中国组织工程研究 ›› 2023, Vol. 27 ›› Issue (33): 5385-5393.doi: 10.12307/2023.687
• 干细胞综述 stem cell review • 上一篇 下一篇
骨再生过程中炎症因素与牙髓干细胞的双向作用
徐溶蔚,王 浩,付秋月,兰兴明,杨 琨
- Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
收稿日期:2022-09-24接受日期:2022-10-12出版日期:2023-11-28发布日期:2023-03-30 -
通讯作者:杨琨,博士,副教授,遵义医科大学附属口腔医院牙周科,贵州省遵义市 563000 -
作者简介:徐溶蔚,女,2001年生,江苏省盐城市人,汉族。 王浩,男,2000年生,贵州省遵义市人,苗族。 -
基金资助:贵州省卫健委科学技术基金项目(gzwkj2022-169),项目负责人:杨琨;贵州省科学技术基金计划(黔科合基础[2020]1Y328),项目负责人:杨琨;遵义市科技计划项目[遵市科合HZ字(2021)302号],项目负责人:杨琨
Bidirectional interaction between inflammatory factors and dental pulp stem cells during bone regeneration
Xu Rongwei, Wang Hao, Fu Qiuyue, Lan Xingming, Yang Kun
- 遵义医科大学附属口腔医院牙周科,贵州省遵义市 563000
-
Received:2022-09-24Accepted:2022-10-12Online:2023-11-28Published:2023-03-30 -
Contact:Yang Kun, MD, Associate professor, Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Xu Rongwei, Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China Wang Hao, Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:Science and Technology Foundation of Guizhou Health Commission, No. gzwkj2022-169 (to YK); Guizhou Provincial Science and Technology Fund Plan, No. [2020]1Y328 (to YK); Zunyi Science and Technology Plan Project, No. HZ(2021)302 (to YK)
摘要:
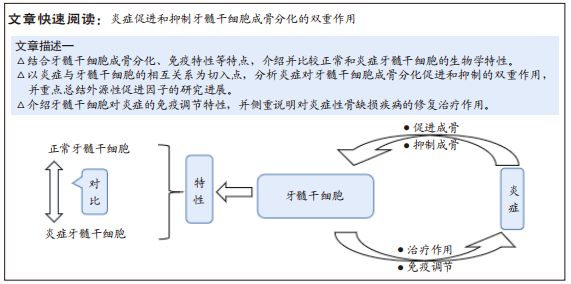
文题释义:
牙髓干细胞:是从牙髓组织分离的,来自神经嵴的外胚间充质干细胞,具有自我更新、免疫调节、多向分化潜能和易获得等特点,一定条件下可分化为神经样细胞、成牙本质细胞和成骨细胞等,目前已被报道在神经、口腔和骨骼等部位的再生医学价值,有良好的临床应用前景。炎症:具有血管系统的活体组织对损伤因子所发生的防御反应,通常引起局部组织呈现发红、肿胀、发热、疼痛和功能丧失的典型炎症反应体征。炎症作为一种病理过程,其特征在于损伤与防御的两面性。对牙髓干细胞而言,炎症微环境与其生物学特性关系密切,其中涉及复杂的炎症递质与信号通路的参与。
背景:牙髓干细胞具有多向分化的优良特性,其中成骨分化潜能在骨组织工程和再生治疗中有较好的前景,而炎症因素与其关系密切。
目的:综述骨再生过程中炎症因素与牙髓干细胞成骨分化双向作用的研究进展。
方法:在PubMed、中国知网和万方数据库中进行检索,中文检索词为“牙髓干细胞、炎症、成骨分化、骨再生”;英文检索词为“dental pulp stem cell*,dpsc*,inflam*,osteogenic differentiation,ossification,osteogenesis,bone formation”,分别检索2012-2022年发表的相关文献,最终纳入76篇进行综述分析。
结果与结论:①牙髓干细胞作为牙源性间充质干细胞,有着众人熟知的多向分化特性,其中成骨分化潜能是骨组织工程的研究热点。②炎症因素不论是作为细胞所处的微环境、可调节的免疫炎症反应、抑或是需要再生修复的炎症性骨组织缺损疾病,都与牙髓干细胞介导的骨再生有着密切的关联,近年来也不乏相关的研究。③文章选取了近十年以来相关的文献报道,发现即使是发炎牙髓组织来源的炎症牙髓干细胞仍然保留一定的成骨分化潜能,而进一步分析骨再生过程中炎症因素与牙髓干细胞的双向关系,发现炎症因素对于牙髓干细胞成骨分化的影响和牙髓干细胞对于炎症的免疫调节都具有促进和抑制的双重作用,这主要与炎症递质的种类、浓度及作用时间等有关,但是具体机制都尚不全面。④近来学者的研究热点主要集中于探索介导炎症环境下牙髓干细胞成骨分化的促进因素,尤其是除了炎症递质以外的外源性因素如药物等。⑤也有一些动物实验和少数临床试验利用牙髓干细胞抗炎促成骨的特性验证了牙髓干细胞对炎症性骨缺损疾病尤其是对牙周炎和关节炎的治疗作用。⑥目前,骨再生过程中炎症因素与牙髓干细胞成骨分化双向作用的相关机制部分的研究还有较大的空缺,未来仍需进一步研究验证。
https://orcid.org/0000-0001-8820-0426 (徐溶蔚);https://orcid.org/0000-0003-1673-3463 (王浩);
https://orcid.org/0000-0002-7720-8681 (杨琨)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
徐溶蔚, 王 浩, 付秋月, 兰兴明, 杨 琨. 骨再生过程中炎症因素与牙髓干细胞的双向作用[J]. 中国组织工程研究, 2023, 27(33): 5385-5393.
Xu Rongwei, Wang Hao, Fu Qiuyue, Lan Xingming, Yang Kun. Bidirectional interaction between inflammatory factors and dental pulp stem cells during bone regeneration[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5385-5393.
