Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (30): 4869-4875.doi: 10.12307/2023.595
Previous Articles Next Articles
Ischemic preconditioning improves exercise performance: methods, applications and mechanisms
Wang Zhou1, 2, Wu Ying1, 2
- 1Beijing Sport University, Beijing 100084, China; 2Laboratory of Sports Stress and Adaptation of General Administration of Sport, Beijing 100084, China
-
Received:2022-10-11Accepted:2022-11-12Online:2023-10-28Published:2023-04-03 -
Contact:Wu Ying, Associate professor, Master’s supervisor, Beijing Sport University, Beijing 100084, China; Laboratory of Sports Stress and Adaptation of General Administration of Sport, Beijing 100084, China -
About author:Wang Zhou, Master, Beijing Sport University, Beijing 100084, China; Laboratory of Sports Stress and Adaptation of General Administration of Sport, Beijing 100084, China -
Supported by:Olympic Science and Technology Project of the State General Administration of Sport, Nos. 2021592 and 2019634 (to WY)
CLC Number:
Cite this article
Wang Zhou, Wu Ying. Ischemic preconditioning improves exercise performance: methods, applications and mechanisms[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4869-4875.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
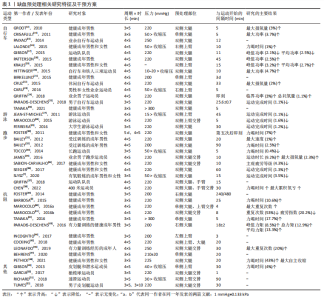
2.1 缺血预处理的干预方法及效应特征 2.1.1 缺血预处理的装置与干预部位 缺血预处理的装置由充气设备和加压载体两部分组成。充气设备包括自动和手动两种类型,压力数值均可量化,加压载体为无弹性尼龙材质充气袖带[3-4]。缺血预处理主要是通过对四肢体近侧端(上臂或大腿)施加压力,从而完全闭塞动脉血流和部分闭塞静脉血流,整个过程中干预对象处于仰卧位。在相关研究中,上下肢使用的袖带宽度存在差异。上肢缺血预处理使用的袖带较窄,宽度为8.0-9.0 cm[5-6],下肢使用的袖带较宽,为11.5-21.5 cm[7-8],袖带长度为42-96 cm[9-10]。加压袖带的尺寸在一定程度上决定了受干预的肌肉面积,这可能对干预效果产生一定影响,但并未见不同袖带尺寸对缺血预处理作用效果影响的研究,但是当受缺血预处理干预的肢体面积不相同时,袖带对肢体的闭塞效果也可能不同,一定范围内,作用效果可能会随着面积的增大而增加,但还需要具体的实证研究来证实袖带尺寸的大小对缺血预处理效果的影响。 2.1.2 缺血预处理的压力 压力大小是影响缺血预处理效果的重要因素。缺血预处理作用于不同人群和部位的压力不存在明显差异,其范围集中在200-240 mmHg (1 mmHg=0.133 kPa),略高于加压训练使用的压力。相关研究指出,至少180 mmHg的压力才可以有效闭塞健康成年男性上下肢动脉血流,引起血氧饱和度(SaO2)、氧分压(PO2)、二氧化碳分压(PCO2)和血液pH值的明显改变[11]。但是,由于袖带尺寸的不同、人体自身结构和肌肉形态的差异,相同的绝对压力施加到人体后,也可能会产生不同效果,即使在250 mmHg的高压下,某些参与者的动脉血流也不能被完全阻断[12]。因此,面对不同个体应采用个性化方案有针对性进行干预。未来针对缺血预处理的研究或应用也可使用多普勒超声设备或近红外光谱分析动脉血流是否被完全阻断,以此确定不同个体的适宜压力,以获得最佳干预效果。 2.1.3 缺血预处理的“剂量” 缺血预处理的“剂量”包括干预的时长和重复次数,这对其作用效果至关重要。不同研究中缺血预处理干预的时长有所不同,大部分研究的干预时长在40 min(4个缺血预处理周期)。该综述分析的研究使用最多的是4个缺血再灌注周期。过多的干预周期并不会对作用效果产生更加显著的影响。研究表明,8×5 min,220 mmHg的干预并没有比4×5 min,220 mmHg的方案对运动表现产生更有益的效果,同等负荷下自行车运动员总的运动时间也并没有延长[13]。因此,过多的干预周期和过长的时间并不会增加缺血预处理的作用效果,反而可能诱导肌肉组织和细胞因长时间缺血发生损伤。现行的缺血预处理方案并未见有对人体产生不适的影响,现有研究中使用最多的是4×5 min、220 mmHg的干预方案。 文章总结了目前各研究中缺血预处理相关研究特征及干预方案[14-52],见表1。"
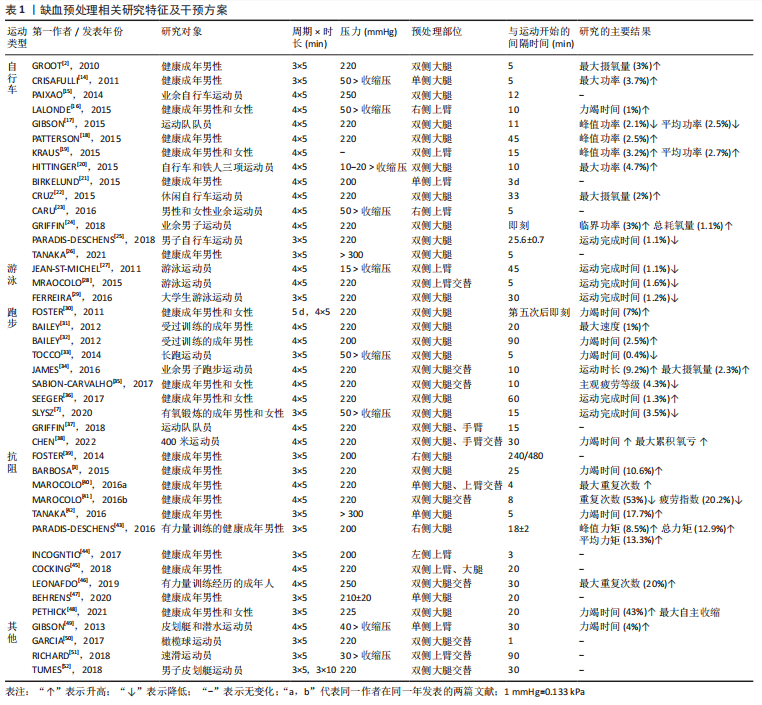
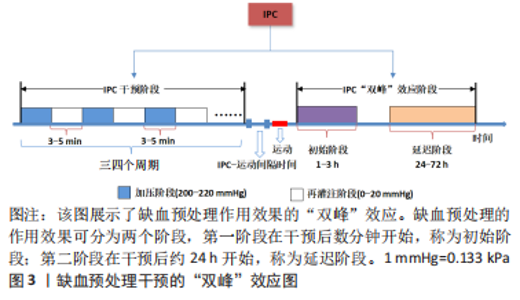
2.1.4 缺血预处理的时程效应和远程效应 缺血预处理的作用效果具有时程效应,主要分为两个阶段,见图3,且呈现出“双峰”特点。第一阶段在缺血预处理后数分钟内开始发挥作用,可持续1-3 h,称为早期阶段,与一氧化氮的合成和阿片类物质的释放有关。第二阶段在缺血预处理后24 h作用效果开始显现,可持续至72 h,称为晚期阶段,主要依赖于缓激肽和腺苷[53]。LISBOA等[54]研究证明了缺血预处理对游泳运动员50 m自由泳运动成绩的影响具有时程特点,从缺血预处理后2 h开始,其效应可持续至8 h。健康成年男性在低氧状态下应用急性缺血预处理对连续2次5 km跑(间隔1 h)完成时间的影响,尽管缺血预处理没有减少第二次5 km跑的完成时间,但两次5 km跑的成绩未表现出显著差异,表明缺血预处理维持了运动表现,同样证实了缺血预处理产生了积极的延迟效应[55]。因此,将缺血预处理运用于运动训练时,作用效果的时程效应被重点考虑,教练员、运动员应根据实际情况,合理安排好缺血预处理和训练之间的间隔时间,使训练、比赛的时间与缺血预处理的效应期吻合,从而有效地发挥缺血预处理的作用。"

