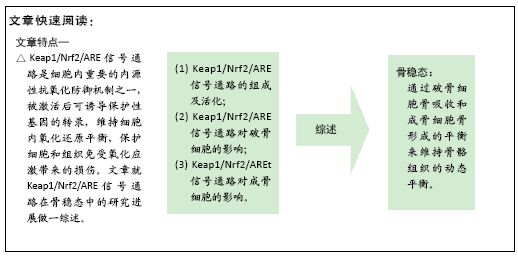
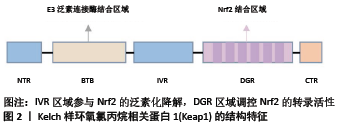
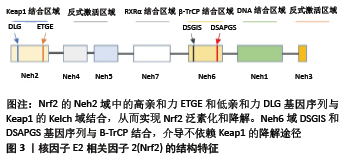
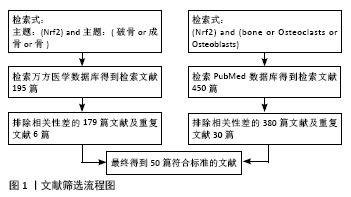
[1] Bone health and nutrition: Introduction to the special issue. Mol Cell Endocrinol. 2015;410:1-2.
[2] KENSLER TW, WAKABAYASHI N, BISWAL S. Cell Survival Responses to Environmental Stresses Via the Keap1-Nrf2-ARE Pathway. Annu Rev Pharmacol Toxicol. 2007;47:89-116.
[3] MOTOHASHI H, YAMAMOTO M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol Med. 2004;10(11): 549-557.
[4] ITOH K, WAKABAYASHI N, KATOH Y, et al. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999;13(1):76-86.
[5] BUGNO M, DANIEL M, CHEPELEV NL, et al. Changing gears in Nrf1 research, from mechanisms of regulation to its role in disease and prevention. Biochim Biophys Acta. 2015;1849(10):1260-1276.
[6] KOPACZ A, KLOSKA D, FORMAN HJ, et al. Beyond repression of Nrf2: An update on Keap1. Free Radic Biol Med. 2020;157:63-74.
[7] NAM LB, KEUM YS. Binding partners of NRF2: Functions and regulatory mechanisms. Arch Biochem Biophys. 2019;678:108184.
[8] MOI P, CHAN K, ASUNIS I, et al. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Proc Natl Acad Sci U S A. 1994;91(21):9926-9930.
[9] LAU A, TIAN W, WHITMAN SA, et al. The Predicted Molecular Weight of Nrf2: It Is What It Is Not. Antioxid Redox Signal. 2013;18(1):91-93.
[10] KATSUOKA F, YAMAMOTO M. Small Maf proteins (MafF, MafG, MafK): History, structure and function. Gene. 2016;586(2):197-205.
[11] HAYES JD, MCMAHON M. NRF2 and KEAP1 mutations: permanent activation of an adaptive response in cancer. Trends Biochem Sci. 2009;34(4):176-188.
[12] KIM JH, YU S, CHEN JD, et al. The nuclear cofactor RAC3/AIB1/SRC-3 enhances Nrf2 signaling by interacting with transactivation domains. Oncogene. 2013;32(4):514-527.
[13] ZENKOV NK, KOZHIN PM, CHECHUSHKOV AV, et al. Mazes of Nrf2 Regulation. Biochemistry (Mosc). 2017;82(5):556-564.
[14] RADA P, ROJO AI, CHOWDHRY S, et al. SCF/{beta}-TrCP promotes glycogen synthase kinase 3-dependent degradation of the Nrf2 transcription factor in a Keap1-independent manner. Mol Cell Biol. 2011;31(6):1121-1133.
[15] CHOWDHRY S, ZHANG Y, MCMAHON M, et al. Nrf2 is controlled by two distinct β-TrCP recognition motifs in its Neh6 domain, one of which can be modulated by GSK-3 activity. Oncogene. 2013;32(32):3765-3781.