2.1.1 正常牙髓干细胞 牙髓干细胞是第一个明确的具有间充质特征和成骨分化潜能的牙源性间充质干细胞,起源于外胚层的神经嵴细胞[2,7]。目前首选酶消化法提取牙髓干细胞[8]。众所周知,在相应的诱导条件下,牙髓干细胞可分化为成牙本质细胞、神经细胞、成骨细胞等多种细胞类型,于牙髓再生、神经修复、组织工程以及转化医学等方面都具有应用价值[9]。其中在骨组织工程领域,研究者对牙髓干细胞的成骨分化潜能关注度很高,不少动物实验和体外实验都相继证明了牙髓干细胞胞外囊泡的促进成骨作用,以及细胞搭载的支架材料、注射凝胶、膜片等在骨再生治疗中的应用前景[6],尤其是针对相关支架的运用,有学者对近年来相关的临床前动物实验进行了回顾和Meta分析,肯定了牙髓干细胞的合成支架在治疗骨缺损中的有效性[10],说明牙髓干细胞成骨应用前景较为可观。与此同时,牙髓干细胞还具有免疫调节特性,它可以通过与T细胞等免疫细胞相互作用来调节免疫系统,还能分泌如白细胞介素6(interleukin-6,IL-6)、白细胞介素10(interleukin-10,IL-10),转化生长因子β(transforming growth factor-β,TGF-β)等抗炎因子,即使是已成骨分化的牙髓干细胞也有免疫调节能力[11],这一免疫特性对炎症反应乃至修复炎症性骨质缺损都有重大意义。
2.1.2 炎症牙髓干细胞 从炎症牙髓组织分离得到的牙髓干细胞为炎症牙髓干细胞(dental pulp stem cells from inflamed pulps,DPSCs-IPs),获取时也可以用酶消化法,且此法相比于生长法更有利于炎症牙髓干细胞成骨分化[12]。对于炎症牙髓干细胞的研究,最早可追溯至2010年学者第一次从患有不可逆性牙髓炎的恒牙牙髓中成功分离出干细胞,此后其相关的生物学特性等也逐渐被报道[13]。近年来的研究表明,炎症牙髓干细胞具有如免疫表型、集落形成能力、牙髓增殖再生、多向分化等牙源性间充质干细胞的典型特征[14],这使得长期被当成医疗废物扔弃的发炎的牙髓组织重获了临床应用价值,一些学者也针对如何人为地提高炎症牙髓干细胞的成骨潜力展开研究。例如由于Wnt信号通路在干细胞成骨分化过程中扮演了关键的角色,ZHONG等[15]构建了Wnt4过表达的炎症性牙髓干细胞(Wnt4-DPSC-IP)并用它治疗大鼠的牙槽骨缺损,发现其骨再生疗效优于一般的炎症牙髓干细胞,为推进日后炎症牙髓干细胞的应用提供了改进方案。类似的可以促进炎症牙髓干细胞成骨的还有中药黄芩苷[16]、人骨形态发生蛋白2(bone morphogenetic protein,BMP-2)[17]、矿物三氧化物凝聚体(mineral trioxide aggregate,MTA)等[18],详细的实验研究方法见表1。

炎症牙髓干细胞也保留了一定的免疫调节特性,比如LEE等[19]发现炎症牙髓干细胞可以抑制巨噬细胞肿瘤坏死因子α(tumor necrosis factor-α,TNF-α)的分泌,这种对巨噬细胞活化的免疫抑制作用可以避免过度炎症带来的损伤,同时吲哚胺2,3-双加氧酶(indoleamine2,3-Dioxygenase,IDO)作为其中重要的介质,其表达水平在炎症牙髓干细胞中也较正常牙髓干细胞(dental pulp stem cells-normal pulps,DPSC-NPs)高。但是在最近的体外研究发现,炎症牙髓干细胞的IDO活性和对T细胞增殖的免疫调节能力却比正常牙髓干细胞要低[14],此发现与LEE等学者的报道产生矛盾,可靠的结果需日后在体内进一步验证和探索。当然,针对受损的免疫抑制能力的改善,SONODA等[20]发现干扰素γ(interferon-γ,IFN-γ)可以作为调节剂有效地提高炎症牙髓干细胞在体外的免疫抑制力。得益于自身的免疫特性,炎症牙髓干细胞可以释放一些炎症相关的细胞因子,值得注意的是,炎症牙髓干细胞表达的白细胞介素6水平高于正常牙髓干细胞,同时由于白细胞介素6的促进作用导致其成骨能力提高,该结果不仅说明了炎症牙髓干细胞较好的成骨潜力,同时也指明了白细胞介素6增强成骨的应用价值[21]。
2.1.3 正常牙髓干细胞对比炎症牙髓干细胞 近来有不少针对正常和炎症牙髓中牙髓干细胞的生物学特性做的对比研究,文章选取NIE等[22]较为全面可信的研究结果作为参考,总结了目前已知的两者差异,见表2。

上述研究数据也有个别存在争议,如炎症牙髓干细胞的增殖速率和分化潜能,有人观察的结果是与正常牙髓干细胞相似[23],这些差异可能与炎症牙髓的状态、炎症来源、炎症强度和持续时间、分离培养方法以及个体之间遗传背景差异等众多因素有关,因此需要未来研究更大的样本量并控制相关因素来解释现下的矛盾之处。
从上述的特性和对比来看,正常牙髓干细胞和炎症牙髓干细胞都具有一定的免疫特性和成骨分化潜能,其他方向的分化潜能也各有强弱。正常牙髓干细胞的应用前景已被人们广泛认可,然而考虑到来源因素,炎症牙髓干细胞在临床上更易被获取应用,但是目前针对炎症性牙髓干细胞生物学特性的研究报道具有一定争议,相关的临床试验也较少,这些都是未来需要进一步研究的方向。
2.2 炎症因素对牙髓干细胞成骨分化的影响 炎症作为一把“双刃剑”,其诱导的损伤和修复往往不可分割[24]。牙髓组织常处于炎症环境中,使得牙髓干细胞的成骨分化能力也受此影响,比如除了众人熟知的牙髓炎以外,牙周炎也可以影响牙髓干细胞。据报道,患有侵袭性牙周炎的患者尽管牙周受损,但只要患牙内的牙髓活力尚未完全丧失,那么从中获取的牙髓干细胞就仍然保留有分化为成骨细胞表型的潜力[25],有研究近一步表明,这种成骨潜力不仅被保留,还由于促炎因子长期维持的炎症环境和应激反应蛋白的表达使得成骨分化潜能不减反增,这其中涉及到细胞骨架重塑的相关机制[26]。但是后来的研究得出,受牙周炎影响的牙髓干细胞成骨潜力没有改变,而软骨生成潜力、生长速度和迁移能力都降低[27]。由此看来,相关结论存在矛盾之处,牙周炎是一个复杂的且因人而异的炎症过程,针对牙周炎究竟对牙髓干细胞成骨分化造成何种影响,还需要更多样本量和更多牙周炎类型的实验去证实。尽管目前知晓的就炎症因素本身如何影响牙髓干细胞成骨的相关机制尚不全面,但是近来很多学者对一些外源性刺激因子如何介导并影响在炎症环境下牙髓干细胞的成骨分化进行了深入探索,包括各类介质、药物、生物材料、调控基因等。
2.2.1 促进成骨分化的炎症递质 炎症环境中存在很多炎症递质,它们是作用于牙髓干细胞成骨分化的重要媒介。白三烯B4(leukotriene B4,LTB4)是一种有效的炎症递质,属于中性粒细胞激活剂,它有刺激免疫反应、招募吞噬细胞、调节细胞因子等众多功能,研究发现白三烯B4能激活LTB4/BLT2机制来诱导牙髓干细胞产生矿化结节,还可通过Runt相关转录因子2(Runt-related transcription factor 2,RUNX2)和整合素结合唾液蛋白(integrinbinding sialoprotein,Ibsp)表达的增加促进细胞增殖和分化[28]。
白细胞介素10是一种抗炎因子,在炎症修复和骨骼稳态中起着关键作用,有研究表明白细胞介素10可激活牙髓干细胞中的氧化磷酸化过程(从糖酵解到氧化磷酸化),同时还能激活ERK/P-ERK/核转录因子κB通路,这些都与促进牙髓干细胞的成骨分化有关[29]。白细胞介素37作为炎症反应和免疫反应的天然抑制剂,与骨代谢相关的炎症因子存在一定联系,据报道[30],它可显著上调牙髓干细胞中牙本质涎磷蛋白(dentin sialophosphoprotein,DSPP)、Runt相关转录因子2、碱性磷酸酶和Osterix蛋白的蛋白质水平,这表明白细胞介素37能够正向诱导牙髓干细胞的成骨分化。
肿瘤坏死因子α是一种主要的促炎细胞因子,有研究表明肿瘤坏死因子α能刺激牙髓干细胞的成骨分化,此过程中,信号转导因子和转录激活因子3(signal transducer and activator of transcription 3,STAT3)在细胞核中表达增加,而在细胞质中表现出相反的现象,与此同时miR-21的表达增加与信号转导因子和转录激活因子3之间存在正反馈作用,二者共同促进了炎症微环境中牙髓干细胞的成骨分化[31]。肿瘤坏死因子α还可激活核转录因子κB通路来刺激牙髓干细胞成骨分化,当使用二硫氨基甲酸肽吡咯烷(核转录因子κB抑制剂)时,可以阻断肿瘤坏死因子α诱导的成骨分化[32]。然而有趣的发现是,低浓度肿瘤坏死因子α通过Wnt/β-catenin信号通路促进人牙髓干细胞的成骨分化,而高浓度则会抑制成骨,此过程与调节因子NAD-依赖性去乙酰化酶(NAD-dependent deacetylase sirtuin-1,SIRT1)密切相
关[33],同时高浓度抑制成骨的潜在机制远不止于此。目前已知的肿瘤坏死因子α双重作用于牙髓干细胞成骨分化的机制见图3。

2.2.2 促进成骨分化的其他外源因子 除了炎症递质的作用,其他的外源因子也可以诱导牙髓干细胞在炎症存在的前提下成骨分化,表3按年份顺序对此进行了总结,需要注明的是,当下能够促进牙髓干细胞成骨分化的外源因素很多,这里只纳入模拟炎症环境的研究[34-45]。