2.2 缺血预处理对运动表现的影响 2.2.1 缺血预处理对有氧能力的影响 有氧能力对长时间(运动时长> 100 s)运动项目的运动表现至关重要,缺血预处理与有氧运动成绩密切相关,可有效提高有氧能力。缺血预处理对有氧能力的积极作用在自行车、长跑等项目中被普遍报道。研究表明,急性缺血预处理后,自行车和长跑运动员最大摄氧量增加0.3%-3.6%,力竭运动的总时间延长1.1%-2.6% [2],主观疲劳感觉显著降低。急性缺血预处理后,人体对随后运动缺血的耐受能力提高,糖原消耗减少,线粒体生物合成功能增强[10],这是急性缺血预处理后有氧能力提升的主要原因。重复使用缺血预处理可以显著增加骨骼肌氧合能力和毛细血管氧合血流量[4, 56],对有氧能力的提升效果优于急性使用。JEFFRIES等[56]报告称,20名健康成年人连续7 d双侧下肢缺血预处理后,静息状态骨骼肌新陈代谢下降,骨骼肌氧化功能提高了13%,毛细血管氧合血流量增多,且干预后72 h的运动表现提升9%。LINDSAY等[4]也证明了重复使用缺血预处理对有氧能力的积极影响,对健康成年人进行7 d下肢缺血预处理后,48 h和7 d后的最大摄氧量相对于基线水平分别提高9.5%和12.8%。 以上研究表明,缺血预处理不仅可以有效提升有氧运动能力,而且其在提升有氧耐力表现时具有时程效应和一定的“剂量”依赖性。单次缺血预处理更多地是通过对机体一次性干预后的应激性发挥作用,而重复缺血预处理更多地是通过对人体较长时间干预后,引起心血管系统和循环系统对长时间缺血、缺氧状态的适应,做出积极性改变,从而提升运动表现。但是,重复缺血预处理和单次缺血预处理在除有氧运动外的其他运动上会表现出怎样的效果,重复次数上的差异是否会引起作用效果和时程效应上的不同,仍需进一步证实。 2.2.2 缺血预处理对力量的影响 力量耐力:缺血预处理不仅对有氧能力产生积极效果,对运动员和健康人群的力量耐力也有明显的促进作用。缺血预处理后,健康成年人80%1RM卧推、深蹲和仰卧45°腿部蹬伸的最大重复次数显著增加,45%1RM握力运动时间延长[5,57-58],表明缺血预处理增强了相关运动肌肉的肌肉耐力,提高了力量耐力的运动表现。游泳运动中,缺血预处理同样具有积极效果。在6次连续50 m(3 min间隔)自由泳冲刺前48 h、24 h和30 min进行3次缺血预处理干预,缺血预处理组的成绩改善(1.17%)明显高于对照组(0.2%)[29],这可能是缺血预处理后骨骼肌抗疲劳能力增加,保持了较高的划水和打腿频率所引起的。 除了作为一种独立的干预方式,缺血预处理和力量、速度训练的协同使用同样对耐力提升表现出积极影响。PARADIS-DESCHENS 等[59]报道,连续4周(2 d/周)的缺血预处理与短跑间歇训练相结合后,耐力运动员在5 km计时跑(完成时间)和30 s的Wingate测试期间的力量耐力表现有更大幅度的改善。缺血预处理干预后,全身性运动过程中肌红蛋白浓度明显升高,肌肉氧合能力增强,保证了运动时氧气的供应和利用率,进而改善运动表现。CARVALHO等[60]研究表明,缺血预处理结合膝关节伸肌阻力训练持续6周(2 d/周),膝关节最大力量伸膝重复次数显著超过了对照组。为了验证机体不同部位与缺血预处理结合干预的作用效果,研究者对健康成年男性进行持续2周(3 d/周)的缺血预处理结合腕伸肌阻力训练,最大重复次数比对照组显著增加[60]。上述研究结果表明,与单纯的缺血预处理干预相比,缺血预处理和运动训练的联合干预可更有效地提高力量耐力,且此种方法具有一定的普适性,在全身性运动、局部关节运动等多种模式中均有良好效果。 与目前传统的力量训练相比,缺血预处理是一种提高力量耐力的非运动性干预手段,如果将缺血预处理与运动干预手段相结合,是否会对力量耐力的提升产生“1+1 > 2”的效果值得深入探讨。 爆发力:缺血预处理对爆发力的影响效果仍存在争议,仅有两项研究证明了缺血预处理对爆发力的积极效果,大部分研究表明缺血预处理对爆发力没有影响。在进行连续7 d缺血预处理后,自行车运动员下肢Wingate峰值功率增加11%,平均功率增加了4.3%[4]。单次使用缺血预处理干预后,健康成年人在最大强度离心运动后的0-24 h,肌肉性能也得到快速恢复,纵跳高度提升4.1%。游泳运动中,运动员进行缺血预处理后,划水频率显著增加,同时50 m自由泳的成绩也得到提升[54]。然而,LALONDE等[16]并未发现缺血预处理后Wingate测试的峰值功率、平均功率和达到峰值功率的时间与对照组有显著差异,30 m冲刺运动的表现未得到提升。同样的结果也被PATTERSON等[18]证明,急性缺血预处理效应的早期阶段,在重复的短跑冲刺中,肌肉氧合的虽然维持得到了改善,但并未提升运动表现,表明缺血预处理对爆发力并没有积极效应,这与之前的研究结果具有一致性。此外,GARCIA等[50]对8名男子橄榄球运动员进行缺血预处理后,运动员T型测试、垂直纵跳及30 s持续跳远等运动表现也均未有所提升。与耐力运动相比,爆发力项目一般持续时间极短,供能方式存在本质区别。通过对上述研究归纳分析发现,缺血预处理和运动起始间隔时间对以爆发力为基础的短时间(< 10 s)运动表现影响较大。有学者认为,缺血预处理对爆发力的影响存在一定的时间依赖性,呈现出正相关趋势,即一定范围内(40-50 min),缺血预处理干预和开始运动之间的间隔越长,效果越明显,如果间隔很短,可能效果甚微或产生负面的影响[16],但仍需进一步证实。 不仅仅是爆发力项目,缺血预处理后ATP、磷酸肌酸和总腺苷核苷酸的含量立即降低,CO2浓度升高,缺血预处理和运动之间的必要间隔可以用能源底物浓度恢复到正常水平所需的时间来解释。这些较低的能源底物浓度会对运动表现产生负面影响,尤其是在更依赖于代谢状态的无氧运动。此外,缺血预处理介导的信号转导到达相关组织所需的最短时间是需要关注的,这也是影响缺血预处理作用效果的关键因素。根据现有研究,建议缺血预处理和开始运动之间应留有至少20 min间隔,爆发力项目应有至少40 min间隔,以保证缺血预处理发挥最大效应。 2.2.3 缺血预处理对无氧耐力的影响 大部分研究表明,缺血预处理对提升无氧耐力有积极作用,主要表现为明显提高下肢Wingate测试的平均输出功率和运动时间的延长。在篮球运动员身上同样发现在热身前进行缺血预处理干预,可以有效提高Wingate下肢无氧功率测试的总输出功率和第三、四次Wingate下肢无氧功率测试的平均功率[62]。除了通过不同形式的冲刺类项目来评价缺血预处理对无氧耐力的影响外,也有研究者采用最大累积氧亏(maximal accumulated oxygen deficit,MAOD)测试来评价缺血预处理对无氧耐力的影响。跑台运动前,对运动员进行缺血预处理干预后,缺血预处理组超最大强度运动(110%VO2max)的力竭时间显著增加,缺血预处理可能通过提高糖酵解系统代谢效率达到提升无氧耐力的目的。与该研究一致,CHEN 等[38]同样发现缺血预处理可提升400 m跑运动员的最大累积氧亏,且该研究比较了远程缺血预处理与局部缺血预处理在提升无氧耐力方面的作用效果,结果表明远程缺血预处理与局部缺血预处理均可提升最大累积氧亏,延长运动力竭时间,两者的作用效果无显著差异,这与 CHENG 等[62]的研究结果一致。"

