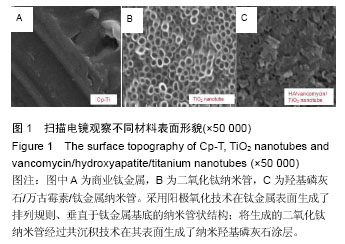
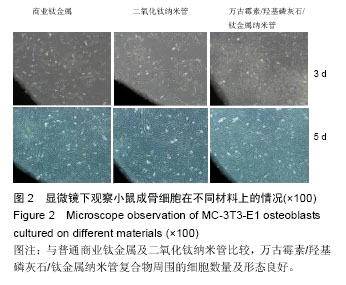
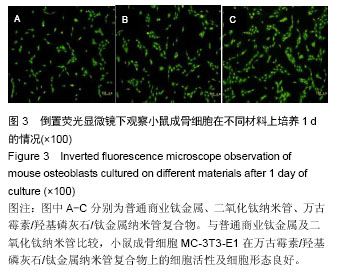
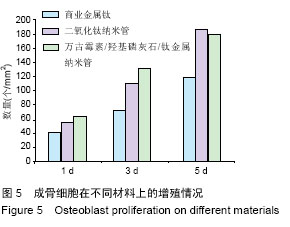
| [1] Glibert M,De Bruyn H,Östman PO.Six-Year Radiographic, Clinical, and Soft Tissue Outcomes of Immediately Loaded, Straight-Walled, Platform-Switched, Titanium-Alloy Implants with Nanosurface Topography.Int J Oral Maxillofac Implants. 2016;31(1):167-171.[2] Raphel J,Karlsson J,Galli S,et al.Engineered protein coatings to improve the osseointegration of dental and orthopaedic implants. Biomaterials. 2016;83(6): 269-282.[3] Ataya A,Kline KP,Cope J,et al.Titanium exposure and yellow nail syndrome.Respir Med Case Rep. 2015;16: 146-147.[4] Hara D, Nakashima Y, Sato T, et al. Bone bonding strength of diamond-structured porous titanium-alloy implants manufactured using the electron beam-melting technique.Mater Sci Eng C Mater Biol Appl.2016;59:1047-1052.[5] Hara D,Nakashima Y,Sato T,et al. Bone bonding strength of diamond-structured porous titanium-alloy implants manufactured using the electron beam-melting technique.Mater Sci Eng C Mater Biol Appl.2016;59:1047-1052.[6] Choi JY,Lee HJ,Jang JU,et al.Comparison between bioactive fluoride modified and bioinert anodically oxidized implant surfaces in early bone response using rabbit tibia model.Implant Dent. 2012;21(2): 124-128.[7] Liu Y,Jiang T,Zhou Y,et al.Evaluation of the attachment, proliferation, and differentiation of osteoblast on a calcium carbonate coating on titanium surface. Materials Science and Engineering C. 2011;31:1055-1061.[8] Park JW,Suh JY,Chung HJ.Effects of calcium ion incorporation on osteoblast gene expression in MC3T3-E1 cells cultured on microstructured titanium surfaces. J Biomed Mater Res A.2008;86(1):117-126.[9] Anselme K.Osteoblast adhesion on biomaterials. Biomaterials.2000;21(7):667-681.[10] Aw MS,Simovic S,Addai-Mensah J,Losic D:.Polymeric micelles in porous and nanotubular implants as a new system for extended delivery of poorly soluble drugs. J Mater Chem.2011;21(20):7082-7089.[11] Bjursten LM,Rasmusson L,Oh S,et al.Titanium dioxide nanotubes enhance bone bonding in vivo.J Biomed Mater Res A.2010;92(3):1218-1224. [12] Burns K,Yao C,Webster TJ.Increased chondrocyte adhesion on nanotubular anodized titanium.J Biomed Mater Res A.2009;88(3):561-568. [13] Chang CH,Lee HC,Chen CC, et al.A novel rotating electrochemically anodizing process to fabricate titanium oxide surface nanostructures enhancing the bioactivity of osteoblastic cells.J Biomed Mater Res A.2012;100(7):1687-1695. [14] Gulati K,Aw MS,Findlay D,et al.Local drug delivery to the bone by drug-releasing implants: perspectives of nano-engineered titania nanotube arrays.Ther Deliv. 2012;3(7):857-873.[15] Gulati K,Ramakrishnan S,Aw MS,et al.Biocompatible polymer coating of titania nanotube arrays for improved drug elution and osteoblast adhesion.Acta Biomater.2012;8(1):449-456.[16] Hu Y,Cai K,Luo Z,et al.TiO2 nanotubes as drug nanoreservoirs for the regulation of mobility and differentiation of mesenchymal stem cells.Acta Biomater. 2012;8(1):439-448. [17] Jayaraman M,Meyer U,Bühner M,et al.Influence of titanium surfaces on attachment of osteoblast-like cells in vitro.Biomaterials.2004;25(4):625-631.[18] Popat KC,Eltgroth M,LaTempa TJ,et al.Titania nanotubes: a novel platform for drug-eluting coatings for medical implants.Small.2007;3(11):1878-1881. [19] Shrestha NK,Macak JM,Schmidt-Stein F,et al. Magnetically guided titania nanotubes for site-selective photocatalysis and drug release.Angew Chem Int Ed Engl. 2009;48(5):969-972. [20] Peng L,Mendelsohn AD,LaTempa TJ,et al.Long-term small molecule and protein elution from TiO2 nanotubes. Nano Lett.2009;9(5):1932-1926.[21] Wang Y,Yuan L,Yao C,et al.Cytotoxicity Evaluation of pH-Controlled Antitumor Drug Release System of Titanium Dioxide Nanotubes.J Nanosci Nanotechnol. 