有学者发现,人β防御素1短基序Pep-B作为有抗炎和免疫调节特性的阳离子肽,对脂多糖模拟的炎症条件下的人牙髓干细胞有促进成骨作用[34],其过程抑制了核转录因子κB和P38丝裂原活化蛋白酶(mitogen-activated protein kinase,MAPK)途径介导的炎症的激活,说明人β防御素1短基序Pep-B有促进炎症环境下的人牙髓干细胞分化与成骨矿化的潜在价值。表皮调节素(epiregulin,EREG)是表皮生长因子家族成员,有学者证实表皮调节素在炎症环境下通过改善磷酸化MAPK和磷酸化的细胞外信号调节激酶(extracellular signal regulated kinases,ERK)的表达来调节MAPK信号通路,维持了人牙髓干细胞的成骨分化水平,增添了用生长因子调节牙髓干细胞于炎症中的成骨潜能的可行性[35]。
青蒿素作为熟知的抗疟疾药物,还能在缺氧和炎症环境下恢复牙髓干细胞的成骨分化能力,同时可以再次激活已失活的Wnt信号通路,具体机制与碳酸酐酶9的上调密切相关,该结果为青蒿素治疗牙髓干细胞奠定了重要基础[36]。姜黄素是一类抗炎抗菌的天然药剂,将其负载在支架上作用于炎症条件下的牙髓干细胞,不仅能发挥其抗炎抗菌的生物特性,还能诱导人牙髓干细胞的成骨矿化,这对于牙髓修复材料的开发提供了新思路[37]。人参皂苷Rb1(Ginsenoside Rb1,GR)是人参的提取物,常用于骨质缺损的修复,有学者将其负载于3D打印的陶瓷复合材料的支架上形成多孔GR支架,并将牙髓干细胞播种其上,植入于人造股骨缺损的兔模型中,发现GR支架不仅能够促进牙髓干细胞的增殖,还具有抑制炎症和促进骨修复的功效[38],类似的研究还有白杨素支架[39],这些都是药物和支架联合运用的良好成果。倍他米松作为糖皮质激素是一种常见的抗炎药物,有文献指出其可抑制核转录因子κB信号的激活帮助牙髓干细胞具有抗炎作用[40],还能在炎症条件下促进人牙髓干细胞成骨,但是需要强调的是,它对乳牙牙髓干细胞有诱导破骨的趋势,而对牙髓干细胞却没有此趋势,两者为何有此区别值得深入研究。雷帕霉素是一种新型大环内酯类免疫抑制剂,它除了抗免疫排斥反应之外,对牙髓干细胞炎症状态下的成骨也具有一定的促进作用,具体机制主要涉及ERK1/2及JNK通路的抑制等[41]。这些研究体现出用药物提高牙髓干细胞炎症下成骨的策略广受学者关注。
磷酸钙水泥作为牙科修复材料一直被不断地改良,有人构建出含有β-磷酸三钙(β-tricalcium phosphate,β-TCP)的新型双相磷酸钙骨水泥[42-43],相比传统的磷酸钙骨水泥,β-磷酸三钙可以使巨噬细胞向M2表型极化从而避免炎症,同时双相磷酸钙骨水泥更能促进人牙髓干细胞的分化与矿物沉积。胶原膜是治疗牙周和种植体周骨缺损、引导骨再生的重要材料,用能影响干细胞增殖分化的氧化石墨烯涂层可提高其生物性能,研究发现涂有氧化石墨烯的胶原膜对牙髓干细胞无细胞毒性,不仅促进人牙髓干细胞的成骨分化,还能控制肿瘤坏死因子α和环氧合酶2介导的炎症,相较于传统的胶原膜无疑是一次成功的修饰和改良[44]。
基因调控对于炎症环境中牙髓干细胞的成骨分化也值得探讨。据报道,SNHG7作为一种可以增强干细胞成骨分化的长非编码RNA,能促进炎症环境下miR-6512-3p的表达来改善人牙髓干细胞的成骨分化,这为牙髓干细胞成骨发掘了潜在的基因调节的靶点[45]。MicroRNA let-7c-5p作为一种内源性非编码调控因子,其对炎症环境下的牙髓干细胞也具有抗炎促成骨的积极作用等[46]。
2.2.3 抑制成骨分化 相较于促进因素,抑制牙髓干细胞炎症下成骨分化的外源性因素的研究较少。炎症过程被认为是牙本质-牙髓复合物愈合和再生的先决条件,早期炎症反应是牙髓细胞的保护机制[24]。然而有研究发现,长期接触促炎因子干扰素γ显著减弱了牙髓干细胞中碱性磷酸酶的活性(成骨标志),而当使用干扰素γ抑制剂预处理时碱性磷酸酶的活性得到了改善[47],这表明长期炎症会损害牙髓干细胞的成骨矿化能力。
肿瘤坏子因子α作为重要炎症递质之一,在低浓度时可以促进牙髓干细胞的成骨分化,在高浓度下则变为抑制作用[48]。FENG等[49]发现,肿瘤坏子因子α可以通过增加RAC1蛋白的表达发挥抑制作用。肿瘤坏死因子α刺激基因6(tumor necrosis factorαstimulated gene 6,TSG-6)的表达水平以及骨形态发生蛋白4/Smad通路也是潜在机制之一,肿瘤坏子因子α诱导TSG-6产生,当TSG-6的表达水平增加时,骨形态发生蛋白4/Smad信号通路相关蛋白质随之减少,成骨标志物的表达也减少,在TSG-6基因敲除后则相反,也就是说,高水平的肿瘤坏子因子α能促进TSG-6的表达来抑制牙髓干细胞的骨形态发生蛋白4 / Smad信号通路,并随之抑制其成骨分化[50]。
G蛋白信号转导激活因子(activators of G protein signaling 3,AGS3)与环磷腺苷效应元件结合蛋白(cAMP-response element binding protein,CREB)的磷酸化有关,它可以负向调控cAMP/PKA/CREB信号通路从而抑制在肿瘤坏死因子α影响下的牙髓干细胞的成骨分化[51],但相关机制局限于肿瘤坏死因子α介导的炎症条件,对于复杂的炎症环境来说略显片面。炎症环境中往往存在一些促进细胞凋亡的因子,B淋巴细胞瘤-2基因(B-cell lymphoma 2,Bcl-2),作为一种抗凋亡基因正好可以提高牙髓干细胞在炎症中的存活率,然而更重要的发现是Bcl-2基因在抗凋亡的同时还损害了牙髓干细胞的成骨分化潜力,这对未来Bcl-2基因的运用明显设限[52]。有些抑制因素的研究,虽未设有炎症前提,但其本身可以触发炎症因子的释放,比如樟脑醌作为一种用于牙科材料的聚合粘接光引发剂,有研究表明其可以诱导牙髓产生促炎因子以引发炎症,同时还能抑制牙髓干细胞的成骨分化,这也启示临床应避免含光引发剂的材料直接盖髓[53]。
总之,炎症因素对牙髓干细胞成骨分化具有双重作用,其中涉及了复杂的炎症递质、细胞因子和信号通路。早期炎症是牙髓干细胞的一种保护机制,可以促进牙髓干细胞成骨分化以抵御骨质破坏,但长期持续的、高强度的炎症则会抑制其成骨分化。近来的研究多集中于探索牙髓干细胞炎症环境下成骨的促进因素,尤其集中于非炎症介质的其他外源因子,由此看来,如何让牙髓干细胞在炎症条件下保持高效的成骨分化能力是当下的一大研究热点。
2.3 牙髓干细胞对炎症的作用
2.3.1 调节炎症的免疫活性 牙髓干细胞的免疫调节能力高度可塑,且由微环境中炎症递质的种类和浓度决定[54]。已有学者较为详细地阐述了口腔间充质干细胞对于炎症微环境的双重反应: 较低炎症水平下促进炎症产生和骨吸收,较高炎症水平则出现相反的现象,即抑制炎症产生和骨吸收,其过程涉及了肥大细胞、树突状细胞、自然杀伤细胞、巨噬细胞(M1和M2表型)、T细胞和B细胞等,干细胞还可以通过细胞间直接接触和微环境中可溶性的细胞因子发挥其免疫调节作用,包括转化生长因子β1、前列腺素E2、吲哚胺2,3-二加氧酶(IDO)、一氧化氮、白细胞介素1、白细胞介素6和白细胞介素10 [55]。不同的表面蛋白可存在不同的调节机制。程序性细胞死亡配体1(programmed death-ligand 1,PD-L1)是抑制免疫系统激活的重要蛋白,炎症微环境下牙髓干细胞的程序性细胞死亡配体1上调,其与程序性死亡受体PD1结合可调节T细胞反应来协调炎症,Fas / FasL途径也参与其中[56],两者的协同作用见图4。
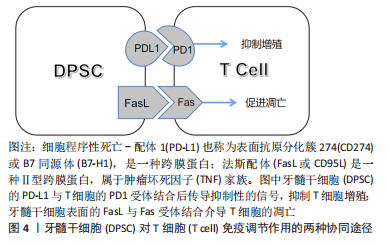
Fas配体(Fas ligand,FasL)是肿瘤坏死因子家族中的一种Ⅱ型跨膜蛋白,与Fas受体结合后可在许多细胞类型中诱导凋亡过程。牙髓干细胞可以表达Fas配体以诱导T细胞凋亡[57](目前只在骨髓间充质干细胞中报道过详细机制[58]),同时Fas/FasL通路还被证明有促进人牙髓干细胞软骨分化的作用[59]。由此可见,在炎症环境下牙髓干细胞的免疫调节机制极其复杂,不同免疫调节途径间存在串扰和协同作用,这些无疑是未来研究炎症调节机制的重要方向。
2.3.2 治疗口腔炎症 由于牙髓干细胞的免疫调节和抗炎潜力,将牙髓干细胞用于治疗炎症性疾病已经成为了一个新兴的、涵盖多学科的研究领域。在口腔范围内,牙髓干细胞的牙髓再生作用对不可逆性牙髓炎有着天然的治疗优势,已有临床试点研究展现出这种再生性治疗的可行性[60-61]。与此同时,牙髓干细胞的成骨分化潜能对于炎症性疾病导致的骨组织缺损的再生修复也值得关注,下文对将牙髓干细胞用于治疗口腔炎症性骨缺损的相关研究进展作总结。
牙髓治疗失败易造成根管内细菌感染,引发根尖周炎症而破坏根尖周骨组织,虽然根管治疗是目前普遍的治疗方案,但是再生性治疗更为理想。有学者通过回顾3份以注射性支架递送的、源于人第三磨牙的正常牙髓干细胞为治疗手段的根尖周病变病例,发现在牙髓干细胞的诱导下,患者不仅能形成成熟的根尖、愈合根尖周的大面积骨缺损,还在没有其他并发症的前提下比传统治疗方式更加省时[62]。这项结果为临床治疗方案提供了新思路,是牙髓干细胞运用的良好开端,但稍有不足的是缺乏了一些炎症水平变化的指标,因而无从知晓牙髓干细胞的免疫调节特性是否在此发挥作用。