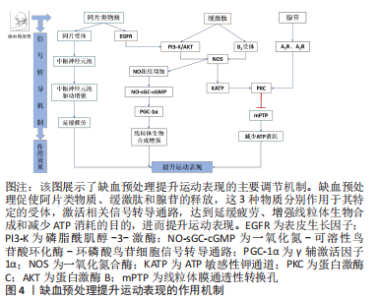
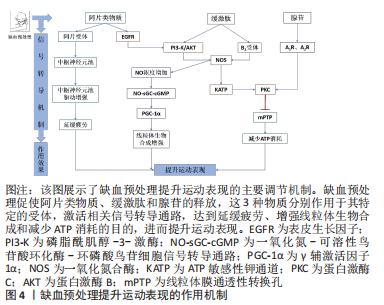
2.3 缺血预处理的作用机制 研究表明,缺血预处理主要是通过改善肌肉氧合、增强线粒体生物合成以及调节神经信号传导提升运动表现,一定量的内源性物质的积累如一氧化氮、阿片类物质、缓激肽(bradykinin,BK)和腺苷,对启动缺血预处理相关的内在反应也是至关重要的。INCOGNITO等[44]认为,当人体因缺血而产生的内源性物质通过血液运输到达中枢神经系统的某个器官时,会作用于它们的特定受体(如腺苷A1R受体),激活细胞保护通路,在提升运动表现中发挥作用。 2.3.1 一氧化氮合成增强 相关研究已经证实补充一氧化氮化合物(如精氨酸和硝酸盐)可增加循环血液中一氧化氮的浓度,提高骨骼肌的氧化能力和耐力运动表现[64]。中等强度运动前对肢体施加缺血预处理刺激,会使运动过程中功能性抗交感作用增强,即运动骨骼肌中血管收缩减弱、血流速度加快、血流量增加。机械刺激(如血管张力、切应力、内皮细胞变形及血液脉冲流动)可引起血管内一氧化氮的释放。同时,缺血预处理过程中肢体的连续缺血引起细胞内cGMP/cAMP比值下降,促使心肌细胞和内皮细胞分泌缓激肽,作用于缓激肽受体B2,增强了一氧化碳合物的活性,加速了一氧化氮的合成速率[65]。一氧化氮是提高全身运动能力和骨骼肌氧化能力(即线粒体功能)的媒介,通过NO-sGC-cGMP信号转导通路调控过氧化物酶体增殖物激活受体γ辅激活因子1α(peroxisome proliferator-activated receptor-γ coactlvator-1α,PGC-1α)转录,进而促进线粒体数目的增加和生物合成的增强[66]。 2.3.2 阿片类物质、缓激肽和腺苷 蛋白激酶C是阿片类物质、缓激肽和腺苷发挥保护作用的共同靶点,蛋白激酶C被阻断后明显抑制保护效果。尽管如此,但这3种物质仍可能通过不同的信号通路发挥作用。缺血预处理刺激机体分泌的内源性物质作用于受体,引起线粒体ATP敏感性钾通道开放,产生氧基自由基,进而激活蛋白激酶C。事实上,仅有阿片类物质和缓激肽的作用机制与此相同。缺血预处理过程中连续的缺血和再灌注刺激阿片类物质、缓激肽和腺苷的分泌,阿片类物质依赖于表皮生长因子(epidermal growth factor receptor,EGFR)增强磷脂酰肌醇-3-激酶(phosphatidylin-ositol-3-kinase,PI3-K)和蛋白激酶B(protein kinase B,Akt)活性,促使ATP敏感性钾通道开放,进而激活蛋白激酶C[67]。但是,缓激肽却绕过了表皮生长因子,直接刺激磷脂酰肌醇-3-激酶和蛋白激酶B发挥作用。相比于阿片类物质和缓激肽,腺苷的作用机制截然不同,它既不依赖于表皮生长因子,也不依赖于磷脂酰肌醇-3-激酶和蛋白激酶B,完全绕过了ATP敏感性钾通道,直接作用于腺苷A1R和A3R受体,激活它们的共同靶点蛋白激酶C,从而调节线粒体膜通透性转换孔(mitochondrial permeability transition pore,mPTP)的开放,减少ATP的消耗[67]。 2.3.3 神经疲劳抑制 缺血预处理可通过降低Ⅲ类和Ⅳ类传入纤维的兴奋性来推迟疲劳感,从而延长受试者的运动时间[22]。Ⅲ类和Ⅳ类传入纤维依靠肌肉至大脑的感觉信息来影响运动性疲劳。运动中肌肉的传入反馈对中枢运动驱动具有抑制性作用,并将外周疲劳的发展限制在个体的临界阈值内。缺血预处理效应的神经调节通路包括脊髓、自主神经系统和体感神经系统,是由内源性阿片类药物激活的,阻断阿片类物质介导的肌肉传入反馈,可增强中枢运动驱动,从而使中枢神经系统对运动引起的外周肌肉疲劳阈值增加。AMANN等[68]研究发现,运动后股四头肌肌电反应增加,表明脊髓运动神经元池有更高的自主神经驱动,这可能是由于运动神经元放电率增加引起的。此外,在接受坐骨神经电刺激的猫后肢上已经观察到缺血预处理后更高的肌电反应,以及在人体耐力运动模型中主观运动感觉(rating of Perceived Exertion,RPE)增长率较低,因此CRISAFULLI等[14]认为缺血预处理提高了身体对疲劳信号感知的阈值,从而延长了受试者的运动时间。 事实上,这种反应发生在缺血预处理的早期阶段,与循环的内源性阿片类物质激活横纹肌组织中的阿片受体引起的肌肉传入电信号减少有关,且阿片是已知的调节耐力表现的物质。研究表明,缺血预处理后机体分泌的内源性阿片物质可以在运动期间激活位于Ⅲ/Ⅳ组肌肉神经组织传入末端的阿片受体[69],抑制传入神经的信号传导,增强了中枢运动神经元池的驱动,降低中枢疲劳的发展速度,这将延缓肌肉神经疲劳的过早发生,提高运动中骨骼肌纤维募集比例,从而维持运动能力,见图4。"
| [1] MURRY CE, JENNINGS RB, REIMER KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74(5):1124-1136. [2] GROOT P, THIJSSEN D, SANCHEZ M, et al. Ischemic preconditioning improves maximal performance in humans. Eur J Appl Physiol. 2010;108(1):141-146. [3] BARBOSA TC, MACHADO AC, BRAZ ID, et al. Remote ischemic preconditioning delays fatigue development during handgrip exercise. Scand J Med Sci Sports. 2015;25(3):356-364. [4] LINDSAY A, PETERSEN C, BLACKWELL G, et al. The effect of 1 week of repeated ischaemic leg preconditioning on simulated Keirin cycling performance: a randomised trial. BMJ Open Sport Exerc Med. 2017;3(1):e000229. [5] GUILHERME L, CARELLI L, I BRAZ I, et al. Effects of ischemic preconditioning as a warm-up on leg press and bench press performance. J Hum Kinet. 