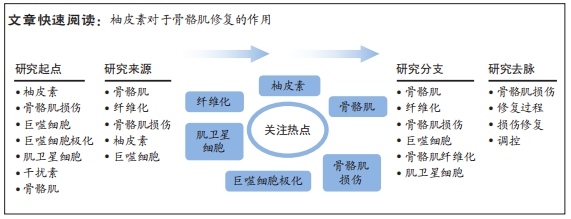
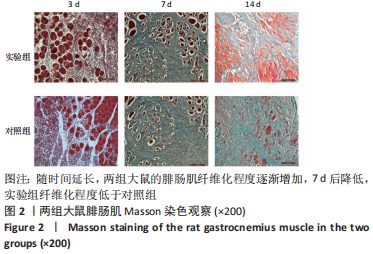
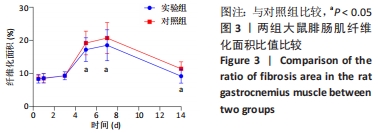
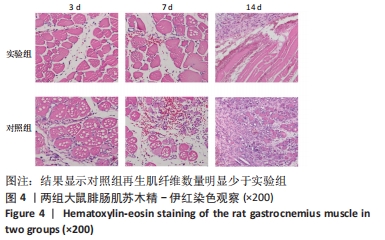
[1] DEGUCHI H, MORLA S, GRIFFIN JH. Novel blood coagulation molecules: Skeletal muscle myosin and cardiac myosin. J Thromb Haemost. 2021;19(1):7-19.
[2] SAHU JK, KUMAR A, PRAKASH K. Randomized controlled trial of electro-acupuncture for autism spectrum disorder. Altern Med Rev. 2010;15(4):302.
[3] LI H, CHEN J, CHEN S, et al. Antifibrotic effects of Smad4 small interfering RNAs in injured skeletal muscle after acute contusion. Int J Sports Med. 2011;32(10):735-742.
[4] BO LI Z, ZHANG J, WAGNER KR. Inhibition of myostatin reverses muscle fibrosis through apoptosis. J Cell Sci. 2012;125(17):3957-3965.
[5] NOGUEIRA JE, AMORIM MR, PINTO AP, et al. Molecular hydrogen downregulates acute exhaustive exercise-induced skeletal muscle damage. Can J Physiol Pharmacol. 2021;99(8):812-820.
[6] YE T, ZHONG L, YE X, et al. miR-221-3p and miR-222-3p regulate the SOCS3/STAT3 signaling pathway to downregulate the expression of NIS and reduce radiosensitivity in thyroid cancer. Exp Ther Med. 2021;21(6):652.
[7] TUSAVITZ S, KEOONELA S, KALKSTEIN M, et al. Macrophage-derived Wnt signaling increases endothelial permeability during skeletal muscle injury. Inflamm Res. 2020;69(12):1235-1244.
[8] SORENSEN JR, KALUHIOKALANI JP, HAFEN PS, et al. An altered response in macrophage phenotype following damage in aged human skeletal muscle: implications for skeletal muscle repair. FASEB J. 2019;33(9):10353-10368.
[9] SUN KT, CHEUNG KK, AU SWN, et al.Overexpression of Mechano-Growth Factor Modulates Inflammatory Cytokine Expression and Macrophage Resolution in Skeletal Muscle Injury. Front Physiol. 2018;9:999.
[10] LE MOAL E, JUBAN G, BERNARD AS, et al. Macrophage-derived superoxide production and antioxidant response following skeletal muscle injury. Free Radic Biol Med. 2018;120:33-40.
[11] WANG X, ZHAO W, RANSOHOFF RM, et al. Infiltrating macrophages are broadly activated at the early stage to support acute skeletal muscle injury repair. J Neuroimmunol. 2018;317:55-66.
[12] RYBALKO V, HSIEH PL, MERSCHAM-BANDA M, et al. The Development of Macrophage-Mediated Cell Therapy to Improve Skeletal Muscle Function after Injury. PLoS One. 2015;10(12):e0145550.
[13] CÔTÉ CH, BOUCHARD P, VAN ROOIJEN N, et al. Monocyte depletion increases local proliferation of macrophage subsets after skeletal muscle injury. BMC Musculoskelet Disord. 2013;14:359.
[14] KRAUSE MP, AL-SAJEE D, D’SOUZA DM, et al. Impaired macrophage and satellite cell infiltration occurs in a muscle-specific fashion following injury in diabetic skeletal muscle. PLoS One. 2013;8(8):e70971.
[15] 程建红,洪莉,洪莎莎,等.巨噬细胞在骨骼肌损伤再生中的研究进展[J].医学综述,2021,27(16):3125-3129.
[16] 贺舟,常青,唐成林,等.大鼠骨骼肌急性损伤后早期运动训练和按摩对肌卫星细胞增殖相关因子的影响[J].中国康复理论与实践,2020,26(1):49-54.