牙周炎是龈下生物膜引起的炎症和免疫反应,会导致牙槽骨和牙周韧带的不可逆性破坏[63]。针对其治疗,相比于牙周膜干细胞来源有限、发炎时免疫调节能力受损的缺点[64],牙髓干细胞更具有应用价值。HU等[65]建立牙周炎猪模型,发现正常的人牙髓干细胞注射和人牙髓干细胞片植入治疗可诱导牙周组织再生,且人牙髓干细胞片疗效更佳。在临床应用中,也有学者用患者自体需拔除的牙的正常牙髓组织作为牙髓干细胞来源,对牙周骨缺陷的案例进行再生治疗,最终的骨增量结果也对牙髓干细胞的疗效给予了肯定[66]。当然,炎症牙髓干细胞作为临床上更易获取的一类干细胞,其对牙周炎所致骨缺损的修复作用也在逐渐被人们探索,比如同一个研究团队先后在临床牙周-牙髓病变患者和小猪标准牙周炎模型中,分别用人和猪自体的炎症牙髓干细胞和支架材料结合进行治疗,结果都显示出炎症牙髓干细胞成骨分化能力和良好的牙周骨质修复效果[67-68],为炎症牙髓干细胞未来应用于临床当中提供了证据。除了牙髓干细胞本身,还有研究表明牙髓干细胞分泌的外泌体也对牙周炎具有一定的治疗作用,实验中,牙髓干细胞来源的外泌体不仅可以加速牙周炎小鼠模型的牙槽骨愈合,还可以将巨噬细胞从促炎表型转化为抗炎表型以调节炎症反应,这种抗炎促成骨的双重作用为牙周炎的治疗提供了新方案[69]。
颞下颌关节骨关节炎(temporomandibular joint osteoarthritis,TMJOA)是口腔颌面部的一种常见的慢性炎症性退行性病变,严重炎症、进行性软骨和髁突骨骼破坏是TMJOA典型病理变化。目前其病因多样且机制未明,炎症因素和机械应力等都与它密切相关,而继发性炎症因素是疾病发生和进展的关键。由于目前对于此病多为药物治疗,虽能缓解症状但不能从根本上修复骨软骨缺损,因此近来不乏干细胞再生疗法的相关报道[70]。有动物实验(在大鼠关节腔内注射化学试剂建立TMJOA模型)发现正常人牙髓干细胞可以减少进行性TMJOA中肿瘤坏死因子α和干扰素γ的表达,并且可缓解痛觉过敏和滑膜炎症,减轻软骨基质降解,并诱导骨再生;由于调节炎症的转录激活因子1 (STAT1) 参与了TMJOA进展过程中基质金属蛋白酶的调节,体外研究中也发现牙髓干细胞可以有效抑制转录激活因子1的激活,导致基质金属蛋白酶3和基质金属蛋白酶13的下调,从而缓解炎症,这证实了牙髓干细胞对TMJOA的局部注射疗效[71]。另外,针对机械应力如颞下颌关节紊乱病导致的TMJOA,牙髓干细胞可以通过分泌一些如肝细胞生长因子等多种治疗因子,不仅能够减轻滑膜和颞肌的炎症,还能再生修复机械应力损伤的髁突软骨和软骨下骨质,由此看来,牙髓干细胞有望成为重度TMJOA患者的再生治疗方案[72]。
2.3.3 治疗全身炎症 不仅在口腔范围内,牙髓干细胞对全身炎性疾病也有极大的治疗价值。比如,作为备受关注且当下仍在传播的感染性肺部炎症——2019年新型冠状病毒病(Corona Virus Disease 2019,COVID-19),牙髓干细胞就被报道可作为其潜在的干细胞治疗策略[73]。诸如此类的研究还有很多[4],此处仅罗列可造成(软)骨破坏的全身炎症性疾病。
骨关节炎是一种伴有软骨丢失的退行性和炎性关节疾病,软骨的完全再生一直是治疗难点。而牙髓干细胞的软骨分化、免疫调节等特性对骨关节炎有着潜在治疗价值,诸多实验皆已证明其应用潜力[74]。深究其治疗机制,发现牙髓干细胞在体外可以通过抑制巨噬细胞的活化来减轻炎症,其中涉及了肝细胞生长因子、转化生长因子β1的分泌和MAPK信号的多条传导途径的失活;而在兔骨关节炎模型的体内实验也进一步印证了巨噬细胞被抑制的结果,同时还发现牙髓干细胞可以减轻软骨损伤和骨软骨病变,最终达到修复骨损伤和抗炎的疗效[75]。
类风湿性关节炎是可导致关节软骨和骨破坏的自身免疫性疾病,具有滑膜增生和慢性炎症的特点。研究类风湿性关节炎的小鼠模型[由胶原Ⅱ型抗体(CAIA)诱导]和来自人乳牙牙髓干细胞的培养基(SHED-CM)的学者发现,SHED-CM可以降低小鼠模型的促炎细胞因子的水平、抑制破骨细胞的形成和骨质破坏,并诱导抗炎M2型巨噬细胞的极化[76],说明在SHED-CM中存在乳牙牙髓干细胞分泌的因子,它们对类风湿性关节炎具有潜在的疗效。乳牙牙髓干细胞是牙髓干细胞的一种特殊类型,可以由此类推牙髓干细胞具有修复类风湿性关节炎的潜力。
综上,牙髓干细胞调节炎症的免疫活性机制还具有很大的研究空间,尤其针对不同调节途径之间的相互协同和串扰作用。牙髓干细胞的成骨分化协同抗炎潜能对于治疗炎症性骨缺损的意义重大,然而相关的临床应用研究却不是很多,见图5。
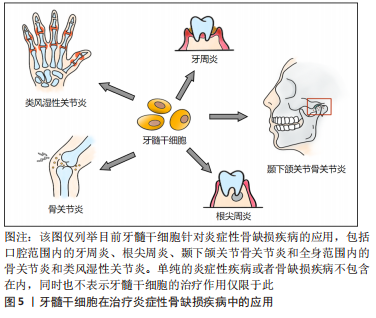
从近期研究来看,牙周炎和关节炎是学者近来较为关注的炎症性骨缺损疾病,这或许也是牙髓干细胞抗炎促成骨功效比较适合的应用方向。

| [1] XUE N, DING X, HUANG R, et al. Bone tissue engineering in the treatment of bone defects. Pharmaceuticals (Basel). 2022;15(7):879. [2] ERCAL P, PEKOZER GG, KOSE GT. Dental stem cells in bone tissue engineering: current overview and challenges. Adv Exp Med Biol. 2018;1107:113-127. [3] FAWZY EK, ELSALAWY R, IBRAHIM N, et al. The dental pulp stem/progenitor cells-mediated inflammatory-regenerative axis. Tissue Eng Part B Rev. 2019;25(5):445-460. [4] YAMADA Y, NAKAMURA-YAMADA S, KUSANO K, et al. Clinical potential and current progress of dental pulp stem cells for various systemic diseases in regenerative medicine: a concise review. Int J Mol Sci. 2019. doi: 10.3390/ijms20051132. [5] 刘雪梅,刘尧,陈旭.牙源性干细胞与炎症相互作用研究进展[J].中国实用口腔科杂志,2015,8(1):52-55. [6] 魏代福,魏馨蕊,赵戬.与牙髓干细胞相关的骨组织工程研究进展[J].中国医学创新,2022,19(14):171-175. [7] HUANG X, LI Z, LIU A, et al. Microenvironment influences odontogenic mesenchymal stem cells mediated dental pulp regeneration. Front Physiol. 2021;12:656588. [8] PILBAUEROVÁ N, SOUKUP T, SUCHÁNKOVÁ KT, et al. Enzymatic isolation, amplification and characterization of dental pulp stem cells. Folia Biol (Praha). 2019;65(3):124-133. [9] NUTI N, CORALLO C, CHAN BM, et al. Multipotent differentiation of human dental pulp stem cells: a literature review. Stem Cell Rev Rep. 2016;12(5):511-523. [10] LORUSSO F, INCHINGOLO F, DIPALMA G, et al. Synthetic scaffold/dental pulp stem cell (dpsc) tissue engineering constructs for bone defect treatment: an animal studies literature review. Int J Mol Sci. 2020. doi:10.3390/ijms21249765. [11] HOSSEIN-KHANNAZER N, HASHEMI SM, NAMAKI S, et al. Study of the immunomodulatory effects of osteogenic differentiated human dental pulp stem cells. Life Sci. 2019;216:111-118. [12] GOPINATH VK, SOUMYA S, JAYAKUMAR MN. Osteogenic and odontogenic differentiation potential of dental pulp stem cells isolated from inflamed dental pulp tissues (I-DPSCs) by two different methods. Acta Odontol Scand. 2020;78(4): 281-289. [13] 赵华翔,赵珊梅,辛欣,等.炎症牙髓干细胞:起步研究与未来发展[J].中国组织工程研究,2014,18(23):3756-3761. [14] INOSTROZA C, VEGA-LETTER AM, BRIZUELA C, et al. Mesenchymal stem cells derived from human inflamed dental pulp exhibit impaired immunomodulatory capacity in vitro. J Endod. 