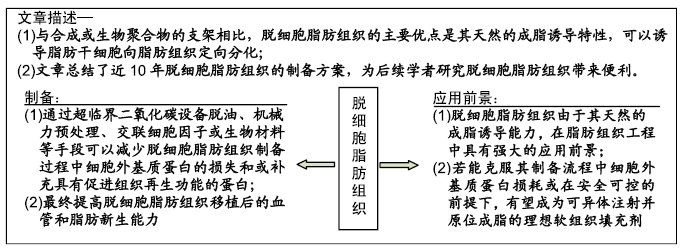
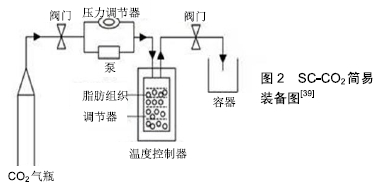
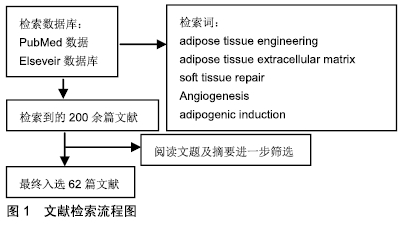
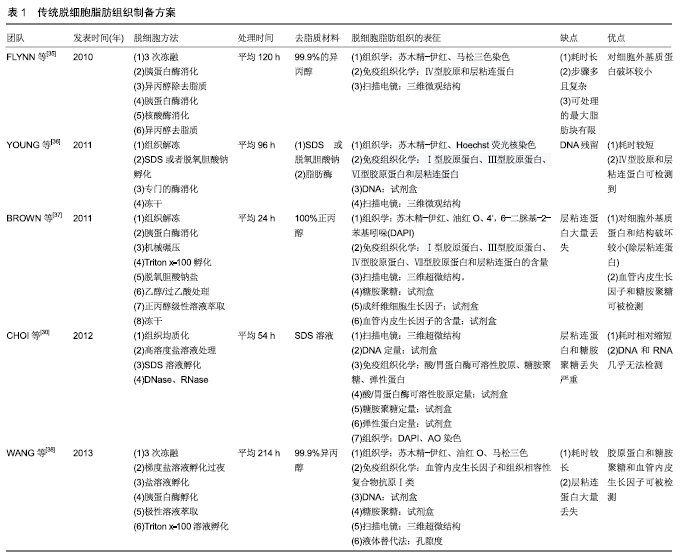
[1] HENKEL J, WOODRUFF MA, EPARI DR,et al. Bone Regeneration Based on Tissue Engineering Conceptions - A 21st Century Perspective. Bone Res.2013;1(3):216-248.
[2] ATALA A, BAUER SB, SOKER S, et al. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet. 2006;367(9518): 1241-1246.
[3] BIRCHALL MA, SEIFALIAN AM.Tissue engineering's green shoots of disruptive innovation. 2014;384(9940):288-290.
[4] FULCO I, MIOT S, HAUG MD, et al. Engineered autologous cartilage tissue for nasal reconstruction after tumour resection: an observational first-in-human trial. Lancet. 2014;384(9940):337-346.
[5] OLAUSSON M, PATIL PB, KUNA VK, et al. Transplantation of an allogeneic vein bioengineered with autologous stem cells: a proof-of-concept study. Lancet. 2012;380(9838):230-237.
[6] RAYA-RIVERA AM, ESQUILIANO D, FIERRO-PASTRANA R, et al. Tissue-engineered autologous vaginal organs in patients: a pilot cohort study. Lancet. 2014;384(9940):329-336.
[7] ELLIOTT RA JR, HOEHN JG. Use of commercial porcine skin for wound dressings. Plast Reconstr Surg. 1973;52(4):401-405.
[8] BADYLAK SF, LANTZ GC, COFFEY A,et al. Small intestinal submucosa as a large diameter vascular graft in the dog. J Surg Res. 1989;47(1):74-80.