2020;75(1):267-277. [6] FRANZ A, BEHRINGER M, HARMSEN JF, et al. Ischemic preconditioning blunts muscle damage responses induced by eccentric exercise. Med Sci Sports Exerc. 2018;50(1):109-115. [7] SLYSZ JT, PETRICK HL, MARROW JP, et al. An examination of individual responses to ischemic preconditioning and the effect of repeated ischemic preconditioning on cycling performance. Eur J Sport Sci. 2020;20(5):633-640. [8] SOUZA H, ARRIEL RA, HOHL R, et al. Is Ischemic preconditioning intervention occlusion-dependent to enhance resistance exercise performance? J Strength Cond Res. 2021;35(10):2706-2712. [9] SOUZA H, ARRIEL RA, MOTA GR, et al. Does ischemic preconditioning really improve performance or it is just a placebo effect? PLoS One. 2021;16(5):e0250572. [10] MANSOUR Z, BOUITBIR J, CHARLES AL, et al. Remote and local ischemic preconditioning equivalently protects rat skeletal muscle mitochondrial function during experimental aortic cross-clamping. J Vasc Surg. 2012;55(2):497-505. [11] SHARMA V, CUNNIFFE B, VERMA AP, et al. Characterization of acute ischemia‐related physiological responses associated with remote ischemic preconditioning: a randomized controlled, crossover human study. Physiol Rep. 2014;2(11):e12200. [12] LOENNEKE JP, FAHS CA, ROSSOW LM, et al. Effects of cuff width on arterial occlusion: implications for blood flow restricted exercise. Eur J Appl Physiol. 2012; 112(8):2903-2912. [13] COCKING S, WILSON MG, NICHOLS D, et al. Is there an optimal ischaemic preconditioning dose to improve cycling performance? Int J Sports Physiol Perform. 2018;13(3):274-282. [14] CRISAFULLI A, TANGIANU F, TOCCO F, et al. Ischemic preconditioning of the muscle improves maximal exercise performance but not maximal oxygen uptake in humans. J Appl Physiol (1985). 2011;111(2):530-536. [15] PAIXAO RC, MOTA GR, MAROCOLO M. Acute effect of ischemic preconditioning is detrimental to anaerobic performance in cyclists. Int J Sports Med. 2014;35(11):912-915. [16] LALONDE F, CURNIER DY. Can anaerobic performance be improved by remote ischemic preconditioning? J Strength Cond Res. 2015;29(1):80-85. [17] GIBSON N, MAHONY B, TRACEY C, et al. Effect of ischemic preconditioning on repeated sprint ability in team sport athletes. J Sports Sci. 2015;33:1182-1188. [18] PATTERSON SD, BEZODIS NE, GLAISTER M, et al. The effect of ischemic preconditioning on repeated sprint cycling performance. Med Sci Sports Exerc. 2015;47(8):1652-1658. [19] KRAUS AS, PASHA EP, MACHIN DR, et al. Bilateral upper limb remote ischemic preconditioning improves anaerobic power. Open Sports Med. 2015;9:1-6. [20] HITTINGER EA, MAHER JL, NASH MS, et al. Ischemic preconditioning does not improve peak exercise capacity at sea level or simulated high altitude in trained male cyclists. Appl Physiol Nutr Metab. 2015;40(1):65-71. [21] BIRKELUND T, OBAD DS, MATEJEC R, et al. Remote ischemic preconditioning does not increase circulating or effector organ concentrations of proopiomelanocortin derivates. Scand Cardiovasc J. 2015;49:257-263. [22] CRUZ RS, AGUIAR RA, TURNES T, et al. Effects of ischemic preconditioning on maximal constant-load cycling performance. J Appl Physiol (1985). 