2020;46(8):1091-1098. [15] ZHONG T, GAO Y, QIAO H, et al. Elevated osteogenic potential of stem cells from inflammatory dental pulp tissues by Wnt4 overexpression for treating bone defect in rats. Ann Palliat Med. 2020;9(5):2962-2969. [16] 薛晶.中药黄芩苷对炎症牙髓干细胞增殖及牙向/骨向分化的影响及机制研究[D].南京:南京医科大学,2015. [17] 邓云贞,李珏丹,魏虹,等.BMP-2促进人炎症牙髓干细胞骨向诱导分化的体外研究[J].口腔生物医学,2015,6(2):71-77. [18] WANG Y, YAN M, FAN Z, et al. Mineral trioxide aggregate enhances the odonto/osteogenic capacity of stem cells from inflammatory dental pulps via NF-κB pathway. Oral Dis. 2014;20(7):650-658. [19] LEE S, ZHANG QZ, KARABUCAK B, et al. DPSCs from inflamed pulp modulate macrophage function via the TNF-α/IDO Axis. J Dent Res. 2016;95(11):1274-1281. [20] SONODA S, YAMAZA H, MA L, et al. Interferon-gamma improves impaired dentinogenic and immunosuppressive functions of irreversible pulpitis-derived human dental pulp stem cells. Sci Rep. 2016;6:19286. [21] PARK YT, LEE SM, KOU X, et al. The role of interleukin 6 in osteogenic and neurogenic differentiation potentials of dental pulp stem cells. J Endod. 2019; 45(11):1342-1348. [22] NIE SC, YANG K, LUAN NN, et al. Unveiling the differences in biological properties of dental pulp stem cells from normal and inflamed pulp: a comprehensive comparative study. Med Sci Monit. 2022;28:e934511. [23] PEREIRA LO, RUBINI MR, SILVA JR, et al. Comparison of stem cell properties of cells isolated from normal and inflamed dental pulps. Int Endod J. 2012;45(12): 1080-1090. [24] COOPER PR, HOLDER MJ, SMITH AJ. Inflammation and regeneration in the dentin-pulp complex: a double-edged sword. J Endod. 2014;40(4 Suppl):S46-S51. [25] SUN HH, CHEN B, ZHU QL, et al. Investigation of dental pulp stem cells isolated from discarded human teeth extracted due to aggressive periodontitis. Biomaterials. 2014;35(35):9459-9472. [26] TOMASELLO L, MAUCERI R, COPPOLA A, et al. Mesenchymal stem cells derived from inflamed dental pulpal and gingival tissue: a potential application for bone formation. Stem Cell Res Ther. 2017;8(1):179. [27] FAGEEH HN. Preliminary evaluation of proliferation, wound healing properties, osteogenic and chondrogenic potential of dental pulp stem cells obtained from healthy and periodontitis affected teeth. Cells. 2021. doi:10.3390/cells10082118. [28] DA SF, DE CAMPOS CLG, DE OLIVEIRA F, et al. Leukotriene B(4) loaded in microspheres regulate the expression of genes related to odontoblastic differentiation and biomineralization by dental pulp stem cells. BMC Oral Health. 2022;22(1):45. [29] YUAN L, YOU H, QIN N, et al. Interleukin-10 modulates the metabolism and osteogenesis of human dental pulp stem cells. Cell Reprogram. 2021;23(5):270-276. [30] LI N, YAN M, CHEN Y, et al. Extracellular IL-37 promotes osteogenic and odontogenic differentiation of human dental pulp stem cells via autophagy. Exp Cell Res. 2021;407(1):112780. [31] XU K, XIAO J, ZHENG K, et al. MiR-21/STAT3 signal is involved in odontoblast differentiation of human dental pulp stem cells mediated by TNF-α. Cell Reprogram. 2018;20(2):107-116. [32] FENG X, FENG G, XING J, et al. TNF-α triggers osteogenic difion of human dental pulp stem cells via the NF-κB signalling pathway. Cell Biol Int. 2013;37(12):1267-1275. [33] FENG G, ZHENG K, SONG D, et al. SIRT1 was involved in TNF-α-promoted osteogenic differentiation of human DPSCs through Wnt/β-catenin signal. In Vitro Cell Dev Biol Anim. 2016;52(10):1001-1011. [34] SHI J, HU Z, ZHOU Y, et al. Therapeutic potential of synthetic human β-defensin 1 short motif pep-B on lipopolysaccharide-stimulated human dental pulp stem cells. Mediators Inflamm. 2022;2022:6141967. [35] RAN R, YANG H, CAO Y, et al. Depletion of EREG enhances the osteo/dentinogenic differentiation ability of dental pulp stem cells via the p38 MAPK and Erk pathways in an inflammatory microenvironment. BMC Oral Health. 