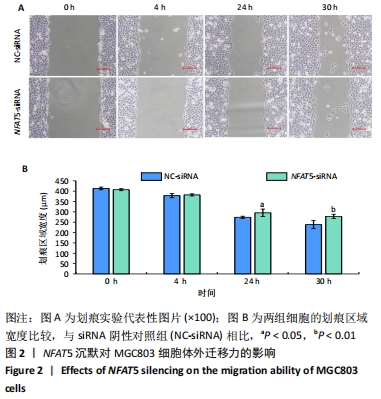
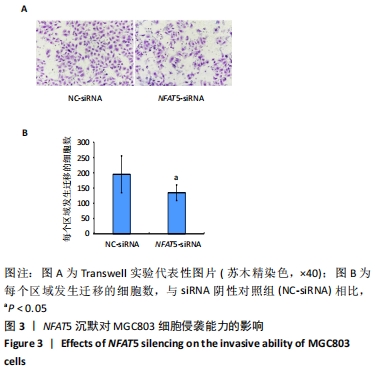
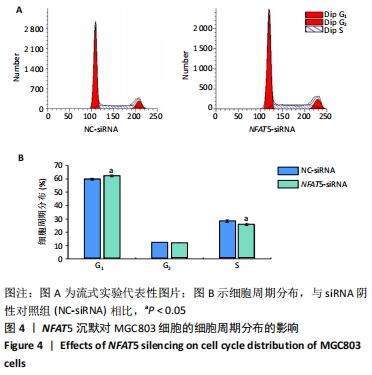
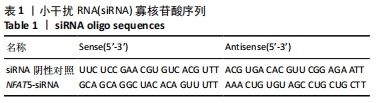
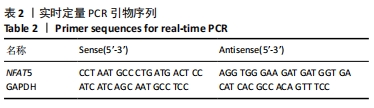
[1] SIEGEL RL, MILLER KD, JEMAL A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7-34.
[2] 侯建忠, 董珊珊, 袁明, 等. 2013年中国肿瘤登记地区胃癌死亡及寿命损失模式分析[J]. 中华肿瘤防治杂志,2019,26(14):986-990.
[3] WANG SM, ZHENG RS, ZHANG SW. Epidemiological characteristics of gastric cancer in China, 2015. Zhong Hua Liu Xing Bing Xue Za Zhi. 2019; 40(12):1517-1521.
[4] 牛闻宇, 康金科, 支永发,等. 胃部分切除手术治疗早期胃癌的临床疗效分析[J]. 中华肿瘤防治杂志,2019,26(S1):72-73.
[5] MAEOKA Y, WU Y, OKAMOTO T, et al. NFAT5 up-regulates expression of the kidney-specific ubiquitin ligase gene Rnf183 under hypertonic conditions in inner-medullary collecting duct cells. J Biol Chem. 2019; 294(1):101-115.
[6] YANG XL, WANG X, PENG BW. NFAT5 Has a Job in the Brain. Dev Neurosci. 2018;40(4):289-300.
[7] KEMPSON SA, ZHOU Y, DANBOLT NC. The betaine/GABA transporter and betaine: roles in brain, kidney, and liver. Front Physiol. 2014;5:159.
[8] HERBELET S, MERCKX C, DE PAEPE B. The PKA-p38MAPK-NFAT5-Organic Osmolytes Pathway in Duchenne Muscular Dystrophy: From Essential Player in Osmotic Homeostasis, Inflammation and Skeletal Muscle Regeneration to Therapeutic Target. Biomedicines. 2021;9(4):350.
[9] AL-ATTAR R, ZHANG Y, STOREY KB. Osmolyte regulation by TonEBP/NFAT5 during anoxia-recovery and dehydration-rehydration stresses in the freeze-tolerant wood frog. Peer J. 2017;5:e2797.
[10] SHOU J, JING J, XIE J, et al. Nuclear factor of activated T cells in cancer development and treatment. Cancer Lett. 2015;361(2):174-184.
