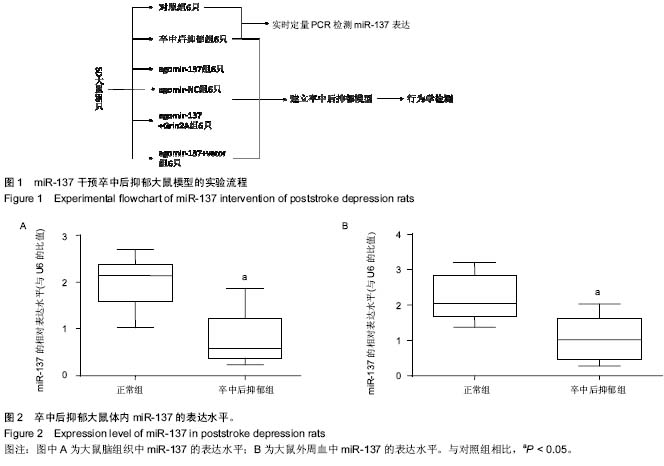
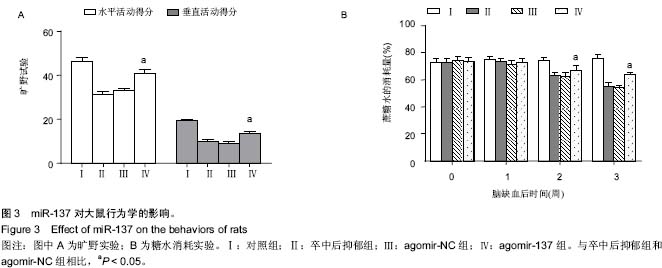
| [1] Whyte EM, Mulsant BH. Post stroke depression: epidemiology, pathophysiology, and biological treatment. Biol Psychiatry. 2002;52(3):253-264.
[2] Souza AC, Rocha MO, Teixeira AL, et al. Depressive symptoms and disability in chagasic stroke patients: impact on functionality and quality of life. J Neurol Sci. 2013;324(1-2): 34-37.
[3] Hama S, Yamashita H, Yamawaki S, et al. Post-stroke depression and apathy: Interactions between functional recovery, lesion location, and emotional response. Psychogeriatrics. 2011;11(1):68-76.
[4] Farner L, Wagle J, Engedal K, et al. Depressive symptoms in stroke patients: a 13 month follow-up study of patients referred to a rehabilitation unit. J Affect Disord. 2010;127(1-3): 211-218.
[5] Van Wynsberghe PM, Chan SP, Slack FJ, et al. Analysis of microRNA expression and function. Methods Cell Biol. 2011; 106:219-252.
[6] Berezikov E. Evolution of microRNA diversity and regulation in animals. Nat Rev Genet. 2011;12(12):846-860.
[7] Ebert MS, Sharp PA. Roles for microRNAs in conferring robustness to biological processes. Cell. 2012;149(3):515-524.
[8] Mendell JT, Olson EN. MicroRNAs in stress signaling and human disease. Cell. 2012;148(6):1172-1187.
[9] Smalheiser NR, Lugli G, Rizavi HS, et al. MicroRNA expression is down-regulated and reorganized in prefrontal cortex of depressed suicide subjects. PLoS One. 2012;7(3): e33201.
[10] Bocchio-Chiavetto L, Maffioletti E, Bettinsoli P, et al. Blood microRNA changes in depressed patients during antidepressant treatment. Eur Neuropsychopharmacol. 2013;23(7):602-611.
[11] Baudry A, Mouillet-Richard S, Schneider B, et al. miR-16 targets the serotonin transporter: a new facet for adaptive responses to antidepressants. Science. 2010;329(5998): 1537-1541.
[12] Mouillet-Richard S, Baudry A, Launay JM, et al. MicroRNAs and depression. Neurobiol Dis. 2012;46(2):272-278.
[13] O'Connor RM, Dinan TG, Cryan JF. Little things on which happiness depends: microRNAs as novel therapeutic targets for the treatment of anxiety and depression. Mol Psychiatry. 2012;17(4):359-376.
[14] Tan KS, Armugam A, Sepramaniam S, et al. Expression profile of MicroRNAs in young stroke patients. PLoS One. 2009;4(11):e7689.
[15] Jeyaseelan K, Lim KY, Armugam A. MicroRNA expression in the blood and brain of rats subjected to transient focal ischemia by middle cerebral artery occlusion. Stroke. 2008; 39(3):959-966.
[16] Dharap A, Vemuganti R. Ischemic pre-conditioning alters cerebral microRNAs that are upstream to neuroprotective signaling pathways. J Neurochem. 2010;113(6):1685-1691.
[17] Nishida F, Morel GR, Hereñú CB, et al. Restorative effect of intracerebroventricular insulin-like growth factor-I gene therapy on motor performance in aging rats. Neuroscience. 2011;177:195-206.
[18] Hahn CD, Manlhiot C, Schmidt MR, et al. Remote ischemic per-conditioning: a novel therapy for acute stroke? Stroke. 2011;42(10):2960-2962.
[19] Longa EZ, Weinstein PR, Carlson S, et al. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke. 1989;20(1):84-91.