2021;21(1):314. [36] HU HM, MAO MH, HU YH, et al. Artemisinin protects DPSC from hypoxia and TNF-α mediated osteogenesis impairments through CA9 and Wnt signaling pathway. Life Sci. 2021;277:119471. [37] ALIPOUR M, FADAKAR S, AGHAZADEH M, et al. Synthesis, characterization, and evaluation of curcumin-loaded endodontic reparative material. J Biochem Mol Toxicol. 2021;35(9):e22854. [38] CHEN CY, SHIE MY, LEE AK, et al. 3D-printed ginsenoside rb1-loaded mesoporous calcium silicate/calcium sulfate scaffolds for inflammation inhibition and bone regeneration. Biomedicines. 2021;9(8):907. [39] ALIPOUR M, POUYA B, AGHAZADEH Z, et al. The antimicrobial, antioxidative, and anti-inflammatory effects of polycaprolactone/gelatin scaffolds containing chrysin for regenerative endodontic purposes. Stem Cells Int. 2021;2021:3828777. [40] WANG D, ZHU NX, QIN M, et al. Betamethasone suppresses the inflammatory response in LPS-stimulated dental pulp cells through inhibition of NF-κB. Arch Oral Biol. 2019;98:156-163. [41] 朱钊,宋佳欣,单兆臣.雷帕霉素在炎症状态下对牙髓干细胞分化能力的影响研究[J].北京口腔医学,2019,27(4):186-190. [42] GU Y, XIE X, ZHUANG R, et al. A biphasic calcium phosphate cement enhances dentin regeneration by dental pulp stem cells and promotes macrophages M2 phenotype in vitro. Tissue Eng Part A. 2021;27(17-18): 1113-1127. [43] GU Y, ZHUANG R, XIE X, et al. Osteogenic stimulation of human dental pulp stem cells with self-setting biphasic calcium phosphate cement. J Biomed Mater Res B Appl Biomater. 2020;108(4):1669-1678. [44] RADUNOVIC M, DE COLLI M, DE MARCO P, et al. Graphene oxide enrichment of collagen membranes improves DPSCs differentiation and controls inflammation occurrence. J Biomed Mater Res A. 2017;105(8):2312-2320. [45] CHEN W. SNHG7 promotes the osteo/dentinogenic differentiation ability of human dental pulp stem cells by interacting with hsa-miR-6512-3p in an inflammatory microenvironment. Biochem Biophys Res Commun. 2021;581:46-52. [46] YUAN H, ZHAO H, WANG J, et al. MicroRNA let-7c-5p promotes osteogenic differentiation of dental pulp stem cells by inhibiting lipopolysaccharide-induced inflammation via HMGA2/PI3K/Akt signal blockade. Clin Exp Pharmacol Physiol. 2019;46(4):389-397. [47] STROJNY C, BOYLE M, BARTHOLOMEW A, et al. Interferon gamma-treated dental pulp stem cells promote human mesenchymal stem cell migration in vitro. J Endod. 2015;41(8):1259-1264. [48] QIN Z, FANG Z, ZHAO L, et al. High dose of TNF-α suppressed osteogenic differentiation of human dental pulp stem cells by activating the Wnt/β-catenin signaling. J Mol Histol. 2015;46(4-5):409-420. [49] FENG G, SHEN Q, LIAN M, et al. RAC1 regulate tumor necrosis factor-α-mediated impaired osteogenic differentiation of dental pulp stem cells. Dev Growth Differ. 2015;57(7):497-506. [50] WANG Y, YUAN S, SUN J, et al. Inhibitory effect of the TSG-6 on the BMP-4/Smad signaling pathway and odonto/osteogenic differentiation of dental pulp stem cells. Biomed Pharmacother. 2020;128:110266. [51] XING J, LIAN M, SHEN Q, et al. AGS3 is involved in TNF-αmedicated osteogenic differentiation of human dental pulp stem cells. Differentiation. 2015;89(5):128-136. [52] HENG B C, YE X, LIU Y, et al. Effects of recombinant overexpression of bcl2 on the proliferation, apoptosis, and osteogenic/odontogenic differentiation potential of dental pulp stem cells. J Endod. 2016;42(4): 575-583. [53] KIM RH, WILLIAMS DW, BAE S, et al. Camphorquinone inhibits odontogenic differentiation of dental pulp cells and triggers release of inflammatory cytokines. J Endod. 2013;39(1):57-61. [54] WANG Y, CHEN X, CAO W, et al. Plasticity of mesenchymal stem cells in immunomodulation:pathological and therapeutic implications. Nat Immunol. 