[11] WANG S, ZHANG Y, LIN Y. Effect of IL-8 on integrin αvβ3 expression and cell motility via PI3K, Akt, and NF-κB-dependent pathway in human breast cells. Clin Oncol. 2012;30:158.
[12] LI XL, LIU L, LI DD, et al. Integrin β4 promotes cell invasion and epithelial-mesenchymal transition through the modulation of Slug expression in hepatocellular carcinoma. Sci Rep. 2017;7:40464.
[13] LEVY C, KHALED M, ILIOPOULOS D, et al. Intronic miR-211 assumes the tumor suppressive function of its host gene in melanoma. Mol Cell. 2010;40:841-849.
[14] MCCARTHY-KEITH DM, MALIK M, BRITTEN J, et al. Gonadotropin-releasing hormone agonist increases expression of osmotic response genes in leiomyoma cells. Fertil Steril. 2011;95(7):2383-2387.
[15] GERMANN S, GRATADOU L, ZONTA E, et al. Dual role of the ddx5/ddx17 RNA helicases in the control of the pro-migratory NFAT5 transcription factor. Oncogene. 2012;31(42):4536-4549.
[16] ALVAREZ-DÍAZ S, VALLE N, FERRER-MAYORGA G, et al. MicroRNA-22 is induced by vitamin D and contributes to its antiproliferative, antimigratory and gene regulatory effects in colon cancer cells. Hum Mol Genet. 2012;21(10):2157-2165.
[17] REMO A, SIMEONE I, PANCIONE M, et al. Systems biology analysis reveals NFAT5 as a novel biomarker and master regulator of inflammatory breast cancer. J Transl Med. 2015;13:138.
[18] CHO HJ, YUN HJ, YANG HC, et al. Prognostic significance of nuclear factor of activated T-cells 5 expression in non-small cell lung cancer patients who underwent surgical resection. J Surg Res. 2018;226:40-47.
[19] FU SW, ZHANG Y, LI S, et al. LncRNA TTN-AS1 promotes the progression of oral squamous cell carcinoma via miR-411-3p/NFAT5 axis. Cancer Cell Int. 2020;20:415.
[20] GUO K, JIN F. NFAT5 promotes proliferation and migration of lung adenocarcinoma cells in part through regulating AQP5 expression. Biochem Biophys Res Commun. 2015;465(3):644-649.
[21] 郭隽馥, 于爽, 王悦, 等. 活化T细胞核因子5对人胃癌MGC803细胞增殖及凋亡能力的影响[J]. 中国肿瘤生物治疗杂志,2018, 25(11):1119-1124.
[22] 訚亚涛, 涂建成, 喻明霞. 活化T细胞核因子的临床研究进展[J]. 医学研究生学报,2011,24(1):109-112.
[23] VIHMA H, LUHAKOODER M, PRUUNSILD P, et al. Regulation of different human NFAT isoforms by neuronal activity. J Neurochem. 2016;137(3): 394-408.
[24] DURAN J, OYARCE C, PAVEZ M, et al. GSK-3beta/NFAT signaling is involved in testosterone-induced cardiac myocyte hypertrophy. PLoS One. 2016;11(12):e168255.
[25] JIANG Y, HE R, JIANG Y, et al. Transcription factor NFAT5 contributes to the glycolytic phenotype rewiring and pancreatic cancer progression via transcription of PGK1. Cell Death Dis. 2019;10(12):948.
[26] MIJATOVIC T, MATHIEU V, GAUSSIN JF, et al. Cardenolide-induced lysosomal membrane permeabilization demonstrates therapeutic benefits in experimental human non-small cell lung cancers. Neoplasia. 2006;8(5):402-412.
[27] BROWN TC, NICOLSON NG, MAN J, et al. Recurrent Amplification of the Osmotic Stress Transcription Factor NFAT5 in Adrenocortical Carcinoma. J Endocr Soc. 2020;4(7):bvaa060.
[28] KIM DH, KIM KS, RAMAKRISHNA S. NFAT5 promotes in vivo development of murine melanoma metastasis. Biochem Biophys Res Commun. 2018;505(3):748-754.