2015;119:961-967. [23] CARU M, LALONDE F, GRAVEL H, et al. Remote ischaemic preconditioning shortens QT intervals during exercise in healthy subjects. Eur J Sport Sci. 2016;16(8):1005-1013. [24] GRIFFIN PJ, FERGUSON RA, GISSANE C, et al. Ischemic preconditioning enhances critical power during a 3 minute all-out cycling test. J Sports Sci. 2018;36(9):1038-1043. [25] PARADIS-DESCHENES P, JOANISSE DR, BILLAUT F. Ischemic preconditioning improves time trial performance at moderate altitude. Med Sci Sports Exerc. 2018,50(3):533-541. [26] TANAKA D, SUGA T, SHIMOHO K, et al. Effect of 2-weeks ischemic preconditioning on exercise performance: a pilot study. Front Sports Act Living. 2021;3:646369. [27] JEAN-ST-MICHEL E, MANLHIOT C, LI J, et al. Remote preconditioning improves maximal performance in highly trained athletes. Med Sci Sports Exerc. 2011;43(7): 1280-1286. [28] MAROCOLO M, MOTA GR, PELEGRINI V, et al. Are the beneficial effects of ischemic preconditioning on performance partly a placebo effect? Int J Sports Med. 2015;36: 822-825. [29] FERRIRA TN, SABINO-CARVALHO J, LOPES TR, et al. Ischemic preconditioning and repeated sprint swimming: a placebo and nocebo study. Med Sci Sports Exerc. 2016;48(10):1967-1975. [30] FOSTER GP, WESTERDAHL DE, FOSTER L A, et al. Ischemic preconditioning of the lower extremity attenuates the normal hypoxic increase in pulmonary artery systolic pressure. Respir Physiol Neurobiol. 2011;179(2-3):248-253. [31] BAILEY TG, JONES H, GREGSON W, et al. Effect of ischemic preconditioning on lactate accumulation and running performance. Med Sci Sports Exerc. 2012;44(11):2084-2089. [32] BAILEY TG, BIRK GK, CABLE NT, et al. Remote ischemic preconditioning prevents reduction in brachial artery flow-mediated dilation after strenuous exercise. Am J Physiol Heart Circ Physiol. 2012;303(5):H533-H538. [33] TOCCO F, MARONGIU E, GHIANI G, et al. Muscle ischemic preconditioning does not improve performance during self-paced exercise. Int J Sports Med. 2014;36(1):9-15. [34] JAMES CA, WILLMOTT A, RICHARDSON AJ, et al. Ischaemic preconditioning does not alter the determinants of endurance running performance in the heat. Eur J Appl Physiol. 2016;116(9):1-11. [35] SABINO-CRAVALHO J, LOPES TR, OBEID-FREITAS T, et al. Effect of ischemic preconditioning on endurance performance does not surpass placebo. Med Sci Sports Exerc. 2017;49(1):124-132. [36] SEEGER JPH, TIMMERS S, PLOEGMAKERS D, et al. Is delayed ischemic preconditioning as effective on running performance during a 5km time trial as acute IPC? J Sci Med Sport. 2017;20(2):208-212. [37] GRIFFIN PJ, HUGHES L, GISSANE C, et al. Effects of local versus remote ischemic preconditioning on repeated sprint running performance. J Sports Med Phys Fitness. 