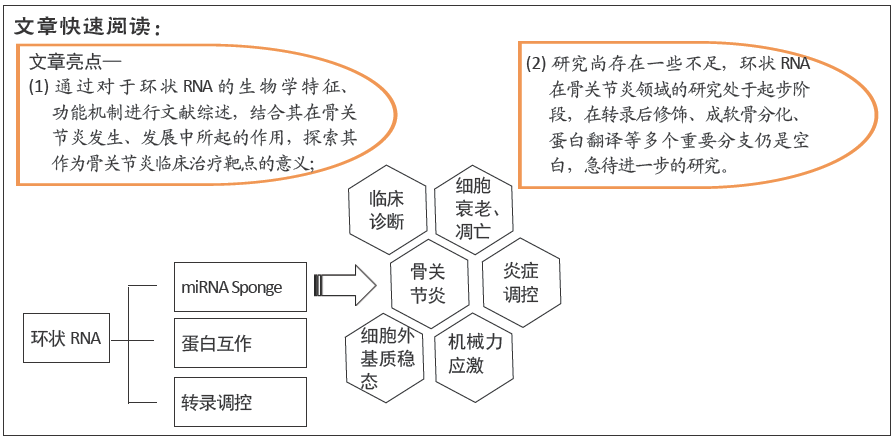
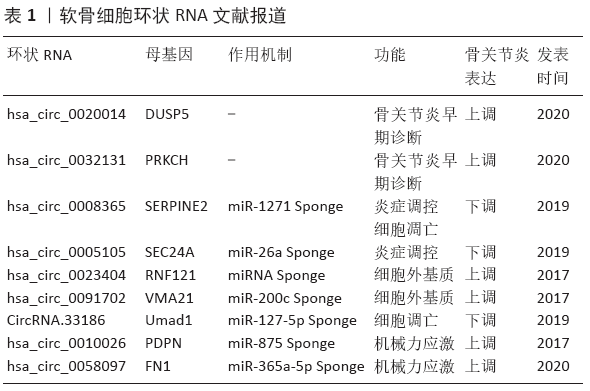
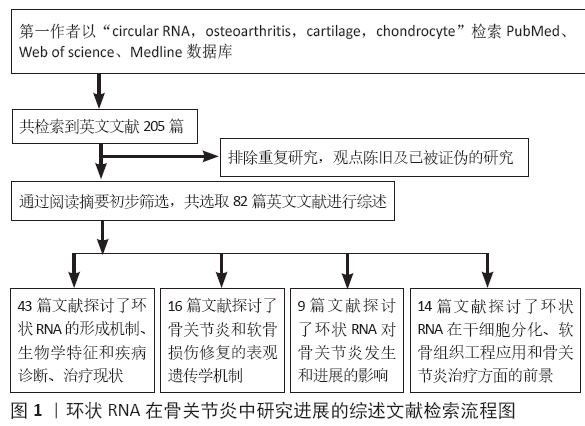
[1] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019; 393 (10182):1745-1759.
[2] DIDOMENICO CD, LINTZ M, BONASSAR LJ. Molecular transport in articular cartilage - what have we learned from the past 50 years? Nat Rev Rheumatol. 2018;14(7):393-403.
[3] ZHU Z, LI J, RUAN G, et al. Investigational drugs for the treatment of osteoarthritis, an update on recent developments. Exp Opin Invest Drugs. 2018;27(11):881-900.
[4] WANG KC, CHANG HY. Epigenomics: Technologies and Applications. Circulation Res.2018;122(9):1191-1199.
[5] RAMOS YF, MEULENBELT I. The role of epigenetics in osteoarthritis: current perspective. Curr Opin Rheumatol. 2017; 29(1):119-129.
[6] NIGRO JM, CHO KR, FEARON ER, et al. Scrambled exons. Cell. 1991; 64(3):607-613.
