| [1]Solomon L. Hereditary multiple exostosis. Am J Hum Genet. 1964;16: 351-363.[2]Jones KB. Glycobiology and the growth plate: current concepts in multiple hereditary exostoses. J Pediatr Orthop. 2011;31(5): 577-586.[3]Tahasildar N, Sudesh P, Goni V, et al. Giant osteochondroma of axis in a child with multiple hereditary exostoses: case report and review of literature. J Pediatr Orthop B. 2012;21(3): 280-285.[4]Franch J, Font J, Ramis A, et al. Multiple cartilaginous exostosis in a Golden Retriever cross-bred puppy. Clinical, radiographic and backscattered scanning microscopy findings. Vet Comp Orthop Traumatol. 2005;18(3):189-193.[5]Kyriazoglou AI, Dimitriadis E, Arnogiannaki N, et al. Similar cytogenetic findings in two synchronous secondary peripheral chondrosarcomas in a patient with multiple osteochondromas. Cancer Genet. 2011;204(12): 677-681.[6]郭永成,王永堂,陆亚东,等. 儿童遗传性多发性骨软骨瘤软骨帽的超微结构观察[J]. 临床与实验病理学杂志, 2013, 29(2): 171-174.[7]Huvos AG, Marcove RC. Chondrosarcoma in the young. A clinicopathologic analysis of 79 patients younger than 21 years of age. Am J Surg Pathol. 1987; 11(12): 930-942.[8]Legeai-Mallet L, Munnich A, Maroteaux P, et al. Incomplete penetrance and expressivity skewing in hereditary multiple exostoses. Clin Genet. 1997;52(1):12-16.[9]肖福英,蒋林彬,刘健翔,等. 遗传性多发性骨软骨瘤一家系11例[J]. 中华医学遗传学杂志,2005,22(5):574.[10]Rozeman LB, de Bruijn IH, Bacchini P, et al. Dedifferentiated peripheral chondrosarcomas: regulation of EXT-downstream molecules and differentiation-related genes. Mod Pathol. 2009; 22(11): 1489-1498.[11]Cook A, Raskind W, Blanton SH, et al. Genetic heterogeneity in families with hereditary multiple exostoses. Am J Hum Genet. 1993;53(1):71-79.[12]Wu YQ, Heutink P, de Vries BB, et al. Assignment of a second locus for multiple exostoses to the pericentromeric region of chromosome 11. Hum Mol Genet. 1994;3(1): 167-171.[13]Wuyts W, Ramlakhan S, Van Hul W, et al. Refinement of the multiple exostoses locus (EXT2) to a 3-cM interval on chromosome 11. Am J Hum Genet. 1995; 57(2): 382-387.[14]Le Merrer M, Legeai-Mallet L, Jeannin PM, et al. A gene for hereditary multiple exostoses maps to chromosome 19p. Hum Mol Genet. 1994;3(5): 717-722.[15]Jennes I, Pedrini E, Zuntini M, et al. Multiple osteochondromas: mutation update and description of the multiple osteochondromas mutation database (MOdb). Hum Mutat. 2009;30(12): 1620-1627.[16]Ishimaru D, Gotoh M, Takayama S, et al. Large-scale mutational analysis in the EXT1 and EXT2 genes for Japanese patients with multiple osteochondromas. BMC Genet.2016;17:52.[17]Ahn J, Lüdecke HJ, Lindow S, et al. Cloning of the putative tumour suppressor gene for hereditary multiple exostoses (EXT1). Nat Genet. 1995;11(2): 137-143.[18]Wuyts W, Van Hul W, Wauters J, et al. Positional cloning of a gene involved in hereditary multiple exostoses. Hum Mol Genet. 1996;5(10): 1547-1557.[19]Szuhai K, Jennes I, de Jong D, et al. Tiling resolution array-CGH shows that somatic mosaic deletion of the EXT gene is causative in EXT gene mutation negative multiple osteochondromas patients. Hum Mutat. 2011; 32(2): E2036-2049.[20]Li Y, Wang J, Tang J, et al. Heterogeneous spectrum of EXT gene mutations in Chinese patients with hereditary multiple osteochondromas. Medicine (Baltimore). 2018;97:e12855.[21]Scl S, Impo R, Takata RI, et al. Analysis of mutations in EXT1 and EXT2 in Brazilian patients with multiple osteochondromas. Mol Genet Genomic Med. 2018;6:382-392.[22]Wise CA, Clines GA, Massa H, et al. Identification and localization of the gene for EXTL, a third member of the multiple exostoses gene family. Genome Res. 1997;7(1): 10-16.[23]Wuyts W, Van Hul W, Hendrickx J, et al. Identification and characterization of a novel member of the EXT gene family, EXTL2. Eur J Hum Genet. 1997;5(6): 382-389.[24]Van Hul W, Wuyts W, Hendrickx J, et al. Identification of a third EXT-like gene (EXTL3) belonging to the EXT gene family. Genomics. 1998;47(2): 230-237.