| [1] Suga S, Goto S, Togari A. Demonstration of Direct Neurite–Osteoclastic Cell Communication In Vitro via the Adrenergic Receptor. J Pharmacol Sci. 2010;112:184-191.[2] Hirai T, Tanaka K, Togari A. alpha1-adrenergic receptor signaling in osteoblasts regulates clock genes and bone morphogenetic protein 4 expression through up-regulation of the transcriptional factor nuclear factor IL-3 (Nfil3)/E4 promoter-binding protein 4 (E4BP4). The Journal of biological chemistry. 2014;289:17174-17183.[3] Kim KA, Kook SH, Song JH,et al.A phenolic acid phenethyl urea derivative protects against irradiation-induced osteoblast damage by modulating intracellular redox state. J Cell Biochem. 2014;115:1877-1887.[4] Domazetovic V, Marcucci G, Iantomasi T, et al. Oxidative stress in bone remodeling: role of antioxidants. Clin Cases Miner Bone. 2017;14:209-216.[5] Hampton MB, Orrenius S. Redox regulation of apoptotic cell death. BioFactors (Oxford, England).1998;8(1-2):1-5.[6] Jin YQ, Li JL, Chen JD, et al. Dalbergioidin (DAL) protects MC3T3-E1 osteoblastic cells against H2O2-induced cell damage through activation of the PI3K/AKT/SMAD1 pathway. Naunyn Schmiedebergs Arch Pharmacol. 2017;390(7):711-720.[7] Lee D, Kook SH, Ji H, et al. N-acetyl cysteine inhibits H2O2-mediated reduction in the mineralization of MC3T3-E1 cells by down-regulating Nrf2/HO-1 pathway. BMB Reports. 2015;48:636-641.[8] 李娟,申广浩,程光,等.组织块法培养SD大鼠的成骨细胞及鉴定[J].中国组织工程研究,2012,16(46):8571-8574.[9] 李丽艳,任塘珂.成骨细胞培养在骨组织工程和运动领域的研究进展[J].中国组织工程研究,2012,16(11):2043-2046.[10] 李菊根,陈建庭,金大地.MTT法分析PDGF对鼠成骨细胞生长作用的影响[J].第一军医大学学报,1998,18(3):69-71.[11] 韩立赤,王雪红,董会,等.苯肾上腺素对大鼠成骨细胞放射损伤的保护作用[J].吉林大学学报,2015,41(6):1113-1117+11.[12] 郭宝磊,杨茂伟,梁单,等.过氧化氢通过线粒体途径诱导成骨细胞凋亡[J].中国老年学杂志,2013,33(22):5661-5663.[13] Wanet A, Arnould T, Najimi M, et al. Connecting Mitochondria, Metabolism, and Stem Cell Fate. Stem Cells Dev. 2015;24: 1957-1971.[14] Mountziaris PM, Spicer PP, Kasper FK,et al. Harnessing and modulating inflammation in strategies for bone regeneration. Tissue Eng Part B Rev. 2011;17(6):393-402.[15] Schett G. Effects of inflammatory and anti-inflammatory cytokines on the bone. Eur J Clin Invest.2011;41:1361-1366.[16] Lacey DC,Simmons PJ,Graves SE,et al. Proinflammatory cytokines inhibitosteogenic differentiation from stem cells: implications for bone repair during inflammation.Osteoarthritis Cartilage.2009;17:735-742.[17] Manolagas SC. From estrogen-centric to aging and oxidative stress: a revised perspective of the pathogenesis of osteoporosis. Endocr Rev. 2010;31:266-300.[18] Banfi G, Iorio EL,Corsi MM.Oxidative stress, free radicals and bone remodeling. Clin Chem Lab Med. 2008;46:1550-1555.[19] Mann V, Huber C, Kogianni G, et al. The antioxidant effect of estrogen and Selective Estrogen Receptor Modulators in the inhibition of osteocyte apoptosis in vitro. Bone.2007;40: 674-684.