord injury in canine. Sci China Life Sci. 2018;61(1):2-13. [55] AN N, YANG J, WANG H, et al. Mechanism of mesenchymal stem cells in spinal cord injury repair through macrophage polarization. Cell Biosci. 2021; 11(1):41. [56] ZENG X, QIU XC, MA YH, et al. Integration of donor mesenchymal stem cell-derived neuronlike cells into host neural network after rat spinal cordtransection. Biomaterials. 2015;53:184-201. [57] WISLET-GENDEBIEN S, WAUTIER F, LEPRINCE P, et al. Astrocytic and neuronal fate of mesenchymal stem cells expressing nestin. Brain Res Bull. 2005; 68(1-2):95-102. [58] MUKHAMEDSHINA YO, GRACHEVA OA, MUKHUTDINOVA DM, et al. Mesenchymal stem cells and the neuronal microenvironment in the area of spinal cord injury. Neural Regen Res. 2019;14(2):227-237. [59] ZHANG K, LIU Z, LI G, et al. Electro-acupuncture promotes the survival and differentiation of transplanted bone marrow mesenchymal stem cells pre-induced with neurotrophin-3 and retinoic acid in gelatin sponge scaffold after rat spinal cord transection. Stem Cell Rev Rep. 2014;10(4):612-625. [60] HAN S, WANG B, LI X, et al. Bone marrow-derived mesenchymal stem cells in three-dimensional culture promote neuronal regeneration by neurotrophic protection and immunomodulation. J Biomed Mater Res A. 2016;104(7):1759-1769. [61] SHIRIAN S, EBRAHIMI-BAROUGH S, SABERI H, et al. Comparison of capability of human bone marrow mesenchymal stem cells and endometrial stem cells to differentiate into motor neurons on electrospun poly (ε-caprolactone) Scaffold. Mol Neurobiol. 2016;53(8):5278-5287. [62] CARON I, ROSSI F, PAPA S, et al. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135-147. [63] FAGHIHI F, MIRZAEI E, AI J, et al. Differentiation potential of human chorion-derived mesenchymal stem cells into motor neuron-like cells in two- and three-dimensional culture systems. Mol Neurobiol. 2016;53(3):1862-1872. [64] YAO S, HE F, CAO Z, et al. Mesenchymal stem cell-laden hydrogel microfibers for promoting nerve fiber regeneration in long-distance spinal cord transection injury. ACS Biomater Sci Eng. 2020;6(2):1165-1175. [65] FAN L, LIU C, CHEN X, et al. Directing induced pluripotent stem cell derived neural stem cell fate with a three-dimensional biomimetic hydrogel for spinal cord injury repair. ACS Appl Mater Interfaces. 2018;10(21):17742-17755. [66] JIANG JP, LIU XY, ZHAO F, et al. Three-dimensional bioprinting collagen/silk fibroin scaffold combined with neural stem cells promotes nerve regeneration after spinal cord injury. Neural Regen Res. 2020;15(5):959-968. [67] KATOH H, YOKOTA K, FEHLINGS MG. Regeneration of spinal cord connectivity through stem cell transplantation and biomaterial scaffolds. Front Cell Neurosci. 2019;13:248. |
| [1] | Chi Wenxin, Zhang Cunxin, Gao Kai, Lyu Chaoliang, Zhang Kefeng. Mechanism by which nobiletin inhibits inflammatory response of BV2 microglia [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1321-1327. |
| [2] | Yang Zhihang, Sun Zuyan, Huang Wenliang, Wan Yu, Chen Shida, Deng Jiang. Nerve growth factor promotes chondrogenic differentiation and inhibits hypertrophic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1336-1342. |
| [3] | Hu Taotao, Liu Bing, Chen Cheng, Yin Zongyin, Kan Daohong, Ni Jie, Ye Lingxiao, Zheng Xiangbing, Yan Min, Zou Yong. Human amniotic mesenchymal stem cells overexpressing neuregulin-1 promote skin wound healing in mice [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1343-1349. |
| [4] | Jin Kai, Tang Ting, Li Meile, Xie Yuan. Effects of conditioned medium and exosomes of human umbilical cord mesenchymal stem cells on proliferation, migration, invasion, and apoptosis of hepatocellular carcinoma cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1350-1355. |
| [5] | Li Dijun, Jiu Jingwei, Liu Haifeng, Yan Lei, Li Songyan, Wang Bin. Three-dimensional gelatin microspheres loaded human umbilical cord mesenchymal stem cells for chronic tendinopathy repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1356-1362. |
| [6] | Liu Qi, Li Linzhen, Li Yusheng, Jiao Hongzhuo, Yang Cheng, Zhang Juntao. Icariin-containing serum promotes chondrocyte proliferation and chondrogenic differentiation of stem cells in the co-culture system of three kinds of cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1371-1379. |
| [7] | Zhang Zhenyu, Liang Qiujian, Yang Jun, Wei Xiangyu, Jiang Jie, Huang Linke, Tan Zhen. Target of neohesperidin in treatment of osteoporosis and its effect on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1437-1447. |
| [8] | Li Jialin, Zhang Yaodong, Lou Yanru, Yu Yang, Yang Rui. Molecular mechanisms underlying role of mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1512-1522. |
| [9] | Zhao Ruihua, Chen Sixian, Guo Yang, Shi Lei, Wu Chengjie, Wu Mao, Yang Guanglu, Zhang Haoheng, Ma Yong. Wen-Shen-Tong-Du Decoction promoting spinal cord injury repair in mice [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1118-1126. |
| [10] | He Bo, Chen Wen, Ma Suilu, He Zhijun, Song Yuan, Li Jinpeng, Liu Tao, Wei Xiaotao, Wang Weiwei, Xie Jing . Pathogenesis and treatment progress of flap ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1230-1238. |
| [11] | Wang Rongrong, Huang Yushan, Li Xiangmiao, Bai Jinzhu. Prostaglandin E1 regulates vascular-related factors and protects microcirculatory function during the acute phase of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 958-967. |
| [12] | Han Haihui, Meng Xiaohu, Xu Bo, Ran Le, Shi Qi, Xiao Lianbo. Effect of fibroblast growth factor receptor 1 inhibitor on bone destruction in rats with collagen-induced arthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 968-977. |
| [13] | Yang Bin, Tao Guangyi, Yang Shun, Xu Junjie, Huang Junqing . Visualization analysis of research hotspots of artificial intelligence in field of spinal cord nerve injury and repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 761-770. |
| [14] | Wang Zilin, Mu Qiuju, Liu Hongjie, Shen Yuxue, Zhu Lili. Protective effects of platelet-rich plasma hydrogel on oxidative damage in L929 cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 771-779. |
| [15] | Zhao Hongxia, Sun Zhengwei, Han Yang, Wu Xuechao , Han Jing. Osteogenic properties of platelet-rich fibrin combined with gelatin methacryloyl hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 809-817. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||