[7] COCQUERELLE C, MASCREZ B, HÉTUIN D, et al. Mis-splicing yields circular RNA molecules. FASEB J.1993;7(1):155-160.
[8] HSU MT, COCA-PRADOS M. Electron microscopic evidence for the circular form of RNA in the cytoplasm of eukaryotic cells. Nature. 1979; 280(5720): 339-340.
[9] WANG PL, BAO Y, YEE MC, et al. Circular RNA is expressed across the eukaryotic tree of life. PloS One. 2014;9(6):e90859.
[10] JECK WR, SORRENTINO JA, WANG K, et al. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA (New York, N.Y.). 2013;19(2): 141-157.
[11] SALZMAN J, GAWAD C, WANG PL, et al. Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types. PloS One. 2012; 7(2):e30733.
[12] SALZMAN J, CHEN RE, OLSEN MN, et al. Cell-type specific features of circular RNA expression. PLoS Genet. 2013; 9(9):e1003777.
[13] REYNARD LN, BARTER MJ. Osteoarthritis year in review 2019: genetics, genomics and epigenetics. Osteoarthritis Cartilage. 2020;28(3): 275-284.
[14] ZHANG XO, DONG R, ZHANG Y, et al. Diverse alternative back-splicing and alternative splicing landscape of circular RNAs. Genome Res. 2016; 26(9):1277-1287.
[15] CONN SJ, PILLMAN KA, TOUBIA J, et al. The RNA binding protein quaking regulates formation of circRNAs. Cell. 2015;160(6):1125-1134.
[16] ERRICHELLI L, DINI MODIGLIANI S, LANEVE P, et al. FUS affects circular RNA expression in murine embryonic stem cell-derived motor neurons. Nat Commun. 2017;8:14741.
[17] ASHWAL-FLUSS R, MEYER M, PAMUDURTI NR, et al. circRNA biogenesis competes with pre-mRNA splicing. Mol Cell. 2014;56(1):55-66.
[18] PAGLIARINI V, JOLLY A, BIELLI P, et al. Sam68 binds Alu-rich introns in SMN and promotes pre-mRNA circularization. Nucl Acids Res. 2020; 48(2):633-645.
[19] YU CY, LI TC, WU YY, et al. The circular RNA circBIRC6 participates in the molecular circuitry controlling human pluripotency. Nat Commun. 2017;8(1):1149.
[20] LIANG D, TATOMER DC, LUO Z, et al. The output of protein-coding genes shifts to circular rnas when the pre-mrna processing machinery is limiting. Mol Cell. 2017;68(5):940-954.e3.
[21] KRAMER MC, LIANG D, TATOMER DC, et al. Combinatorial control of Drosophila circular RNA expression by intronic repeats, hnRNPs, and SR proteins. Genes Dev. 2015;29(20):2168-2182.
[22] LI X, LIU CX, XUE W, et al. Coordinated circRNA Biogenesis and Function with NF90/NF110 in Viral Infection. Mol Cell. 2017;67(2): 214-227.e7.
[23] EISENBERG E, LEVANON EY. A-to-I RNA editing - immune protector and transcriptome diversifier. Nature reviews. Genetics. 2018;19(8): 473-490.
[24] GLAZAR P, PAPAVASILEIOU P, RAJEWSKY N. circBase: a database for circular RNAs. RNA. 2014;20(11):1666-1670.
[25] YOU X, VLATKOVIC I, BABIC A, et al. Neural circular RNAs are derived from synaptic genes and regulated by development and plasticity. Nat Neurosci. 2015;18(4):603-610.
[26] RYBAK-WOLF A, STOTTMEISTER C, GLAŽAR P, et al. Circular RNAs in the Mammalian Brain Are Highly Abundant, Conserved, and Dynamically Expressed. Mol Cell. 2015;58(5):870-885.
[27] DANAN M, SCHWARTZ S, EDELHEIT S, et al. Transcriptome-wide discovery of circular RNAs in Archaea. Nucleic Acids Res. 2012;40(7): 3131-3142.