[25]Kang QL, Xu J, Zhang Z, et al. Mutation screening for the EXT1 and EXT2 genes in Chinese patients with multiple osteochondromas. Arch Med Res. 2013;44(7): 542-548.[26]Stancheva-Ivanova MK, Wuyts W, van Hul E, et al. Clinical and molecular studies of EXT1/EXT2 in Bulgaria. J Inherit Metab Dis. 2011;34(4): 917-921.[27]Lindahl U. Heparan sulfate-protein interactions--a concept for drug design. Thromb Haemost. 2007;98(1): 109-115.[28]Li JP. Heparin, heparan sulfate and heparanase in cancer: remedy for metastasis. Anticancer Agents Med Chem. 2008. 8(1): 64-76.[29]McCormick C, Leduc Y, Martindale D, et al. The putative tumour suppressor EXT1 alters the expression of cell-surface heparan sulfate. Nat Genet. 1998;19(2): 158-161.[30]de Andrea CE, Prins FA, Wiweger MI, et al. Growth plate regulation and osteochondroma formation: insights from tracing proteoglycans in zebrafish models and human cartilage. J Pathol. 2011; 224(2): 160-168.[31]Hameetman L, David G, Yavas A, et al. Decreased EXT expression and intracellular accumulation of heparan sulphate proteoglycan in osteochondromas and peripheral chondrosarcomas. J Pathol. 2007;211(4): 399-409.[32]Clément A, Wiweger M, der Hardt SV, et al. Regulation of zebrafish skeletogenesis by ext2/dackel and papst1/pinscher. PLoS Genet. 2008;4(7): e1000136.[33]Huegel J, Enomoto-Iwamoto M, Sgariglia F, et al. Heparanase stimulates chondrogenesis and is up-regulated in human ectopic cartilage: a mechanism possibly involved in hereditary multiple exostoses. Am J Pathol. 2015;185:1676-1685.[34]Schwartz NB, Domowicz M. Chondrodysplasias due to proteoglycan defects. Glycobiology. 2002;12(4): 57R-68R.[35]Turnbull J, Powell A, Guimond S. Heparan sulfate: decoding a dynamic multifunctional cell regulator. Trends Cell Biol. 2001; 11(2): 75-82.[36]Hecht JT, Hall CR, Snuggs M, et al. Heparan sulfate abnormalities in exostosis growth plates. Bone. 2002; 31(1): 199-204.[37]Zak BM, Schuksz M, Koyama E, et al. Compound heterozygous loss of Ext1 and Ext2 is sufficient for formation of multiple exostoses in mouse ribs and long bones. Bone. 2011;48(5): 979-987.[38]Jones KB, Pacifici M, Hilton MJ. Multiple hereditary exostoses (MHE): elucidating the pathogenesis of a rare skeletal disorder through interdisciplinary research. Connect Tissue Res. 2014;55(2): 80-88.[39]Duncan G, McCormick C, Tufaro F. The link between heparan sulfate and hereditary bone disease: finding a function for the EXT family of putative tumor suppressor proteins. J Clin Invest. 2001; 108(4): 511-516.[40]Beltrami G, Ristori G, Scoccianti G, et al. Hereditary Multiple Exostoses: a review of clinical appearance and metabolic pattern. Clin Cases Miner Bone Metab. 2016;13(2): 110-118.[41]Lin X, Wei G, Shi Z, et al. Disruption of gastrulation and heparan sulfate biosynthesis in EXT1-deficient mice. Dev Biol. 2000;224(2): 299-311.[42]Hameetman L, Kok P, Eilers PH, et al. The use of Bcl-2 and PTHLH immunohistochemistry in the diagnosis of peripheral chondrosarcoma in a clinicopathological setting. Virchows Arch. 2005;446(4): 430-437.[43]Modrowski D, Orosco A, Thévenard J, Fromigué O, Marie PJ. Syndecan-2 overexpression induces osteosarcoma cell apoptosis: Implication of syndecan-2 cytoplasmic domain and JNK signaling. Bone. 2005;37(2): 180-189.[44]Mundy C, Bello A, Sgariglia F, et al. HhAntag, a Hedgehog Signaling Antagonist, Suppresses Chondrogenesis and Modulates Canonical and Non-Canonical BMP Signaling. J Cell Physiol. 2016;231:1033-1044.[45]Koziel L, Kunath M, Kelly OG, et al. Ext1-dependent heparan sulfate regulates the range of Ihh signaling during endochondral ossification. Dev Cell. 2004; 6(6): 801-813.[46]Benoist-Lasselin C, de Margerie E, Gibbs L, et al. Defective chondrocyte proliferation and differentiation in osteochondromas of MHE patients. Bone. 2006;39(1): 17-26.[47]Osterholm C, Barczyk MM, Busse M, et al. Mutation in the heparan sulfate biosynthesis enzyme EXT1 influences growth factor signaling and fibroblast interactions with the extracellular matrix. J Biol Chem. 2009; 284(50): 34935-34943.