2014;15(11):1009-1016. [55] ZHOU LL, LIU W, WU YM, et al. Oral mesenchymal stem/progenitor cells:the immunomodulatory masters. Stem Cells Int. 2020;2020:1327405. [56] DI TINCO R, BERTANI G, PISCIOTTA A, et al. Role of PD-L1 in licensing immunoregulatory function of dental pulp mesenchymal stem cells. Stem Cell Res Ther. 2021;12(1):598. [57] ZHAO Y, WANG L, JIN Y, et al. Fas ligand regulates the immunomodulatory properties of dental pulp stem cells. J Dent Res. 2012;91(10):948-954. [58] AKIYAMA K, CHEN C, WANG D, et al. Mesenchymal-stem-cell-induced immunoregulation involves FAS-ligand-/FAS-mediated T cell apoptosis. Cell Stem Cell. 2012;10(5):544-555. [59] PISCIOTTA A, BERTANI G, BERTONI L, et al. Modulation of cell death and promotion of chondrogenic differentiation by Fas/FasL in human dental pulp stem cells (hDPSCs). Front Cell Dev Biol. 2020;8:279. [60] MEZA G, URREJOLA D, SAINT J N, et al. Personalized cell therapy for pulpitis using autologous dental pulp stem cells and leukocyte platelet-rich fibrin: a case report. J Endod. 2019;45(2):144-149. [61] NAKASHIMA M, IOHARA K, MURAKAMI M, et al. Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: a pilot clinical study. Stem Cell Res Ther. 2017;8(1):61. [62] SHIEHZADEH V, AGHMASHEH F, SHIEHZADEH F, et al. Healing of large periapical lesions following delivery of dental stem cells with an injectable scaffold: new method and three case reports. Indian J Dent Res. 2014;25(2):248-253. [63] KWON T, LAMSTER IB, LEVIN L. Current concepts in the management of periodontitis. Int Dent J. 2021;71(6):462-476. [64] LIU D, XU J, LIU O, et al. Mesenchymal stem cells derived from inflamed periodontal ligaments exhibit impaired immunomodulation. J Clin Periodontol. 2012;39(12):1174-1182. [65] HU J, CAO Y, XIE Y, et al. Periodontal regeneration in swine after cell injection and cell sheet transplantation of human dental pulp stem cells following good manufacturing practice. Stem Cell Res Ther. 2016;7(1):130. [66] AIMETTI M, FERRAROTTI F, GAMBA M N, et al. Regenerative treatment of periodontal intrabony defects using autologous dental pulp stem cells: a 1-year follow-up case series. Int J Periodontics Restorative Dent. 2018;38(1):51-58. [67] LI Y, ZHAO S, NAN X, et al. Repair of human periodontal bone defects by autologous grafting stem cells derived from inflammatory dental pulp tissues. Stem Cell Res Ther. 2016;7(1):141. [68] LI Y, NAN X, ZHONG TY, et al. Treatment of periodontal bone defects with stem cells from inflammatory dental pulp tissues in miniature swine. Tissue Eng Regen Med. 2019;16(2):191-200. [69] SHEN Z, KUANG S, ZHANG Y, et al. Chitosan hydrogel incorporated with dental pulp stem cell-derived exosomes alleviates periodontitis in mice via a macrophage-dependent mechanism. Bioact Mater. 2020;5(4):1113-1126. [70] ZHAO Y, XIE L. An update on mesenchymal stem cell-centered therapies in temporomandibular joint osteoarthritis. Stem Cells Int. 2021;2021:6619527. [71] CUI S J, ZHANG T, FU Y, et al. DPSCs attenuate experimental progressive tmj arthritis by inhibiting the STAT1 pathway. J Dent Res. 2020;99(4):446-455. [72] NOGASAWARA N, KANK F, HASHIMOTO N, et al. Factors secreted from dental pulp stem cells show multifaceted benefits for treating experimental temporomandibular joint osteoarthritis. Osteoarthritis Cartilage. 2020;28(6): 831-841. [73] ZAYED M, IOHARA K. Immunomodulation and regeneration properties of dental pulp stem cells: a potential therapy to treat coronavirus disease 2019. Cell Transplant. 2020;29:2138944793. [74] FERNANDES TL, CORTEZ D SJ, FRISENE I, et al. Systematic review of human dental pulp stem cells for cartilage regeneration. Tissue Eng Part B Rev. 2020;26(1):1-12. [75] LI PL, WANG YX, ZHAO ZD, et al. Clinical-grade human dental pulp stem cells suppressed the activation of osteoarthritic macrophages and attenuated cartilaginous damage in a rabbit osteoarthritis model. Stem Cell Res Ther. 2021; 12(1):260. [76] ISHIKAWA J, TAKAHASHI N, MATSUMOTO T, et al. Factors secreted from dental pulp stem cells show multifaceted benefits for treating experimental rheumatoid arthritis. Bone. 2016;83:210-219. |
| [1] | 方兴艳, 田侦丽, 赵哲仪, 文平, 谢婷婷. 亚砷酸钠对人脐静脉内皮细胞损伤及鞘氨醇激酶1/1-磷酸鞘氨醇信号轴的影响[J]. 中国组织工程研究, 2023, 27(在线): 1-7. |
| [2] | 农复香, 蒋志雄, 李英豪, 许文聪, 施智兰, 罗 慧, 张晴朗, 钟 爽, 唐梅文. 外泌体调控铁死亡在疾病诊断治疗中的应用与作用[J]. 中国组织工程研究, 2023, 27(在线): 1-10. |
| [3] | 郭淑慧, 杨晔, 江杨洋, 许建文. 神经源性膀胱miRNA-mRNA调控网络的筛选与验证[J]. 中国组织工程研究, 2023, 27(在线): 1-8. |
| [4] | 潘钟杰, 秦志鸿, 郑铁军, 丁晓飞, 廖世杰. 股骨头坏死发病机制中非编码RNA的靶标性[J]. 中国组织工程研究, 2023, 27(9): 1441-1447. |