[17] 胡久婷,朱道立.慢肌卫星细胞移植修复骨骼肌的钝挫损伤[J].中国组织工程研究与临床康复,2010,14(23):4271-4274.
[18] 谢春燕,谢刚,季语竹.柚皮素通过miR-22抑制NLRP3炎症小体并减轻溃疡性结肠炎大鼠模型肠屏障损伤[J].中国病理生理杂志,2021,37(9):1573-1581.
[19] 郭诗哲,孙亚英,刘少华, 等.柚皮素抑制小鼠骨骼肌急性钝挫伤后纤维化[J].中国运动医学杂志,2017,36(3):201-206.
[20] CHAZAUD B. Inflammation and Skeletal Muscle Regeneration: Leave It to the Macrophages!. Trends Immunol. 2020;41(6):481-492.
[21] TIAN ZL, JIANG SK, ZHANG M, et al. Detection of satellite cells during skeletal muscle wound healing in rats: time-dependent expressions of Pax7 and MyoD in relation to wound age. Int J Legal Med. 2016;130(1):163-172.
[22] SATO T, HIGASHIOKA K, SAKURAI H, et al. Core Transcription Factors Promote Induction of PAX3-Positive Skeletal Muscle Stem Cells. Stem Cell Reports. 2019; 13(2):352-365.
[23] ALMEIDA CF, FERNANDES SA, RIBEIRO JUNIOR AF, et al. Muscle Satellite Cells: Exploring the Basic Biology to Rule Them. Stem Cells Int. 2016;16(3):1078686.
[24] FUJIMAKI S, SEKO D, KITAJIMA Y, et al. Notch1 and Notch2 Coordinately Regulate Stem Cell Function in the Quiescent and Activated States of Muscle Satellite Cells.Stem Cells. 2018;36(2):278-285.
[25] HUŠÁKOVÁ M, BAY-JENSEN AC, FOREJTOVÁ Š, et al. Metabolites of type I, II, III, and IV collagen may serve as markers of disease activity in axial spondyloarthritis. Sci Rep. 2019;9(1):11218.
[26] RHO SW, CHOI GS, KO EJ, et al. Molecular changes in remote tissues induced by electro-acupuncture stimulation at acupoint ST36. Mol Cells. 2008,25(2):178-183.
[27] ARANGO DUQUE G, DESCOTEAUX A. Macrophage cytokines: involvement in immunity and infectious diseases. Front Immunol. 2014;7(5):491.
[28] DU H, SHIH CH, WOSCZYNA MN, et al. Macrophage-released ADAMTS1 promotes muscle stem cell activation. Nat Commun. 2017;8(1):669.
[29] SU Y, YU Y, LIU C, et al. Fate decision of satellite cell differentiation and self-renewal by miR-31-IL34 axis. Cell Death Differ. 2020;27(3):949-965.
[30] MCLENNAN IS. Degenerating and regenerating skeletal muscles contain several subpopulations of macrophages with distinct spatial and temporal distributions. J Anat. 1996;188( Pt 1):17-28.
[31] WANG X, SATHE AA, SMITH GR, et al. Heterogeneous origins and functions of mouse skeletal muscle-resident macrophages. Proc Natl Acad Sci U S A. 2020; 117(34):20729-20740.
[32] CARLETON MM, SEFTON MV. Injectable and degradable methacrylic acid hydrogel alters macrophage response in skeletal muscle. Biomaterials. 2020;223:119477.
[33] SONG S, AN J, LI Y, et al. Electroacupuncture at ST-36 ameliorates DSS-induced acute colitis via regulating macrophage polarization induced by suppressing NLRP3/IL-1beta and promoting Nrf2/HO-1. Mol Immunol. 2019;106:143-152.
[34] MCAVOY M, TSOSIE JK, VYAS KN, et al. Flexible Multielectrode Array for Skeletal Muscle Conditioning, Acetylcholine Receptor Stabilization and Epimysial Recording After Critical Peripheral Nerve Injury. Theranostics. 2019;9(23):7099-7107.
[35] CUI CY, DRISCOLL RK, PIAO Y, et al. Skewed macrophage polarization in aging skeletal muscle. Aging Cell. 2019;18(6):e13032.
[36] LI T, GARCIA-GOMEZ A, MORANTE-PALACIOS O, et al. SIRT1/2 orchestrate acquisition of DNA methylation and loss of histone H3 activating marks to prevent premature activation of inflammatory genes in macrophages. Nucleic Acids Res. 2020;48(2):665-681.
[37] HE L, ZHAO X, HE L. LINC01140 Alleviates the Oxidized Low-Density Lipoprotein-Induced Inflammatory Response in Macrophages via Suppressing miR-23b. Inflammation. 2020;43(1):66-73.
[38] MCCLAIN CALDWELL I, HOGDEN C, NEMETH K, et al. Bone Marrow-Derived Mesenchymal Stromal Cells (MSCs) Modulate the Inflammatory Character of Alveolar Macrophages from Sarcoidosis Patients. J Clin Med. 2020;9(1):278.
|