2015;15(6):4143-4148.[22] Doadrio AL,Conde A,Arenas MA,et al.Use of anodized titanium alloy as drug carrier: Ibuprofen as model of drug releasing.Int J Pharm. 2015;492(1-2): 207-212[23] Przekora A,Benko A,Nocun M,et al.Titanium coated with functionalized carbon nanotubes--a promising novel material for biomedical application as an implantable orthopaedic electronic device.Mater Sci Eng C Mater Biol Appl.2014;45:287-296.[24] Yan L,Jiang DM.Study of bone-like hydroxyapatite/polyamino acid composite materials for their biological properties and effects on the reconstruction of long bone defects.Drug Des Devel Ther.2015;17(9):6497-6508[25] Clauss M,Van Der Straeten C,Goossens M. Prospective five-year subsidence analysis of a cementless fully hydroxyapatite-coated femoral hip arthroplasty component.Hip Int. 2014;24(1):91-97.[26] Li S,Huang B,Chen Y,et al. Hydroxyapatite-coated femoral stems in primary total hip arthroplasty: a meta-analysis of randomized controlled trials.Int J Surg. 2013;11(6):477-482.[27] Oonishi H. A long term histological analysis of effect of interposed hydroxyapatite between bone and bone cement in THA and TKA.J Long Term Eff Med Implants. 2012;22(2):165-176.[28] Rajesh P,Muraleedharan CV,Komath M,et al.Laser surface modification of titanium substrate for pulsed laser deposition of highly adherent hydroxyapatite.J Mater Sci Mater Med.2011;22(7):1671-1679.[29] Cooper LF. A role for surface topography in creating and maintaining bone at titanium endosseous implants.J Prosthet Dent.2000;84(5):522-534.[30] von Wilmowsky C,Bauer S,Lutz R,et al.In vivo evaluation of anodic TiO2 nanotubes: an experimental study in the pig. J Biomed Mater Res B Appl Biomater. 2009;89(1):165-171.[31] Bjursten LM, Rasmusson L, Oh S, et al. Titanium dioxide nanotubes enhance bone bonding in vivo. J Biomed Mater Res A. 2010; 92(3):1218-1224.[32] Tang J,Hu J,Kang L,et al.The use of vancomycin in the treatment of adult patients with methicillin-resistant Staphylococcus aureus (MRSA) infection: a survey in a tertiary hospital in China.Int J Clin Exp Med. 2015; 8(10): 19436-19441.[33] Jeon YL,Kim MH,Yang HS,et al.Optimum initial loading dose of vancomycin for pneumonia caused by methicillin-resistant Staphylococcus aureus.J Infect. 2016;72(1):115-118.[34] Kim SH,Kim MG,Kim SS,et al.Change in Detection Rate of Methicillin-Resistant Staphylococcus aureus and Pseudomonas aeruginosa and Their Antibiotic Sensitivities in Patients with Chronic Suppurative Otitis Media.J Int Adv Otol.2015;11(2):151-156.[35] Hahn A,Frenck RW Jr,Allen-Staat M,et al.Evaluation of Target Attainment of Vancomycin Area Under the Curve in Children With Methicillin-Resistant Staphylococcus Aureus Bacteremia.Ther Drug Monit. 2015;37(5):619-625. [36] Hanrahan TP,Kotapati C,Roberts MJ,et al.Factors associated with vancomycin nephrotoxicity in the critically ill.Anaesth Intensive Care. 2015;43(5): 594-599.[37] Jacqz-Aigrain E,Leroux S,Zhao W,et al.How to use vancomycin optimally in neonates: remaining questions. Expert Rev Clin Pharmacol. 2015;8(5): 635-648. [38] Hanrahan T,Whitehouse T,Lipman J,et al. Vancomycin-associated nephrotoxicity: A meta-analysis of administration by continuous versus intermittent infusion.Int J Antimicrob Agents. 2015; 46(3):249-253. [39] Liu Y,Yin Y,Liu XZ,et al.Retrospective Analysis of Vancomycin Nephrotoxicity in Elderly Chinese Patients.Pharmacology.2015;95(5-6):279-284. [40] Edin ML,Miclau T,Lester GE,et al.Effect of cefazolin and vancomycin on osteoblasts in vitro. Clin Orthop Relat Res.1996;(333):245-251.[41] Rathbone CR,Cross JD,Brown KV,et al. Effect of various concentrations of antibiotics on osteogenic cell viability and activity.J Orthop Res. 2011;29(7): 1070-1074. |
.jpg)


.jpg)