[48]Häcker U, Nybakken K, Perrimon N. Heparan sulphate proteoglycans: the sweet side of development. Nat Rev Mol Cell Biol. 2005; 6(7): 530-541.[49]Shimokawa K, Kimura-Yoshida C, Nagai N, et al. Cell surface heparan sulfate chains regulate local reception of FGF signaling in the mouse embryo. Dev Cell. 2011;21(2): 257-272.[50]Stickens D, Zak BM, Rougier N, et al. Mice deficient in Ext2 lack heparan sulfate and develop exostoses. Development. 2005; 132(22): 5055-5068.[51]Huegel J, Mundy C, Sgariglia F, et al. Perichondrium phenotype and border function are regulated by Ext1 and heparan sulfate in developing long bones: a mechanism likely deranged in Hereditary Multiple Exostoses. Dev Biol. 2013; 377(1): 100-12.[52]Matsumoto Y, Matsumoto K, Irie F, et al. Conditional ablation of the heparan sulfate-synthesizing enzyme Ext1 leads to dysregulation of bone morphogenic protein signaling and severe skeletal defects. J Biol Chem. 2010; 285(25): 19227-19234.[53]Sinha S, Mundy C, Bechtold T, et al. Unsuspected osteochondroma-like outgrowths in the cranial base of Hereditary Multiple Exostoses patients and modeling and treatment with a BMP antagonist in mice. PLoS Genet. 2017; 13(4): e1006742.[54]Inubushi T, Lemire I, Irie F, et al. Palovarotene Inhibits Osteochondroma Formation in a Mouse Model of Multiple Hereditary Exostoses. J Bone Miner Res. 2018;33:658-666.[55]Mundy C, Yang E, Takano H, et al. Heparan sulfate antagonism alters bone morphogenetic protein signaling and receptor dynamics, suggesting a mechanism in hereditary multiple exostoses. J Biol Chem, 2018;293:7703-7716.[56]Day TF, Guo X, Garrett-Beal L, et al. Wnt/beta-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev Cell. 2005;8(5): 739-750.[57]Sgariglia F, Pedrini E, Bradfield JP, et al. The type 2 diabetes associated rs7903146 T allele within TCF7L2 is significantly under-represented in Hereditary Multiple Exostoses: insights into pathogenesis. Bone. 2015;72:123-127.[58]Kikuchi A, Yamamoto H, Sato A, et al. New insights into the mechanism of Wnt signaling pathway activation. Int Rev Cell Mol Biol. 2011; 291: 21-71.[59]Yuasa T, Kondo N, Yasuhara R, et al. Transient activation of Wnt/{beta}-catenin signaling induces abnormal growth plate closure and articular cartilage thickening in postnatal mice. Am J Pathol. 2009;175(5): 1993-2003.[60]Bishop JR, Schuksz M, Esko JD. Heparan sulphate proteoglycans fine-tune mammalian physiology. Nature. 2007; 446(7139): 1030-1037.[61]Pedrini E, Jennes I, Tremosini M, et al. Genotype-phenotype correlation study in 529 patients with multiple hereditary exostoses: identification of "protective" and "risk" factors. J Bone Joint Surg Am. 2011;93(24): 2294-2302.[62]Rider CC, Mulloy B. Bone morphogenetic protein and growth differentiation factor cytokine families and their protein antagonists. Biochem J. 2010;429(1): 1-12.[63]Bornemann DJ, Duncan JE, Staatz W, et al. Abrogation of heparan sulfate synthesis in Drosophila disrupts the Wingless, Hedgehog and Decapentaplegic signaling pathways. Development. 2004;131(9): 1927-1938.[64]Bornemann DJ, Park S, Phin S, et al. A translational block to HSPG synthesis permits BMP signaling in the early Drosophila embryo. Development. 2008;135(6): 1039-1047.[65]Freeman SD, Moore WM, Guiral EC, et al. Extracellular regulation of developmental cell signaling by XtSulf1. Dev Biol. 2008; 320(2): 436-445.[66]Jones KB, Piombo V, Searby C, et al. A mouse model of osteochondromagenesis from clonal inactivation of Ext1 in chondrocytes. Proc Natl Acad Sci U S A. 2010;107(5): 2054-2059.[67]Nozawa S, Inubushi T, Irie F, et al. Osteoblastic heparan sulfate regulates osteoprotegerin function and bone mass. JCI Insight. 2018; 3(3). pii: 89624.[68]Matsumoto K, Irie F, Mackem S, et al. A mouse model of chondrocyte-specific somatic mutation reveals a role for Ext1 loss of heterozygosity in multiple hereditary exostoses. Proc Natl Acad Sci U S A. 2010; 107(24): 10932-10937. |
.jpg)
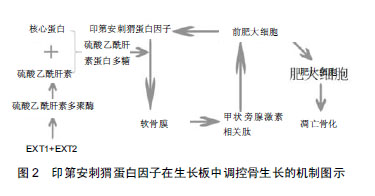

.jpg)
.jpg)