[20] Willner P. Chronic mild stress (CMS) revisited: consistency and behavioural-neurobiological concordance in the effects of CMS. Neuropsychobiology. 2005;52(2):90-110.
[21] Wang SH, Zhang ZJ, Guo YJ, et al. Decreased expression of serotonin 1A receptor in the dentate gyrus in association with chronic mild stress: a rat model of post-stroke depression. Psychiatry Res. 2009;170(2-3):245-251.
[22] Duan Q, Wang X, Gong W, et al. ER stress negatively modulates the expression of the miR-199a/214 cluster to regulates tumor survival and progression in human hepatocellular cancer. PLoS One. 2012;7(2):e31518.
[23] Geekiyanage H, Jicha GA, Nelson PT, et al. Blood serum miRNA: non-invasive biomarkers for Alzheimer's disease. Exp Neurol. 2012;235(2):491-496.
[24] Smrt RD, Szulwach KE, Pfeiffer RL, et al. MicroRNA miR-137 regulates neuronal maturation by targeting ubiquitin ligase mind bomb-1. Stem Cells. 2010;28(6):1060-1070.
[25] Cummings E, Donohoe G, Hargreaves A, et al. Mood congruent psychotic symptoms and specific cognitive deficits in carriers of the novel schizophrenia risk variant at MIR-137. Neurosci Lett. 2013;532:33-38.
[26] Kwon E, Wang W, Tsai LH. Validation of schizophrenia- associated genes CSMD1, C10orf26, CACNA1C and TCF4 as miR-137 targets. Mol Psychiatry. 2013;18(1):11-12.
[27] Mozhui K, Karlsson RM, Kash TL, et al. Strain differences in stress responsivity are associated with divergent amygdala gene expression and glutamate-mediated neuronal excitability. J Neurosci. 2010;30(15):5357-5367.
[28] Reutlinger C, Helbig I, Gawelczyk B, et al. Deletions in 16p13 including GRIN2A in patients with intellectual disability, various dysmorphic features, and seizure disorders of the rolandic region. Epilepsia. 2010;51(9):1870-1873.
[29] Cherlyn SY, Woon PS, Liu JJ, et al. Genetic association studies of glutamate, GABA and related genes in schizophrenia and bipolar disorder: a decade of advance. Neurosci Biobehav Rev. 2010;34(6):958-977.
[30] Paoletti P. Molecular basis of NMDA receptor functional diversity. Eur J Neurosci. 2011;33(8):1351-1365.
[31] Szakacs R, Janka Z, Kalman J. The "blue" side of glutamatergic neurotransmission: NMDA receptor antagonists as possible novel therapeutics for major depression. Neuropsychopharmacol Hung. 2012;14(1):29-40.
[32] Autry AE, Adachi M, Nosyreva E, et al. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature. 2011;475(7354):91-95.
[33] Kavalali ET, Monteggia LM. Synaptic mechanisms underlying rapid antidepressant action of ketamine. Am J Psychiatry. 2012;169(11):1150-1156.
[34] Monteggia LM, Gideons E, Kavalali ET. The role of eukaryotic elongation factor 2 kinase in rapid antidepressant action of ketamine. Biol Psychiatry. 2013;73(12):1199-1203.
[35] Naughton M, Clarke G, O'Leary OF, et al. A review of ketamine in affective disorders: current evidence of clinical efficacy, limitations of use and pre-clinical evidence on proposed mechanisms of action. J Affect Disord. 2014;156: 24-35.
[36] Castillo CS, Schultz SK, Robinson RG. Clinical correlates of early-onset and late-onset poststroke generalized anxiety. Am J Psychiatry. 1995;152(8):1174-1179.
[37] Cam E, Yulug B, Ozan E. MK 801: a possible neuroprotective agent by poststroke depression? J Neuropsychiatry Clin Neurosci. 2008;20(3):367-368.
[38] Robinson RG, Jorge RE, Clarence-Smith K, et al. Double-blind treatment of apathy in patients with poststroke depression using nefiracetam. J Neuropsychiatry Clin Neurosci. 2009;21(2):144-151.
[39] Hama S, Yamashita H, Yamawaki S, et al. Post-stroke depression and apathy: Interactions between functional recovery, lesion location, and emotional response. Psychogeriatrics. 2011;11(1):68-76.
[40] Lim AL, Taylor DA, Malone DT. Consequences of early life MK-801 administration: long-term behavioural effects and relevance to schizophrenia research. Behav Brain Res. 2012;227(1):276-286.
[41] Pae CU, Marks DM, Han C, et al. Does neurotropin-3 have a therapeutic implication in major depression? Int J Neurosci. 2008;118(11):1515-1522.
[42] Yulug B. Neuroprotective treatment strategies for poststroke mood disorders: A minireview on atypical neuroleptic drugs and selective serotonin re-uptake inhibitors. Brain Res Bull. 2009;80(3):95-99.
[43] Buller KM, Carty ML, Reinebrant HE, et al. Minocycline: a neuroprotective agent for hypoxic-ischemic brain injury in the neonate? J Neurosci Res. 2009;87(3):599-608
[44] Meloni BP, Campbell K, Zhu H, et al. In search of clinical neuroprotection after brain ischemia: the case for mild hypothermia (35 degrees C) and magnesium. Stroke. 2009; 40(6):2236-2240. |