2018;59(2):187-194. [38] CHEN Y, YANG J, MURADOV O, et al. Effect of ischemic preconditioning on maximum accumulated oxygen deficit in 400-meter runners. Eur J Sport Sci. 2022. doi: 10.1080/17461391.2022.2064769. [39] FOSTER GP, GIRI PC, ROGERS DM, et al. Ischemic preconditioning improves oxygen saturation and attenuates hypoxic pulmonary vasoconstriction at high altitude. High Alt Med Biol. 2014;15(2):155-161. [40] MAROCOLO M, DA MOTA GR, SIMIM MA, et al. Myths and facts about the effects of ischemic preconditioning on performance. International Sports Med. 2016a;37:87-96. [41] MRAOCOLO M, WILLARDSON JM, MAROCOLO IC, et al. Ischemic preconditioning and placebo intervention improves resistance exercise performance. J Strength Cond Res. 2016b;30:1462-1469. [42] TANAKA D, SUGA T, TANAKA T, et al. Ischemic preconditioning enhances muscle endurance during sustained isometric exercise. Int J Sports Med. 2016;37(8):614-618. [43] PARADIS-DESCHENES, JOANISSE DR, BILLAUT F. Ischemic preconditioning increases muscle perfusion, oxygen uptake, and force in strength-trained athletes. Appl Physiol Nutr Metab. 2016;41(9):938-944. [44] INCOGNITO AV, BURR JF, MILLAR PJ. The effects of ischemic preconditioning on human exercise performance. Sports Med. 2017;46(4):531-544. [45] COCKING S, CABLE NT, WILSON MG, et al. Conduit artery diameter during exercise is enhanced after local, but not remote, ischemic preconditioning. Front Physiol. 2018;9:435. [46] LEONRADO, CARVAIHO, RENATO, et al. Ischemic preconditioning improves strength endurance performance. J Strength Cond Res. 2019;33(12):3332-3337. [47] BEHRENS M, ZSCHORLICH V, MITTLMEIER T, et al. Ischemic preconditioning did not affect central and peripheral factors of performance fatigability after submaximal isometric exercise. Front Physiol. 2020;11:371. [48] PETHICK J, CASSELTON C, WINTER SL, et al. Ischemic preconditioning blunts loss of knee extensor torque complexity with fatigue. Med Sci Sports Exerc. 2021;53(2): 306-315. [49] GIBSON N, WHITE J, NEISH M, et al. Effect of ischemic preconditioning on land-based sprinting in team-sport athletes. Int J Sports Physiol Perform. 2013;8(6):671-676. [50] GARCIA C, MOTA GD, LEICHT A, et al. Ischemic preconditioning and acute recovery of performance in rugby union players. Sports Med Int Open. 2017;1(3):E107-E112. [51] RICHARD P, BILLAUT F. Time-trial performance in elite speed skaters after remote ischemic preconditioning. Int J Sports Physiol Perform. 2018;16:1-9. [52] TURNES T, AGUIAR RA, de OLIVEIRA CRUZ RS, et al. Impact of ischaemia-reperfusion cycles during ischaemic preconditioning on 2000-m rowing ergometer performance. Eur J Appl Physiol. 2018;118:1599-1607. [53] HAUSENLOY DJ, YELLON DM. The Second Window of Preconditioning (SWOP) where are we now? Cardiovasc Drugs Ther. 2010;24(3):235-254. [54] LISBOA FD, TURNES T, Cruz R, et al. The time dependence of the effect of ischemic preconditioning on successive sprint swimming performance. J Sci Med Sport. 2016; 20(5):507-511. [55] MOTA G, WILLIS SJ, SANTOS N, et al. Ischemic preconditioning maintains performance on two 5-km time trials in hypoxia. Med Sci Sports Exerc. 