[16] KUMAR H, KIM IS, MORE SV, et al. Natural product-derived pharmacological modulators of Nrf2/ARE pathway for chronic diseases. Nat Prod Rep. 2014;31(1):109-139.
[17] SCHMIDLIN CJ, DODSON MB, MADHAVAN L, et al. Redox regulation by NRF2 in aging and disease. Free Radic Biol Med. 2019;134:702-707.
[18] DINKOVA-KOSTOVA AT, HOLTZCLAW WD, COLE RN, et al. Direct evidence that sulfhydryl groups of Keap1 are the sensors regulating induction of phase 2 enzymes that protect against carcinogens and oxidants. Proc Natl Acad Sci U S A. 2002;99(18):11908-11913.
[19] NEZU M, SUZUKI N, YAMAMOTO M. Targeting the KEAP1-NRF2 System to Prevent Kidney Disease Progression. Am J Nephrol. 2017;45(6): 473-483.
[20] KEUM YS, CHOI BY. Molecular and chemical regulation of the Keap1-Nrf2 signaling pathway. Molecules. 2014;19(7):10074-10089.
[21] KIMURA M, YAMAMOTO T, ZHANG J, et al. Molecular basis distinguishing the DNA binding profile of Nrf2-Maf heterodimer from that of Maf homodimer. J Biol Chem. 2015;290(17):10644.
[22] GAN L, JOHNSON JA. Oxidative damage and the Nrf2-ARE pathway in neurodegenerative diseases. Biochim Biophys Acta. 2014;1842(8): 1208-1218.
[23] KAGEYAMA S, SAITO T, OBATA M, et al. Negative Regulation of the Keap1-Nrf2 Pathway by a p62/Sqstm1 Splicing Variant. Mol Cell Biol. 2018;38(7):e00642-e00617.
[24] ANDERICA C, HERNANDEZ J, VAZQUEZ I, et al. The MLN4924 inhibitor exerts a neuroprotective effect against oxidative stress injury via Nrf2 protein accumulation. Redox Biol. 2016;8:341-347.
[25] WANG R, YU Z, SUNCHU B, et al. Rapamycin inhibits the secretory phenotype of senescent cells by a Nrf2-independent mechanism. Aging Cell. 2017;16(3):564-574.
[26] TIAN D, SHI Y, CHEN D, et al. The Wnt inhibitor LGK-974 enhances radiosensitivity of HepG2 cells by modulating Nrf2 signaling. Int J Oncol. 2017;51(2):545-554.
[27] MOBASHER A, GONZALEZ A, SANTAMARIA B, et al. Protein tyrosine phosphatase 1B modulates GSK3β/Nrf2 and IGFIR signaling pathways in acetaminophen-induced hepatotoxicity. Cell Death Dis. 2013;4(5): e626.
[28] DODSON M, DE LA VEGA MR, CHOLANIANS AB, et al. Modulating NRF2 in Disease: Timing Is Everything. Annu Rev Pharmacol Toxicol. 2019;59:555-575.
[29] LERNER UH. Osteoclasts in Health and Disease. Pediatr Endocrinol Rev. 2019;17(2):84-99.
[30] AMIN N, BOCCARDI V, TAGHIZADEH M, et al. Probiotics and bone disorders: the role of RANKL/RANK/OPG pathway. Aging Clin Exp Res. 2020;32(3):363-371.
[31] 谢冰洁,冯捷,韩向龙.破骨细胞生物学特征的研究与进展[J].中国组织工程研究,2017,21(11):1770-1775.
[32] SUN X, ZHANG B, PAN X, et al. Octyl itaconate inhibits osteoclastogenesis by suppressing Hrd1 and activating Nrf2 signaling. Faseb J. 2019;33: 12929-12940.
[33] KIM HJ, PARK C, KIM GY, et al. Sargassum serratifolium attenuates RANKL-induced osteoclast differentiation and oxidative stress through inhibition of NF-κB and activation of the Nrf2/HO-1 signaling pathway. Biosci Trends. 2018;12(3):257-265.