[29] MENG X, LI Z, ZHOU S, et al. miR-194 suppresses high glucose-induced non-small cell lung cancer cell progression by targeting NFAT5. Thorac Cancer. 2019;10(5):1051-1059.
[30] 李剑锋, 倪莎, 刘笑默. 干扰NFAT5的表达对宫颈癌增殖和转移能力的影响及机制研究[J].标记免疫分析与临床,2021,28(1):117-121.
[31] 于珊, 郑熙. 四君子汤对胃癌MGC803细胞NFAT5及凋亡因子表达的影响[J]. 中国中医药现代远程教育,2021,19(11):165-168.
[32] 王建, 柴嘉穗, 张雅敏. 活化T细胞核因子与肿瘤关系的研究进展[J]. 实用器官移植电子杂志,2017,5(4):315-320.
[33] LEE S, KONG JS, YOU S, et al. Transcription Factor NFAT5 Promotes Migration and Invasion of Rheumatoid Synoviocytes via Coagulation Factor III and CCL2. J Immunol. 2018;201(2):359-370.
[34] DOU Y, TIAN W, WANG H, et al. Circ_0001944 Contributes to Glycolysis and Tumor Growth by Upregulating NFAT5 Through Acting as a Decoy for miR-142-5p in Non-Small Cell Lung Cancer. Cancer Manag Res. 2021;13: 3775-3787.
[35] TIAN L, CAI D, ZHUANG D, et al. miR-96-5p Regulates Proliferation, Migration, and Apoptosis of Vascular Smooth Muscle Cell Induced by Angiotensin II via Targeting NFAT5. J Vasc Res. 2020;57(2):86-96.
[36] YOSHIMOTO S, MORITA H, MATSUDA M, et al. NFAT5 promotes oral squamous cell carcinoma progression in a hyperosmotic environment. Lab Invest. 2021;101(1):38-50.
[37] YANG M, KE H, ZHOU W. LncRNA RMRP Promotes Cell Proliferation and Invasion Through miR-613/NFAT5 Axis in Non-Small Cell Lung Cancer. Onco Targets Ther. 2020;13:8941-8950.
[38] KÜPER C, BECK FX, NEUHOFER W. NFAT5-mediated expression of S100A4 contributes to proliferation and migration of renal carcinoma cells. Front Physiol. 2014;5:293.
[39] MISHRA SK, SIDDIQUE HR, SALEEM M. S100A4 calcium-binding protein is key player in tumor progression and metastasis: preclinical and clinical evidence. Cancer Metastasis Rev. 2012;31(1-2):163-172.
[40] XU J, GROSS N, ZANG Y, et al. Overexpression of S100A4 Predicts Migration, Invasion, and Poor Prognosis of Hypopharyngeal Squamous Cell Carcinoma. Mol Diagn Ther. 2019;23(3):407-417.
[41] LI F, SHI J, XU Z, et al. S100A4-MYH9 Axis Promote Migration and Invasion of Gastric Cancer Cells by Inducing TGF-β-Mediated Epithelial-Mesenchymal Transition. J Cancer. 2018;9(21):3839-3849.
[42] CHEN M, SASTRY SK, O’CONNOR KL. Src kinase pathway is involved in NFAT5-mediated S100A4 induction by hyperosmotic stress in colon cancer cells. Am J Physiol Cell Physiol. 2011;300(5):C1155-1163.
[43] GUO J, BIAN Y, WANG Y, et al. S100A4 influences cancer stem cell-like properties of MGC803 gastric cancer cells by regulating GDF15 expression. Int J Oncol. 2016;49(2):559-568.
[44] GUO J, BIAN Y, WANG Y, et al. FAM107B is regulated by S100A4 and mediates the effect of S100A4 on the proliferation and migration of MGC803 gastric cancer cells. Cell Biol Int. 2017;41(10):1103-1109.
[45] 高立红, 张天彪, 赵滢. siRNA干扰S100A4蛋白引起钙离子信号改变后对人胃癌细胞的影响[J]. 现代肿瘤医学,2019,27(10):1675-1680.
|