[9] CHEN RN, HO HO, TSAI YT, et al. Process development of an acellular dermal matrix (ADM) for biomedical applications. Biomaterials. 2004;25(13):2679-2686.
[10] SCHMIDT CE, BAIER JM. Acellular vascular tissues: natural biomaterials for tissue repair and tissue engineering. Biomaterials. 2000;21(22):2215-2231.
[11] UCHIMURA E, SAWA Y, TAKETANI S, et al. Novel method of preparing acellular cardiovascular grafts by decellularization with poly(ethylene glycol). J Biomed Mater Res A.2003;67:834-837.
[12] DAHL SL, KOH J, PRABHAKAR V, et al.Decellularized native and engineered arterial scaffolds for transplantation. Cell Transplant.2003; 12:659-666.
[13] CONKLIN BS, RICHTER ER, KREUTZIGER KL,et al.Development and evaluation of a novel decellularized vascular xenograft. Med Eng Phys 2002;24:173-183.
[14] BORSCHEL GH, DENNIS RG, KUZON WM. Contractile skeletal muscle tissue-engineered on an acellular scaffold. Plast Reconstr Surg.2004;113:595-602.
[15] LIN P, CHAN WC, BADYLAK SF, et al. Assessing porcine liverderived biomatrix for hepatic tissue engineering. Tissue Eng.2004;10:1046–53.
[16] KROPP BP, EPPLEY BL, PREVEL CD,et al. Experimental assessment of small intestinal submucosa as a bladder wall substitute. Urology 1995;46:396-400.
[17] BADER A, SCHILLING T, TEEBKEN OE, et al. Tissue engineering of heart valves-human endothelial cell seeding of detergent acellularized porcine valves. Eur J Cardiothorac Surg. 1998;14:279-284.
[18] BOOTH C, KOROSSIS SA, WILCOX HE, et al. Tissue engineering of cardiac valve prostheses I:development and histological characterization of an acellular porcine scaffold.J Heart Valve Dis. 2002;11:457-462.
[19] GRAUSS RW, HAZEKAMP MG, OPPENHUIZEN F, et al. Histological evaluation of decellularised porcine aortic valves: matrix changes due to different decellularisation methods. Eur J Cardiothorac Surg. 2005; 27:566-571.
[20] KASIMIR MT, RIEDER E, SEEBACHER G, et al. Comparison of different decellularization procedures of porcine heart valves. Int J Artif Organs. 2003;26:421-427.
[21] KOROSSIS SA, BOOTH C, WILCOX HE, et al. Tissue engineering of cardiac valve prostheses II:biomechanical characterization of decellularized porcine aortic heart valves.J Heart Valve Dis. 2002;11: 463-471.
[22] RIEDER E, KASIMIR MT, SILBERHUMER G, et al. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J Thorac Cardiovasc Surg. 2004;127:399-405.
[23] BOYCE M, RADTKE C, VOGT PM. The volumetric analysis of fat graft survival in breast reconstruction. Plast Reconstr Surg. 2013;132(5): 862e-863e.
[24] CHACON AH. Fillers in dermatology: from past to present. Cutis. 2015; 96(5):E17-19.
[25] CHIANG YZ, PIERONE G, AL-NIAIMI F. Dermal fillers: pathophysiology, prevention and treatment of complications. J Eur Acad Dermatol Venereol.2017;31(3):405-413.
[26] VAN NIEUWENHOVE I, TYTGAT L, RYX M, et al. Soft tissue fillers for adipose tissue regeneration: From hydrogel development toward clinical applications.Acta Biomater.2017;63:37-49.
[27] DZIKI JL, WANG DS, PINEDA C, et al. Solubilized extracellular matrix bioscaffolds derived from diverse source tissues differentially influence macrophage phenotype.J Biomed Mater Res A. 2017;105(1):138-147.
[28] YU Y, ALKHAWAJI A, DING Y, et al. Decellularized scaffolds in regenerative medicine. Oncotarget.2016;7(36):58671-58683.
