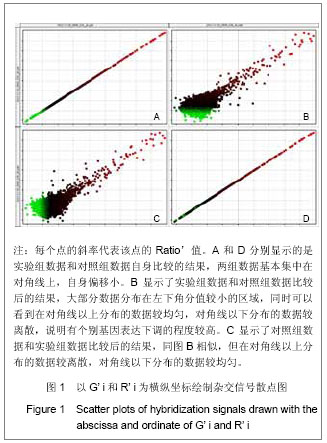
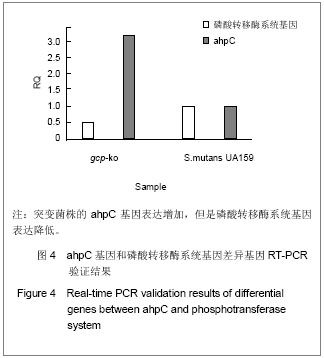
| [1] Yamada T, Kose H, Ohta T, et al. Genetic dissection of complex genetic factor involved in NIDDM of OLETF rat. Exp Diabetes Res. 2012;2012:582546.[2] Mueller TF, Mas VR. Microarray applications in nephrology with special focus on transplantation. J Nephrol. 2012;25(5): 589-602.[3] Fujita K, Fukuda M, Iwahashi H. Significance of comprehensive gene expression analysis for evaluation of biological effects of manufactured nanomaterials. Nihon Eiseigaku Zasshi. 2012;67(3):390-395.[4] 闫文娟.外源性单磷酸鸟苷环二聚体抑制变形链球菌生物膜的形成能力[J]. 中国组织工程研究,2012,16(16): 2939-2942.[5] 闫文娟,徐树军,谢苗苗,等.外源性单磷酸鸟苷环二聚体对变形链球菌基因表达的影响[J].中国组织工程研究, 2012,16(8): 1451-1454.[6] Li Z, Chen JH, Hao Y, et al. Structures of the PelD cyclic diguanylate effector involved in pellicle formation in Pseudomonas aeruginosa PAO1. J Biol Chem. 2012;287(36): 30191-30204.[7] Zaade D, Schmitz J, Benke E, et al. Distinct signal transduction pathways downstream of the (P)RR revealed by microarray and ChIP-chip analyses. PLoS One. 2013;8(3):e57674. [8] Zhou J, Sayre DA, Wang J, et al. Endo-S-c-di-GMP analogues-polymorphism and binding studies with class I riboswitch. Molecules. 2012;17(11):13376-13389.[9] Li W, He ZG. LtmA, a novel cyclic di-GMP-responsive activator, broadly regulates the expression of lipid transport and metabolism genes in Mycobacterium smegmatis. Nucleic Acids Res. 2012;40(22):11292-11307.[10] Petrova OE, Sauer K. Dispersion by Pseudomonas aeruginosa requires an unusual posttranslational modification of BdlA. Proc Natl Acad Sci U S A. 2012;109(41):16690- 16695. [11] Stelitano V, Brandt A, Fernicola S, et al. Probing the activity of diguanylate cyclases and c-di-GMP phosphodiesterases in real-time by CD spectroscopy. Nucleic Acids Res. 2013;41(7): e79. [12] Chen Y, Chai Y, Guo JH,et al. Evidence for cyclic Di-GMP-mediated signaling in Bacillus subtilis. J Bacteriol. 2012;194(18):5080-5090.[13] Smith KD,Lipchock SV,Livingston AL,et al. Structural and biochemical determinants of ligand binding by the c-di-GMP riboswitch . Biochemistry. 2010;49(34):7351-7359. [14] Tschowri N, Lindenberg S, Hengge R. Molecular function and potential evolution of the biofilm-modulating blue light-signalling pathway of Escherichia coli. Mol Microbiol. 2012;85(5):893-906. [15] Cruz DP, Huertas MG, Lozano M, et al. omparative analysis of diguanylate cyclase and phosphodiesterase genes in Klebsiella pneumoniae. BMC Microbiol. 2012;12:139.[16] Chen MW, Kotaka M, Vonrhein C, et al. Structural insights into the regulatory mechanism of the response regulator RocR from Pseudomonas aeruginosa in cyclic Di-GMP signaling. J Bacteriol. 2012;194(18):4837-4846.[17] Miller MB, Tang YW. Basic concepts of microarrays and potential applications in clinical microbiology. Clin Microbiol Rev. 2009;22(4):611-633.[18] Procop GW. Molecular diagnostics for the detection and characterization of microbial pathogens. Clin Infect Dis. 2007;45 Suppl 2:S99-S111. [19] Charoenlap N, Shen Z, McBee ME, et al. Alkyl hydroperoxide reductase is required for Helicobacter cinaedi intestinal colonization and survival under oxidative stress in BALB/c and BALB/c interleukin-10-/- mice. Infect Immun. 2012;80(3): 921-928.[20] Higuchi M, Yamamoto Y, Kamio Y. Molecular biology of oxygen tolerance in lactic acid bacteria: Functions of NADH oxidases and Dpr in oxidative stress. J Biosci Bioeng. 2000; 90(5):484-493.[21] Mohammadian T, Doosti M, Paknejad M, et al. Preparative SDS-PAGE Electroelution for Rapid Purification of Alkyl Hydroperoxide Reductase from Helicobacter pylori. Iran J Public Health. 2010;39(1):85-91.[22] Lechardeur D, Fernandez A, Robert B, et al. The 2-Cys peroxiredoxin alkyl hydroperoxide reductase c binds heme and participates in its intracellular availability in Streptococcus agalactiae. J Biol Chem. 2010;285(21):16032-16041.[23] Yamamoto Y, Higuchi M, Poole LB, et al. Identification of a new gene responsible for the oxygen tolerance in aerobic life of Streptococcus mutans.Biosci Biotechnol Biochem. 2000; 64(5):1106-1109.[24] Wasim M, Bible AN, Xie Z, et al. Alkyl hydroperoxide reductase has a role in oxidative stress resistance and in modulating changes in cell-surface properties in Azospirillum brasilense Sp245. Microbiology. 2009;155(Pt 4):1192-1202.[25] Jönsson TJ, Ellis HR, Poole LB. Cysteine reactivity and thiol-disulfide interchange pathways in AhpF and AhpC of the bacterial alkyl hydroperoxide reductase system. Biochemistry. 2007;46(19):5709-5721.[26] Yamamoto Y, Sato Y, Takahashi-Abbe S, et al. Cloning and sequence analysis of the pfl gene encoding pyruvate formate-lyase from Streptococcus mutans.Infect Immun. 1996;64(2):385-391.[27] Elizarov SM, Alekseeva MG, Novikov FN, et al. Identification of phosphorylation sites in aminoglycoside phosphotransferase VIII from Streptomyces rimosus. Biochemistry.2012;77(11):1258-1265.[28] Neumann S, Grosse K, Sourjik V. Chemotactic signaling via carbohydrate phosphotransferase systems in Escherichia coli. Proc Natl Acad Sci U S A. 2012;109(30):12159-12164.[29] Zeng L, Burne RA. Multiple sugar: phosphotransferase system permeases participate in catabolite modification of gene expression in Streptococcus mutans. Mol Microbiol. 2008;70(1):197-208.[30] Webb AJ, Homer KA, Hosie AH. A phosphoenolpyruvate- dependent phosphotransferase system is the principal maltose transporter in Streptococcus mutans. J Bacteriol. 2007;189(8):3322-3327. |

.jpg)
.jpg)
.jpg)