| [5] | 蔡志浩, 谢召勇. 股骨颈前倾角测量评估:如何建立统一的方法和标准[J]. 中国组织工程研究, 2023, 27(9): 1448-1454. |
| [6] | 党 祎, 杜成砚, 姚红林, 袁能华, 曹 金, 熊 山, 张顶梅, 王 信. 激素型骨坏死与氧化应激[J]. 中国组织工程研究, 2023, 27(9): 1469-1476. |
| [7] | 杨芷姗, 唐正龙. Hippo信号通路中的核心因子YAP/TAZ参与骨形成的作用与机制[J]. 中国组织工程研究, 2023, 27(8): 1264-1271. |
| [8] | 王 继, 张 敏, 杨中亚, 张 龙. 体力活动干预2型糖尿病肌少症的研究现状[J]. 中国组织工程研究, 2023, 27(8): 1272-1277. |
| [9] | 宋荷花, 魏在荣. 糖尿病的周围神经病变:研究与治疗[J]. 中国组织工程研究, 2023, 27(8): 1278-1285. |
| [10] | 聂晨晨, 苏凯奇, 高 静, 凡勇福, 阮晓迪, 袁 洁, 段昭远, 冯晓东. 环状RNA调控脑缺血发病的作用与机制[J]. 中国组织工程研究, 2023, 27(8): 1286-1291. |
| [11] | 高 煜, 韩佳慧, 葛 新. 脊髓缺血再灌注损伤后的免疫炎性微环境[J]. 中国组织工程研究, 2023, 27(8): 1300-1305. |
| [12] | 杨九杰, 李 治, 王树杰, 田 野, 赵 伟. 神经电生理监测硬脊膜切开减压治疗急性脊髓损伤过程中脊髓的功能变化[J]. 中国组织工程研究, 2023, 27(8): 1232-1236. |
| [13] | 徐星星, 文超举, 孟茂花, 王勤英, 陈镜桥, 董 强. 口腔种植中的碳纳米材料[J]. 中国组织工程研究, 2023, 27(7): 1062-1070. |
| [14] | 李 诚, 郑国爽, 蒯贤东, 于炜婷. 海藻酸盐支架修复关节软骨[J]. 中国组织工程研究, 2023, 27(7): 1080-1088. |
| [15] | 陈世崧, 刘晓红, 徐志云. 人工生物瓣膜的研究现状及展望[J]. 中国组织工程研究, 2023, 27(7): 1096-1102. |
牙髓干细胞是牙源性间充质干细胞,其来源广泛取材方便,具有成骨分化等诸多良好的生物学特性,是骨组织工程领域中极具潜质的种子细胞[2]。实际上,发炎牙髓组织来源的牙髓干细胞在临床来源和研究中并不少见,而炎症环境对牙髓干细胞的增殖、分化以及免疫调节特性都有着不可忽视的影响[3],尤其对成骨分化特性的影响于骨组织工程而言意义重大;与此同时,牙髓干细胞对炎症又有着复杂的免疫反应和调节作用,对某些炎症性疾病也有潜在的疗效[4]。由此可见,明确炎症与牙髓干细胞成骨分化的双向作用对于骨再生而言显得尤为重要。近年来相关的研究进展迅速,却鲜有文章对上述的双向关系做总结分析,文章内容最为贴切的一篇综述也仅仅针对于牙源性干细胞和炎症的相互作用,并没有将牙髓干细胞作为主要论述对象,也没有将成骨分化和骨组织工程相关应用作为侧重点[5]。当然也有文章直接论述与牙髓干细胞相关的骨组织工程的研究进展,而炎症因素却提及甚少[6]。
文章以炎症因素与牙髓干细胞成骨分化双向作用作为论述的主线,在篇头概述牙髓干细胞特性的同时,将来源广泛、更具应用潜能的炎症牙髓干细胞也作了对比介绍,希望通过综述近十年的研究成果,为骨组织工程的推进和牙髓干细胞在临床上的应用提供进一步的研究思路。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1.1 检索人及检索时间 第一作者在2022年8月进行检索。
1.1.2 检索文献时限 检索文献时限设置为2012-2022年。
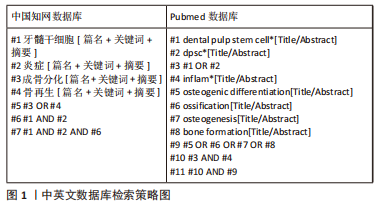
1.1.3 检索数据库 PubMed、中国知网和万方数据库。
1.1.4 检索词 中文检索词为“牙髓干细胞、炎症、成骨分化、骨再生”;英文检索词为“dental pulp stem cell*,dpsc*,inflam*,osteogenic differentiation,ossification,osteogenesis,bone formation”。
1.1.5 检索文献类型 研究原著、综述、述评、病例报告和荟萃分析。
1.1.6 手工检索情况 无。
1.1.7 检索策略 具体检索策略见图1。
1.2 入组标准
1.2.1 纳入标准 ①有关牙髓干细胞特性和临床应用的文献;②有关牙髓干细胞成骨分化的文献;③有关炎症环境、炎症性疾病的文献;④有关炎症与牙髓干细胞关系的文献;⑤同一领域中论点、证据可靠的文献。
1.2.2 排除标准 ①与文章相关性较低或无关的文献;②内容较陈旧的文献;③重复性研究;④资料无法提取的文献。
1.3 文献质量评估与数据的提取 初步检索出文献919篇,阅读摘要和全文后,剔除不符合纳入标准的文献,排除与研究目的相关性差及内容重复、资料陈旧的文献,最终纳入70篇英文文献和6篇中文文献,共计76篇进行综述,见图2。
3.2 作者综述区别于他人他篇的特点 目前,牙髓干细胞相关的综述日渐增多,多数都是围绕牙髓干细胞在某一方向的应用基础研究,比如有文章论述与牙髓干细胞相关的骨组织工程的研究进展,然而炎症因素在其中却未被深入剖析。与此文章较为类似的一篇综述也仅针对于炎症和牙源性干细胞的相互作用,并没有将牙髓干细胞作为论述的主体,也没有将成骨分化和骨组织工程作为侧重点。由此可见,目前还没有一篇文章较有针对性地阐述炎症因素与牙髓干细胞在诱导骨再生过程中的相互关系。因此文章以炎症因素与牙髓干细胞成骨分化间双向关系作为论述的主线,考虑到炎症因素对牙髓干细胞来源的影响,在篇头还将正常牙髓干细胞与临床上更常见且易获取的炎症牙髓干细胞作了对比介绍,以期望拓宽日后的研究方向。
3.3 综述的局限性 由于该综述有较强的侧重点,因此在筛选文献时排除了许多如炎症因素影响牙髓干细胞其他特性的研究、只研究牙髓干细胞成骨分化却未谈及炎症的文献、牙髓干细胞对非炎症性骨缺损疾病的治疗进展等,这可能会让读者产生一些片面的理解。由于目前挖掘机制的研究较少,该综述在罗列双向关系的机制时仅仅基于10年内现有的结论,并不代表机制仅限于此。还有一些观点比如关于炎症牙髓干细胞生物特性的研究存在矛盾,而最终的结论只能由未来更严谨的研究去证实,却不能在此综述中主观臆断。
3.4 综述的重要意义 文章重点对比介绍了炎症牙髓干细胞,不仅列举了现有的研究结论争议,还强调了它的潜在应用价值,以期望引起更多学者的关注从而加强研究,让临床上的炎症牙髓组织变废为宝。文章还总结了目前已知的可以促进牙髓干细胞炎症条件下成骨分化的外源因子,这些研究结果或许将成为骨组织工程中良好的调节剂。与此同时,文章巧妙地提出将牙髓干细胞抗炎的免疫特性和成骨的分化潜力联合运用,筛选了一些具有骨缺损特征的炎症性疾病以突出牙髓干细胞针对性的疗效,尤其是牙周炎和关节炎,可能是目前牙髓干细胞抗炎促成骨潜能较为合适的应用方向。
3.5 课题专家组对未来的建议 牙髓干细胞是骨组织工程研究领域中极具潜力的种子细胞,而炎症作为影响牙髓干细胞成骨分化的重要因素之一,二者的双向关系值得被深入研究,尤其是目前仍处于探索阶段的内在机制如信号通路及细胞因子等。当然,对于文中所提及的一些存在争议的结论,也需要日后更多的研究做支撑完善。目前,有关牙髓干细胞炎症条件下成骨分化的促进因素广为研究,但是缺乏横向的对比和对多种促进因素的联合效应的探索,或许这也是可期的一类研究方向。最后,为了让牙髓干细胞的抗炎和骨再生潜力恰到好处地应用于临床,针对其治疗炎症性骨缺损疾病的疗效还需更多的动物和临床试验来验证。也许牙髓干细胞抗炎促成骨的应用远不止文中提及的这些疾病,期待着未来该领域的研究进展也展望着牙髓干细胞能早日应用于临床实践中。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
 #br#
#br#
文题释义:
牙髓干细胞:是从牙髓组织分离的,来自神经嵴的外胚间充质干细胞,具有自我更新、免疫调节、多向分化潜能和易获得等特点,一定条件下可分化为神经样细胞、成牙本质细胞和成骨细胞等,目前已被报道在神经、口腔和骨骼等部位的再生医学价值,有良好的临床应用前景。炎症:具有血管系统的活体组织对损伤因子所发生的防御反应,通常引起局部组织呈现发红、肿胀、发热、疼痛和功能丧失的典型炎症反应体征。炎症作为一种病理过程,其特征在于损伤与防御的两面性。对牙髓干细胞而言,炎症微环境与其生物学特性关系密切,其中涉及复杂的炎症递质与信号通路的参与。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
文章紧抓“炎症”“牙髓干细胞”和“骨再生”这3个要素,以骨再生过程中,炎症因素和牙髓干细胞的双向作用为主线展开分析。不同于现有的综述只阐述炎症与干细胞的相互作用或单纯分析牙髓干细胞的成骨分化潜能,文章关注牙髓干细胞免疫调节和成骨分化两个潜能,对极具应用前景的炎症牙髓干细胞也作了相关介绍,侧重于探讨炎症因素对牙髓干细胞成骨分化的双重影响以及牙髓干细胞联合抗炎和成骨两项特性对于炎症性骨缺损疾病的治疗作用,于骨组织工程和日后的临床研究都具有一定导向意义。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||