[29] LIN CY, LIU TY, CHEN MH, et al. An injectable extracellular matrix for the reconstruction of epidural fat and the prevention of epidural fibrosis. Biomed Mater.2016;11(3):035010.
[30] CHOI JS, KIM BS, KIM JY, et al. Decellularized extracellular matrix derived from human adipose tissue as a potential scaffold for allograft tissue engineering. J Biomed Mater Res A.2011;97(3):292-299.
[31] GILBERT TW, SELLARO TL, BADYLAK SF.Decellularization of tissues and organs. Biomaterials.2006;27(19):3675-3683.
[32] SEDDON AM, CURNOW P, BOOTH PJ. Membrane proteins, lipids and detergents: not just a soap opera. Biochim Biophys Acta. 2004; 1666(1-2):105-117.
[33] WANG L, JOHNSON JA, CHANG DW, et al. Decellularized musculofascial extracellular matrix for tissue engineering. Biomaterials. 2013;34(11):2641-2654.
[34] SANO H, ORBAY H, TERASHI H, et al. Acellular adipose matrix as a natural scaffold for tissue engineering. J Plast Reconstr Aesthet Surg. 2014;67(1):99-106.
[35] FLYNN LE. The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials.2010;31(17):4715-4724.
[36] YOUNG DA, IBRAHIM DO, HU D, et al.Injectable hydrogel scaffold from decellularized human lipoaspirate. Acta Biomater. 2011;7(3): 1040-1049.
[37] BROWN BN, FREUND JM, HAN L, et al.Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix. Tissue Eng Part C Methods.2011;17(4):411-421.
[38] WANG L, JOHNSON JA, ZHANG Q, et al. Combining decellularized human adipose tissue extracellular matrix and adipose-derived stem cells for adipose tissue engineering.Acta Biomater. 2013;9(11):8921-8931.
[39] WANG JK, LUO B, GUNETA V, et al. Supercritical carbon dioxide extracted extracellular matrix material from adipose tissue. Mater Sci Eng C Mater Biol Appl.2017;75:349-358.
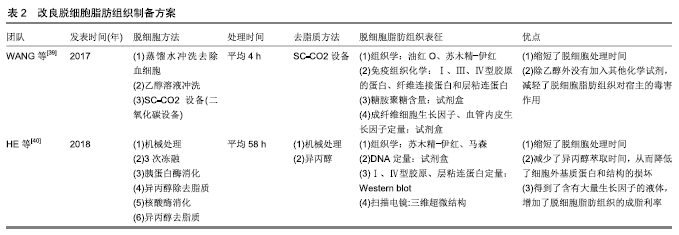
[40] HE Y, LIN M, WANG X, et al.Optimized adipose tissue engineering strategy based on a neo-mechanical processing method. Wound Repair Regen.2018;26(2):163-171.
[41] REN H, SHI X, TAO L, et al. Evaluation of two decellularization methods in the development of a whole-organ decellularized rat liver scaffold. Liver Int.2013;33(3):448-458.
[42] CEBOTARI S, TUDORACHE I, JAEKEL T, et al.Detergent decellularization of heart valves for tissue engineering: toxicological effects of residual detergents on human endothelial cells. Artif Organs. 2010;34(3):206-210.
[43] CERQUEIRA MT, MARQUES AP, REIS RL. Using stem cells in skin regeneration: possibilities and reality. Stem Cells Dev. 2012;21(8): 1201-1214.
[44] POON CJ, PEREIRA E COTTA MV, SINHA S, et al. Preparation of an adipogenic hydrogel from subcutaneous adipose tissue.Acta Biomater. 2013;9(3):5609-5620.
[45] ADAM YOUNG D, BAJAJ V, CHRISTMAN KL. Award winner for outstanding research in the PhD category, 2014 Society for Biomaterials annual meeting and exposition, Denver, Colorado, April 16-19, 2014: Decellularized adipose matrix hydrogels stimulate in vivo neovascularization and adipose formation. J Biomed Mater Res A. 2014;102(6):1641-1651.