[28] LI Y, ZHENG Q, BAO C, et al. Circular RNA is enriched and stable in exosomes: a promising biomarker for cancer diagnosis. Cell Res. 2015; 25(8): 981-984.
[29] BAHN JH, ZHANG Q, LI F, et al. The landscape of microRNA, Piwi-interacting RNA, and circular RNA in human saliva. Clin Chem. 2015; 61(1):221-230.
[30] YU F, XIE C, SUN J, et al. Circular RNA expression profiles in synovial fluid: a promising new class of diagnostic biomarkers for osteoarthritis. Int J Clin Exp Pathol. 2018;11(3):1338-1346.
[31] SUZUKI H, ZUO Y, WANG J, et al. Characterization of RNase R-digested cellular RNA source that consists of lariat and circular RNAs from pre-mRNA splicing. Nucleic Acids Res. 2006;34(8): e63.
[32] NICOLET BP, ENGELS S, AGLIALORO F, et al. Circular RNA expression in human hematopoietic cells is widespread and cell-type specific. Nucleic Acids Res. 2018;46(16):8168-8180.
[33] OU R, LV J, ZHANG Q, et al. circAMOTL1 Motivates AMOTL1 Expression to Facilitate Cervical Cancer Growth. Molecular therapy. Nucleic Acids. 2020;19:50-60.
[34] YANG Q, DU WW, WU N, et al. A circular RNA promotes tumorigenesis by inducing c-myc nuclear translocation. Cell Death Differ. 2017;24(9): 1609-1620.
[35] TAY Y, RINN J, PANDOLFI PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014;505(7483):344-352.
[36] KRISTENSEN LS, OKHOLM TLH, VENØ MT, et al. Circular RNAs are abundantly expressed and upregulated during human epidermal stem cell differentiation. RNA Biol. 2018;15(2):280-291.
[37] WU N, YUAN Z, DU KY, et al. Translation of yes-associated protein (YAP) was antagonized by its circular RNA via suppressing the assembly of the translation initiation machinery. Cell Death Differ. 2019;26(12): 2758-2773.
[38] ZENG Y, DU WW, WU Y, et al. A Circular RNA Binds To and Activates AKT Phosphorylation and Nuclear Localization Reducing Apoptosis and Enhancing Cardiac Repair. Theranostics. 2017;7(16):3842-3855.
[39] LI Z, HUANG C, BAO C, et al. Exon-intron circular RNAs regulate transcription in the nucleus. Nat Struct Mol Biol. 2015; 22(3): 256-264.
[40] YANG Y, FAN X, MAO M, et al. Extensive translation of circular RNAs driven by N(6)-methyladenosine. Cell Res. 2017; 27(5): 626-641.
[41] ZHANG M, HUANG N, YANG X, et al. A novel protein encoded by the circular form of the SHPRH gene suppresses glioma tumorigenesis. Oncogene. 2018; 37(13): 1805-1814.
[42] HAQUE S, HARRIES LW. Circular RNAs (circRNAs) in health and disease. Genes (Basel). 2017;8(12). pii: E353.
[43] HADDAD G, LORENZEN JM. Biogenesis and Function of Circular RNAs in Health and in Disease. Front Pharmacol. 2019;10:428.
[44] KRISTENSEN LS, HANSEN TB, VENO MT, et al. Circular RNAs in cancer: opportunities and challenges in the field. Oncogene. 2018; 37(5): 555-565.
[45] ALTESHA MA, NI T, KHAN A, et al. Circular RNA in cardiovascular disease. J Cell Physiol. 2019; 234(5): 5588-5600.
[46] LU S, YANG X, WANG C, et al. Current status and potential role of circular RNAs in neurological disorders. J Neurochem. 2019;150(3): 237-248.