2019;51(11):2309-2317. [56] JEFFRIES O, WALDRON M, PATTISON JR, et al. Enhanced local skeletal muscle oxidative capacity and microvascular blood flow following 7-day ischemic preconditioning in healthy humans. Front Physiol. 2018;9:463. [57] MAROCOLO M, MAROCOLO IC, MOTA GR. Beneficial effects of ischemic preconditioning in resistance exercise fade over time. Int J Sports Med. 2016;37(10):819-824. [58] CARVALHO L, BARROSO R. Ischemic preconditioning improves strength endurance performance. J Strength Cond Res. 2019;33(12):3332-3337. [59] PARADIS-DESCHENES, JOANISSE DR, MAURIEGE P, et al. Ischemic preconditioning enhances aerobic adaptations to sprint interval training in athletes without altering systemic hypoxic signaling and immune function. Front Sports Act Living. 2020;2:41. [60] CARVALHO L, CONCON V, MELONI M, et al. Effects of resistance training combined with ischemic preconditioning on muscle size and strength in resistance-trained individuals. J Sports Med Phys Fitness. 2020;60(11):1431-1436. [61] SURKAR SM, BLAND MD, MATTLAGE AE, et al. Effects of remote limb ischemic conditioning on muscle strength in healthy young adults: a randomized controlled trial. PLoS One. 2020;15(2):1-19. [62] CHENG CF, KUO YH, HSU WC, et al. Local and remote ischemic preconditioning improves sprint interval exercise performance in team sport athletes. Int J Environ Res Public Health. 2021;18(20):10653. [63] INCOGNITO AV, BURR JF, MILLAR PJ. The effects of ischemic preconditioning on human exercise performance. Sports Med. 2017;46(4):531-544. [64] LARSEN FJ, SCHIFFER TA, BORNIQUEL S, et al. Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metab. 2011;13(2):149-159. [65] RASSAF T, TOTEACK M, HENDGEN-COTTA UB, et al. Circulating nitrite contributes to cardio protection by remote ischemic preconditioning. Circ Res. 2014;114(10): 1601-1610. [66] 余克威,吴毅.一氧化氮对线粒体生物功能的影响[J].神经损伤与功能重建, 2012,7(5):373-376. [67] DOWNEY JM, DAVIS AM, COHEN MV. Signaling pathways in ischemic preconditioning. Heart Fail Rev. 2007;12(3-4):181-188. [68] AMANN M, DEMPSEY JA. Locomotor muscle fatigue modifies central motor drive in healthy humans and imposes a limitation to exercise performance. J Physiol. 2008;586(1):161-173. [69] LEAL AK, YAMAUCHI K, KIM J, et al. Peripheral δ-opioid receptors attenuate the exercise pressor reflex. Am J Physiol Heart Circ Physiol. 2013;305(8):H1246-H1255. |
| [1] | Zhang Zixian, Xu Youliang, Wu Shaokui, Wang Xiangying. Effects of blood flow restriction training combined with resistance training on muscle indicators in college athletes: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1705-1713. |
| [2] | Chen Yuning, Jiang Ying, Liao Xiangyu, Chen Qiongjun, Xiong Liang, Liu Yue, Liu Tong. Buqi Huoxue Compounds intervene with the expression of related factors and autophagy related proteins in a rat model of cerebral ischemia/reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1152-1158. |