[20] Baek KH, Oh KW, Lee WY,et al. Association of oxidative stress with postmenopausal osteoporosis and the effects of hydrogen peroxide on osteoclast formation in human bone marrow cell cultures. Calcif Tissue Int. 2010;87:226-235.[21] Ichiseki T, Matsumoto T, Nishino M,et al. Oxidative stress and vascular permeability in steroid-induced osteonecrosis model. J Orthop Sci. 2004;9(5):509-515.[22] Komori T. Cell Death in Chondrocytes, Osteoblasts, and Osteocytes. Int J Mol Sci. 2016;17(12). pii: E2045.[23] Hu K, Olsen BR. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J Clin Invest. 2016;126(2):509-526.[24] Lean JM, Jagger CJ, Kirstein B,et al. Hydrogen peroxide is essential for estrogen-deficiency bone loss and osteoclast formation. Endocrinology. 2005;146:728-735.[25] Huang RL, Yuan Y, Tu J, et al. Opposing TNF-α/IL-1β- and BMP-2-activated MAPK signaling pathways converge on Runx2 to regulate BMP-2-induced osteoblastic differentiation. Cell Death Dis. 2014;5:e1187.[26] Pawlowska E, Wysokiński D, Tokarz P, et al. Dexamethasone and 1,25-Dihydroxyvitamin D3 Reduce Oxidative Stress-Related DNA Damage in Differentiating Osteoblasts. Int J Mol Sci. 2014;15:16649-16664.[27] Almubarak S, Nethercott H, Freeberg M, et al. Tissue engineering strategies for promoting vascularized bone regeneration. Bone.2016;83:197-209.[28] Chen CT, Shih YR, Kuo TK,et al.Coordinated changes ofmitochondrial biogenesis and antioxidant enzymes during osteogenicdifferentiation of human mesenchymal stem cells, Stem Cells. 2008;(26):960-968.[29] Wang D,ChristensenK,Chawla G,et al. Franceschi,Isolation and characterization of MC3T3-E1 preosteoblast subclones withdistinct in vitro and in vivo differentiation/mineralization potential. J Bone Miner Res.1999;14(6):893-903.[30] Vasin MV,Ushakov IB,Semenova LA,et al.Radiation protective efficacy of alpha-adrenomimetics during local gamma irradiation of the skin. Radiats Biol Radioecol. 1999;39(2-3): 249-253.[31] Xiang B, Li YJ, Zhao XB, et al. Mechanism of the protective effect of phenylephrine pretreatment against irradiation-induced damage in the submandibular gland. Exp Ther Med. 2013;5:875-879.[32] Tanaka K, Hirai T, Kodama D, et al. alpha1B -Adrenoceptor signalling regulates bone formation through the up-regulation of CCAAT/enhancer-binding protein delta expression in osteoblasts. Br J Pharmacol. 2016;173:1058-1069.[33] Olarescu NC, Ueland T, Lekva T, et al. Adipocytes as a source of increased circulating levels of nicotinamide phosphoribosyltransferase/visfatin in active acromegaly.J Clin Endocrinol Metab. 2012;97(4):1355-1362.