[47] WANG Y, WU C, ZHANG Y, et al. Screening for differentially expressed circRNA between Kashin-Beck disease and osteoarthritis patients based on circRNA chips. Clin Chim Acta. 2020;501:92-101.
[48] WANG Y, WU C, YANG Y, et al. Preliminary Exploration of hsa_circ_0032131 Levels in Peripheral Blood as a Potential Diagnostic Biomarker of Osteoarthritis. Genet Test Mol Biomarkers. 2019;23(10): 717-721.
[49] SCANZELLO CR. Role of low-grade inflammation in osteoarthritis. Curr Opin Rheumatol. 2017;29(1):79-85.
[50] GREENE MA, LOESER RF. Aging-related inflammation in osteoarthritis. Osteoarthritis Cartilage. 2015;23(11):1966-1971.
[51] ZHOU ZB, HUANG GX, FU Q, et al. circRNA.33186 Contributes to the Pathogenesis of Osteoarthritis by Sponging miR-127-5p. Mol Ther. 2019;27(3):531-541.
[52] SHEN S, WU Y, CHEN J, et al. CircSERPINE2 protects against osteoarthritis by targeting miR-1271 and ETS-related gene. Ann Rheum Dis. 2019;78(6):826-836.
[53] STEGEN S, LAPERRE K, EELEN G, et al. HIF-1α metabolically controls collagen synthesis and modification in chondrocytes. Nature. 2019; 565(7740): 511-515.
[54] WU Y, ZHANG Y, ZHANG Y, et al. CircRNA hsa_circ_0005105 upregulates NAMPT expression and promotes chondrocyte extracellular matrix degradation by sponging miR-26a. Cell Biol Int. 2017; 41(12): 1283-1289.
[55] LIU Q, ZHANG X, HU X, et al. Circular RNA Related to the Chondrocyte ECM Regulates MMP13 Expression by Functioning as a MiR-136 ‘Sponge’ in Human Cartilage Degradation. Sci Rep. 2016; 6:22572.
[56] CHENG X, ZHANG L, ZHANG K, et al. Circular RNA VMA21 protects against intervertebral disc degeneration through targeting miR-200c and X linked inhibitor-of-apoptosis protein. Ann Rheum Dis. 2018; 77(5):770-779.
[57] HARBO M, DELAISSE JM, KJAERSGAARD-ANDERSEN P, et al. The relationship between ultra-short telomeres, aging of articular cartilage and the development of human hip osteoarthritis. Mech Ageing Dev. 2013; 134(9): 367-372.
[58] LIU L, RANDO TA. Manifestations and mechanisms of stem cell aging. J Cell Biol. 2011; 193(2): 257-266.
[59] JURK D, WANG C, MIWA S, et al. Postmitotic neurons develop a p21-dependent senescence-like phenotype driven by a DNA damage response. Aging Cell. 2012;11(6):996-1004.
[60] DU WW, FANG L, YANG W, et al. Induction of tumor apoptosis through a circular RNA enhancing Foxo3 activity. Cell Death Differ. 2017; 24(2): 357-370.
[61] LI T, WANG G. Computer-aided targeting of the PI3K/Akt/mTOR pathway: toxicity reduction and therapeutic opportunities. Int J Mol Sci. 2014;15(10): 18856-18891.
[62] JERE SW, HOURELD NN, ABRAHAMSE H. Role of the PI3K/AKT (mTOR and GSK3β) signalling pathway and photobiomodulation in diabetic wound healing. Cytokine Growth Factor Rev. 2019;50:52-59.
[63] MI B, XIONG Y, CHEN L, et al. CircRNA AFF4 promotes osteoblast cells proliferation and inhibits apoptosis via the Mir-7223-5p/PIK3R1 axis. Aging. 2019; 11(24):11988-12001.
[64] THYSEN S, LUYTEN FP, LORIES RJU. Targets, models and challenges in osteoarthritis research. Dis Model Mech. 2015;8(1):17-30.