| [3] | Zhao Xiaoxuan, Liu Shuaiyi, Li Qi, Xing Zheng, Li Qingwen, Chu Xiaolei. Different exercise modalities promote functional recovery after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1248-1256. |
| [4] | Peng Yong, Hu Jiangping, Zhu Huan. Effects of low-load blood flow restriction exercise and high-intensity resistance exercise on the thigh microcirculation function of athletic young men [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(2): 393-401. |
| [5] | Yu Yanyan, Liu Juan, Yang Yue, Wang Qianhui, Li Shan, Jiang Jie. Moxibustion inhibits NLRP3/Caspase-1 pathway mediated cell pyroptosis and alleviates cerebral ischemia/reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(34): 5473-5479. |
| [6] | Yan Hailong, Huo Jiangtao, Zhou Wucheng, Bai Xuehua, Liang Yuanyuan. Application and mechanism of tissue flossing in sports and rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 464-471. |
| [7] | Dong Zhaojing, Jiang Dongting, Luo Xinjian, Yan Bing, Wang Yang, Ling Xiaoyu. Research progress on effects of wearable resistance training on lower limb movement ability [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4677-4684. |
| [8] | Kong Jianda, Xie Yingao, Ma Wen, Liu Youhan, Wang Qinglu. Mitochondrial dysfunction in Parkinson’s disease and the potential ameliorative effects of exercise [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(27): 4413-4420. |
| [9] | Dong Kuan, Xu Chengli, Tian Jing, Xu Changchun. Effects of endurance training with blood flow restriction on aerobic capacity, lower limb muscle strength, and sports performance: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3766-3772. |
| [10] | Kong Jianda, Xie Yingao, Chen Shijuan, Zhu Lei. Blood flow restriction training interventions for sarcopenia in older adults: biological mechanisms and proposed application protocols [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3743-3750. |
| [11] | Sun Yuan, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Zhang Yan. Effects of early and late aerobic exercise on right heart failure induced by monocrotaline in rats with pulmonary hypertension [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 177-185. |
| [12] | Xie Enli, Tao Huimin. Application trends of blood flow restriction training in clinical rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 258-262. |
| [13] | Dai Xinyu, Yan Jihong, Hua Lingjun, Zheng Xiaohong. Resistance exercise improves body composition in overweight and obese people: an umbrella review [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 267-271. |
| [14] | Zhang Yue, Guo Yingjie, Cheng Yang, Yang Tingting. Effect of blood flow restriction training on the fitness benefit of upper limb muscles [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2248-2253. |
| [15] | Zuo Huiwu, Geng Zhizhong, Chen Peng, Lin Xikai, Chen Jian. Knee function recovery in patients with anterior cruciate ligament reconstruction after blood flow restriction training: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(12): 1962-1968. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||