[34] He X, He J, Shi Y, et al. Nicotinamide phosphoribosyltransferase (Nampt) may serve as the marker for osteoblast differentiation of bone marrow-derived mesenchymal stem cells. Exp Cell Res. 2017;352:45-52.[35] Li Y, He J, He X, et al. Nampt expression increases during osteogenic differentiation of multi- and omnipotent progenitors.Biochem Biophys Res Commun. 2013;434(1): 117-123.[36] Marie-Charlotte Laiguillon XH, Carole Bougault, et al. Expression and function of visfatin (Nampt), an adipokine-enzyme involved in inflammatory pathways of osteoarthritis. Arthritis Res Ther. 2014;16:1-12.[37] Lin YC, Wu HC, Liao CC, et al. Secretion of one adipokine Nampt/Visfatin suppresses the inflammatory stress-induced NF-kappaB activity and affects Nampt-dependent cell viability in Huh-7 cells. Mediators Inflamm. 2015;2015:392471.[38] Romacho T, Villalobos LA, Cercas E, et al. Visfatin as a novel mediator released by inflamed human endothelial cells. PloS one. 2013;8:e78283.[39] Garten A, Petzold S, Korner A, et al. Nampt: linking NAD biology, metabolism and cancer. Trends Endocrinol Metab. 2009;20(3):130-138.[40] Imai S. Nicotinamide phosphoribosyltransferase (Nampt): A link between NAD biology, metabolism, and diseases. Curr Pharm Des.2009;15:20-28.[41] 吴梦然,张红胜.NAD+和Nampt与SIRT1的研究进展[J].医学综述, 2010;16(7):968-971.[42] Romanello M, Bicego M, Pirulli D, et al.ExtracellularNAD+: a novel autocrine/paracrine signal in osteoblast physiology. Biochem Biophys Res Commun. 2002;(299):424-431.[43] Ling M, Huang P, Islam S, et al. Epigenetic regulation of Runx2 transcription and osteoblast differentiation by nicotinamide phosphoribosyltransferase. Cell Biosci. 2017;7: 27.[44] Xie H, Tang SY, Luo XH, et al. Insulin?like efects of visfatin on human osteoblasts. Calcif Tissue Int. 2007;80:201-210. |
.jpg) 文题释义:
苯肾上腺素:苯肾上腺素是α1-肾上腺素受体(α1-Adrenergic receptor,α1-AR)特异性激动剂。α1-AR 广泛分布于机体的器官、组织和细胞中,介导多种生理反应。α1-AR能够通过增加成骨细胞转录因子的启动子或增强子结合蛋白的表达从而诱导其增殖,苯肾上腺素对成骨细胞的生长具有重要作用。
氧化应激损伤成骨细胞:H2O2氧化应激不仅影响成骨细胞的活性还对细胞分化产生作用,氧化应激可减少成骨细胞的成熟和细胞内矿化物质的合成,使其在成骨细胞形态学上表现为数量减少和体积皱缩等。
文题释义:
苯肾上腺素:苯肾上腺素是α1-肾上腺素受体(α1-Adrenergic receptor,α1-AR)特异性激动剂。α1-AR 广泛分布于机体的器官、组织和细胞中,介导多种生理反应。α1-AR能够通过增加成骨细胞转录因子的启动子或增强子结合蛋白的表达从而诱导其增殖,苯肾上腺素对成骨细胞的生长具有重要作用。
氧化应激损伤成骨细胞:H2O2氧化应激不仅影响成骨细胞的活性还对细胞分化产生作用,氧化应激可减少成骨细胞的成熟和细胞内矿化物质的合成,使其在成骨细胞形态学上表现为数量减少和体积皱缩等。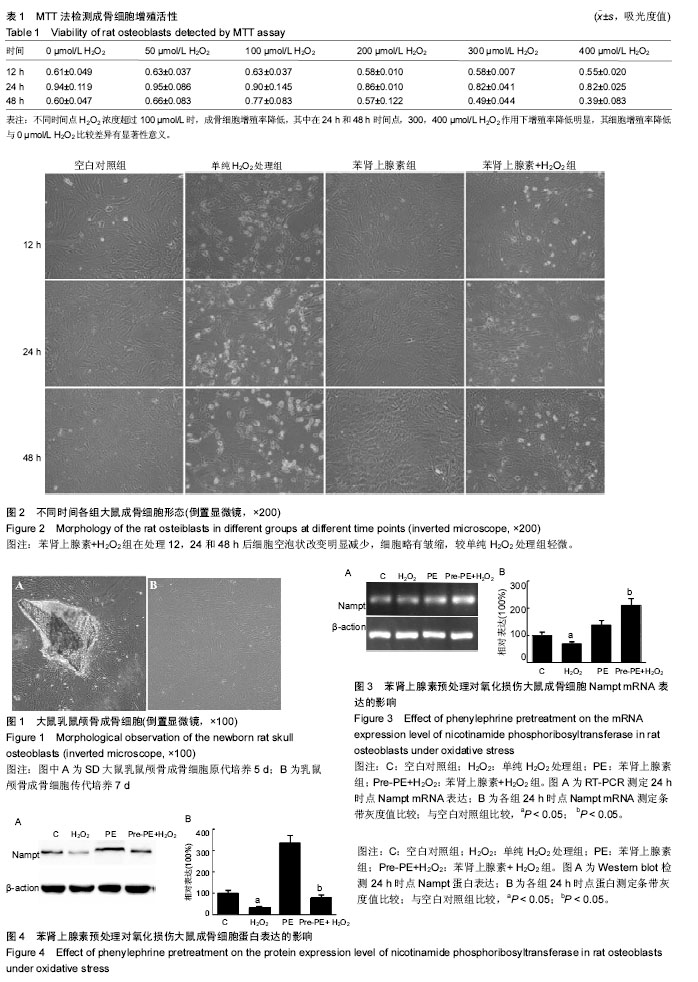
.jpg) 文题释义:
苯肾上腺素:苯肾上腺素是α1-肾上腺素受体(α1-Adrenergic receptor,α1-AR)特异性激动剂。α1-AR 广泛分布于机体的器官、组织和细胞中,介导多种生理反应。α1-AR能够通过增加成骨细胞转录因子的启动子或增强子结合蛋白的表达从而诱导其增殖,苯肾上腺素对成骨细胞的生长具有重要作用。
氧化应激损伤成骨细胞:H2O2氧化应激不仅影响成骨细胞的活性还对细胞分化产生作用,氧化应激可减少成骨细胞的成熟和细胞内矿化物质的合成,使其在成骨细胞形态学上表现为数量减少和体积皱缩等。
文题释义:
苯肾上腺素:苯肾上腺素是α1-肾上腺素受体(α1-Adrenergic receptor,α1-AR)特异性激动剂。α1-AR 广泛分布于机体的器官、组织和细胞中,介导多种生理反应。α1-AR能够通过增加成骨细胞转录因子的启动子或增强子结合蛋白的表达从而诱导其增殖,苯肾上腺素对成骨细胞的生长具有重要作用。
氧化应激损伤成骨细胞:H2O2氧化应激不仅影响成骨细胞的活性还对细胞分化产生作用,氧化应激可减少成骨细胞的成熟和细胞内矿化物质的合成,使其在成骨细胞形态学上表现为数量减少和体积皱缩等。