[65] LIU Q, ZHANG X, HU X, et al. Emerging Roles of circRNA Related to the Mechanical Stress in Human Cartilage Degradation of Osteoarthritis. Mol Ther Nucleic Acids. 2017;7:223-230.
[66] XIAO L, DING B, XU S, et al. circRNA_0058097 promotes tension-induced degeneration of endplate chondrocytes by regulating HDAC4 expression through sponge adsorption of miR-365a-5p. J Cell Biochem, 2020;121(1):418-429.
[67] NISHIMORI S, LAI F, SHIRAISHI M, et al. PTHrP targets HDAC4 and HDAC5 to repress chondrocyte hypertrophy. JCI Insight. 2019;4(5). pii: 97903.
[68] WANG H, ZHANG H, SUN Q, et al. Chondrocyte mTORC1 activation stimulates miR-483-5p via HDAC4 in osteoarthritis progression. J Cell Physiol. 2019;234(3):2730-2740.
[69] XIAO W, ADHIKARI S, DAHAL U, et al. Nuclear m(6)A Reader YTHDC1 Regulates mRNA Splicing. Mol Cell. 2016;61(4):507-519.
[70] COOTS RA, LIU XM, MAO Y, et al. m6A Facilitates eIF4F-Independent mRNA Translation. Mol Cell. 2017;68(3):504-514.e7.
[71] XIAO CL, ZHU S, HE M, et al. N-Methyladenine DNA Modification in the Human Genome. Mol Cell. 2018;71(2):306-318.e7.
[72] MIN KW, ZEALY RW, DAVILA S, et al. Profiling of m6A RNA modifications identified an age-associated regulation of AGO2 mRNA stability. Aging Cell. 2018;17(3):e12753.
[73] LI Z, ZHANG R, YANG X, et al. Analysis of gene expression and methylation datasets identified ADAMTS9, FKBP5, and PFKBF3 as biomarkers for osteoarthritis. J Cell Physiol. 2019;234(6):8908-8917.
[74] TENG H, MAO F, LIANG J, et al. Transcriptomic signature associated with carcinogenesis and aggressiveness of papillary thyroid carcinoma. Theranostics. 2018;8(16):4345-4358.
[75] YANG Y, FAN X, MAO M, et al. Extensive translation of circular RNAs driven by N-methyladenosine. Cell research. 2017;27(5):626-641.
[76] CHEN YG, CHEN R, AHMAD S, et al. N6-Methyladenosine Modification Controls Circular RNA Immunity. Mol Cell. 2019;76(1):96-109.e9.
[77] CHEN RX, CHEN X, XIA LP, et al. N-methyladenosine modification of circNSUN2 facilitates cytoplasmic export and stabilizes HMGA2 to promote colorectal liver metastasis. Nat Commun. 2019;10(1):4695.
[78] GARIKIPATI VNS, VERMA SK, CHENG Z, et al. Circular RNA CircFndc3b modulates cardiac repair after myocardial infarction via FUS/VEGF-A axis. Nat Commun. 2019;10(1):4317.
[79] HUANG A, ZHENG H, WU Z, et al. Circular RNA-protein interactions: functions, mechanisms, and identification. Theranostics. 2020;10(8): 3503-3517.
[80] ZHOU W, LIN J, ZHAO K, et al. Single-cell profiles and clinically useful properties of human mesenchymal stem cells of adipose and bone marrow origin. Am J Sports Med. 2019;47(7):1722-1733.
[81] ZHU Y, GUI W, LIN X, et al. Knock-down of circular RNA H19 induces human adipose-derived stem cells adipogenic differentiation via a mechanism involving the polypyrimidine tract-binding protein 1. Exp Cell Res. 2020;387(2):111753.
[82] CHERUBINI A, BARILANI M, ROSSI RL, et al. FOXP1 circular RNA sustains mesenchymal stem cell identity via microRNA inhibition. Nucleic Acids Res. 2019;47(10):5325-5340.
|