[34] MA Q, LIANG M, TANG X, et al. Vitamin B5 inhibit RANKL induced osteoclastogenesis and ovariectomy induced osteoporosis by scavenging ROS generation. Am J Transl Res. 2019;11:5008-5018.
[35] PARK CK, LEE Y, KIM KH, et al. Nrf2 is a novel regulator of bone acquisition. Bone. 2014;63:36-46.
[36] CHUNG S, KIM S, SON M, et al. Inhibition of p300/CBP-Associated Factor Attenuates Renal Tubulointerstitial Fibrosis through Modulation of NF-kB and Nrf2. Int J Mol Sci. 2019;20(7):1554.
[37] LI Z, CHEN C, ZHU X, et al. Glycyrrhizin Suppresses RANKL-Induced Osteoclastogenesis and Oxidative Stress Through Inhibiting NF-κB and MAPK and Activating AMPK/Nrf2. Calcif Tissue Int. 2018;103(3): 324-337.
[38] KANZAKI H, SHINOHARA F, KAJIYA M, et al. The Keap1/Nrf2 protein axis plays a role in osteoclast differentiation by regulating intracellular reactive oxygen species signaling. J Biol Chem. 2013;288(32): 23009-23020.
[39] HYEON S, LEE H, YANG Y, et al. Nrf2 deficiency induces oxidative stress and promotes RANKL-induced osteoclast differentiation. Free Radic Biol Med. 2013;65:789-799.
[40] LI W, SUN Y. Nrf2 is required for suppressing osteoclast RANKL-induced differentiation in RAW 264.7 cells via inactivating cannabinoid receptor type 2 with AM630. Regen Ther. 2020;14:191-195.
[41] LIU Z, HOU Y, LI L, et al. Nrf2 deficiency aggravates the increase in osteoclastogenesis and bone loss induced by inorganic arsenic. Toxicol Appl Pharmacol. 2019;367:62-70.
[42] MAEDA K, KOBAYASHI Y, KOIDE M, et al. The Regulation of Bone Metabolism and Disorders by Wnt Signaling. Int J Mol Sci. 2019;20(22): 5525.
[43] TRESGUERRES FGF, TORRES J, LÓPEZ-QUILES J, et al. The osteocyte: A multifunctional cell within the bone. Ann Anat. 2020;227:151422.
[44] KOOK SH, KIM KA, JI H, et al. Irradiation inhibits the maturation and mineralization of osteoblasts via the activation of Nrf2/HO-1 pathway. Mol Cell Biochem. 2015;410(1-2):255-266.
[45] HINOI E, FUJIMORI S, WANG L, et al. Nrf2 negatively regulates osteoblast differentiation via interfering with Runx2-dependent transcriptional activation. J Biol Chem. 2006;281(26):18015-18024.
[46] YOSHIDA E, SUZUKI T, MORITA M, et al. Hyperactivation of Nrf2 leads to hypoplasia of bone in vivo. Genes Cells. 2018;23(5):386-392.
[47] SUN YX, LI L, CORRY KA, et al. Deletion of Nrf2 reduces skeletal mechanical properties and decreases load-driven bone formation. Bone. 2015;74:1-9.
[48] KIM JH, SINGHAL V, BISWAL S, et al. Nrf2 is required for normal postnatal bone acquisition in mice. Bone Res. 2014;2:14033.
[49] IBANEZ L, FERRANDIZ ML, BRINES R, et al. Effects of Nrf2 deficiency on bone microarchitecture in an experimental model of osteoporosis. Oxid Med Cell Longev. 2014;2014:726590.
[50] YIN Y, CORRY KA, LOUGHRAN JP, et al. Moderate Nrf2 Activation by Genetic Disruption of Keap1 Has Sex-Specific Effects on Bone Mass in Mice. Sci Rep. 2020;10(1):348.
|