[46] HAN TT, TOUTOUNJI S, AMSDEN BG, et al. Adipose-derived stromal cells mediate in vivo adipogenesis, angiogenesis and inflammation in decellularized adipose tissue bioscaffolds.Biomaterials.2015; pii: S0142-9612(15)00735-8.
[47] BAJEK A, GURTOWSKA N, OLKOWSKA J, et al. Adipose-Derived Stem Cells as a Tool in Cell-Based Therapies, Arch Immunol Ther Exp (Warsz).2016;64(6):443-454.
[48] CHOI JS, YANG HJ, KIM BS, et al. Human extracellular matrix (ECM) powders for injectable cell delivery and adipose tissue engineering.J Control Release.2009;139(1):2-7.
[49] TURNER AE, YU C, BIANCO J, et al. The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells. Biomaterials. 2012;33(18):4490-4499.
[50] TING AC, CRAFT RO, PALMER JA, et al.The adipogenic potential of various extracellular matrices under the influence of an angiogenic growth factor combination in a mouse tissue engineering chamber. Acta Biomater.2014;10(5):1907-1918.
[51] LU Q, LI M, ZOU Y, et al. Delivery of basic fibroblast growth factors from heparinized decellularized adipose tissue stimulates potent de novo adipogenesis.J Control Release.2014;174:43-50.
[52] ZHANG S, LU Q, CAO T, et al. Adipose Tissue and Extracellular Matrix Development by Injectable Decellularized Adipose Matrix Loaded with Basic Fibroblast Growth Factor. Plast Reconstr Surg.2016; 137(4): 1171-1180.
[53] ZENG F, HARRIS RC.Epidermal growth factor,from gene organization to bedside.Semin Cell Dev Biol.2017;28:2-11.
[54] BERNARDES VF, GLEBER-NETTO FO, SOUSA SF, et al, EGF insaliva and tumor samples of oral squamous cell carcinoma.Appl Immunohistochem Mol Morphol.2017;19(6):528-533.
[55] ZHOU X, WANG J, HUANG X, et al. Injectable decellularized nucleus pulposus-based cell delivery system for differentiation of adipose-derived stem cells and nucleus pulposus regeneration.Acta Biomater. 2018;81: 115-128.
[56] WU I, NAHAS Z, KIMMERLING KA, et al. An injectable adipose matrix for soft-tissue reconstruction. Plast Reconstr Surg. 2012;129(6):1247-1257.
[57] LI S, POCHE JN, LIU Y, et al. Hybrid Synthetic-Biological Hydrogel System for Adipose Tissue Regeneration. Macromol Biosci. 2018; 18(11):e1800122.
[58] MAUNEY JR, NGUYEN T, GILLEN K, et al.Engineering adipose-like tissue in vitro and in vivo utilizing human bone marrow and adipose-derived mesenchymal stem cells with silk fibroin 3D scaffolds. Biomaterials.2007;28(35):5280-5290.
[59] KANG JH, GIMBLE JM, KAPLAN DL. In vitro 3D model for human vascularized adipose tissue. Tissue Eng Part A.2009;15(8):2227-2236.
[60] KAYABOLEN A, KESKIN D, AYKAN A, et al. Native extracellular matrix/fibroin hydrogels for adipose tissue engineering with enhanced vascularization. Biomed Mater.2017;12(3):035007.
[61] ZHAO Y, FAN J, BAI S. Biocompatibility of injectable hydrogel from decellularized human adipose tissue in vitro and in vivo.J Biomed Mater Res B Appl Biomater. 2019;107(5):1684-1694.
[62] MORI S, KIUCHI S, OUCHI A, et al. Characteristic expression of extracellular matrix in subcutaneous adipose tissue development and adipogenesis; comparison with visceral adipose tissue." Int J Biol Sci. 2014;10(8):825-833.
|