| [1] Krause-Titz UR, Warneke N, Freitag-Wolf S, et al. Factors influencing the outcome (GOS) in reconstructive cranioplasty. Neurosurg Rev. 2016;39(1):133-139.[2] Kurland DB, Khaladj-Ghom A, Stokum JA, et al. Complications associated with decompressive craniectomy: a systematic review. Neurocrit Care. 2015;23:292-304.[3] Yamaura A, Makino H. Neurological deficits in the presence of the sinking skin flap following decompressive craniectomy. Neurol Med Chir (Tokyo). 1977;17(1Pt1):43-53.[4] Kasprzak P, Ormezowska E, Jaskólski D. Cranioplasty as the return-to-work factor-112 patients with cranial defects treated in the Department of Neurosurgery at the Medical University Of Lodz. Int J Occup Med Environ Health. 2017;30(5):803-809.[5] 刁云锋,杨细平.几种颅骨修补材料的临床应用及不良反应防治[J].中国组织工程研究与临床康复,2011,15(16):2985-2989.[6] 王玖,郑新瑞,费舟.颅骨修补时机和应用材料的研究进展[J].临床神经外科杂志,2016,13(5):397-400.[7] Smith DM, Cooper GM, Afifi AM, et al. Regenerative surgery in cranioplasty revisited. Plast Reconstr Surg. 2011;128(5):1053-1060.[8] Khader BA, Towler MR. Materials and techniques used in cranioplasty fixation: a review. Mater Sci Eng. 2016;66:315-322.[9] Zanaty M, Chalouhi N, Starke RM, et al. Complications following cranioplasty: incidence and predictors in 348 cases. J Neurosurg. 2015;123(1):182-188.[10] Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Br Med J. 2009;6(7):e1000100.[11] OCEBM Levels of Evidence Working Group. The Oxford Levels of Evidence 2. Oxford Cent Evidence-Based Med 2012. 2015.[12] Wells G, Shea B, O’Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Heal Res Inst 2006. 2016.[13] 程序曲,赵富文,楮荣涛,等.自体骨瓣、普通钛网、预塑二维钛网修补颅骨缺损比较[J].中国临床神经外科杂志,2012,17(1):24-26.[14] 曹轲,李宗正,刘玉飞,等.自体骨与钛网两种颅骨修补方案的疗效分析[J].中华神经外科杂志,2015,31(9):918-922.[15] Lee L, Ker J, Quah BL, et al. A retrospective analysis and review of an institution's experience with the complications of cranioplasty. Br J Neurosurg. 2013;27(5):629-635.[16] Bobinski L, Koskinen LD, Lindvall P. Complications following cranioplasty using autologous bone or polymethylmethacrylate- Retrospective experience from a single center. Clin Neurol Neurosurg. 2013;115(9):1788-1791.[17] Ng ZY, Ang WJJ, Nawaz I. Computer-Designed Polyetheretherketone Implants Versus Titanium Mesh (±Acrylic Cement) in Alloplastic Cranioplasty. J Craniofac Surg. 2014;25(2):e185-e189.[18] Klinger DR, Madden C. Autologous and acrylic cranioplasty: a review of 10 years and 258 cases. World Neurosurg. 2014;82(3-4):e525-e530.[19] Gilardino MS, Karunanayake M, Al-Humsi T, et al. A Comparison and cost analysis of cranioplasty techniques. J Craniofac Surg. 2015;26(1):113-117.[20] Morton RP, Abecassis IJ, Hanson JF, et al. Predictors of infection after 754 cranioplasty operations and the value of intraoperative cultures for cryopreserved bone flaps. J Neurosurg. 2016;125(3):766-770.[21] Honeybul S, Morrison DA, Ho KM, et al. A randomized controlled trial comparing autologous cranioplasty with custom-made titanium cranioplasty. J Neurosurg. 2017;126(1):81-90.[22] Kim SH, Kang DS, Cheong JH, et al. Comparison of complications following cranioplasty using a sterilized autologous bone flap or polymethyl methacrylate. Korean J Neurotrauma. 2017;13(1):15.[23] Sakamoto S, Eguchi K, Kiura Y, et al. CT perfusion imaging in the syndrome of the sinking skin flap before and after cranioplasty. Clin Neurol Neurosurg. 2006;108(6):583-585.[24] Di Stefano C, Rinaldesi M L, Quinquinio C, et al. Neuropsychological changes and cranioplasty: a group analysis. Brain Inj. 2016;30(2):164-171.[25] Paredes I, Castaño AM, Cepeda S, et al. The effect of cranioplasty on cerebral hemodynamics as measured by perfusion computed tomography and doppler ultrasonography. J Neurotrauma. 2016; 33(17):1586-1597.[26] Gooch MR, Gin GE, Kenning TJ, et al. Complications of cranioplasty following decompressive craniectomy: analysis of 62 cases. Neurosurg Focus. 2009;26(6):E9.[27] Schwarz F, Dünisch P, Walter J, et al. Cranioplasty after decompressive craniectomy: is there a rationale for an initial artificial bone-substitute implant? A single-center experience after 631 procedures. J Neurosurg. 2016;124(3):710-715.[28] Schoekler B, Trummer M. Prediction parameters of bone flap resorption following cranioplasty with autologous bone. Clin Neurol Neurosurg. 2014;120:64-67.[29] Chang TJ, Choi JW, Ra YS, et al. Changes in graft thickness after skull defect reconstruction with autogenous split calvarial bone graft. J Craniofac Surg. 2014;25(4):1241-1244.[30] Corliss B, Gooldy T, Vaziri S, et al. Complications after in vivo and ex vivo autologous bone flap storage for cranioplasty: a comparative analysis of the literature. World Neurosurg. 2016;96:510-515.[31] Kim JS, Park IS, Kim SK, et al. Analysis of the risk factors affecting the surgical site infection after cranioplasty following decompressive craniectomy. Korean J Neurotrauma. 2015;11(2):100.[32] Tsang AC, Hui VK, Lui W, et al. Complications of post-craniectomy cranioplasty: Risk factor analysis and implications for treatment planning. J Clin Neurosci. 2015;22(5):834-837.[33] Riordan MA, Simpson VM, Hall WA. Analysis of factors contributing to infections after cranioplasty: a single-institution retrospective chart review. World Neurosurg. 2016;87:207-213.[34] Thesleff T, Lehtimaki K, Niskakangas T, et al. Cranioplasty with adipose-derived stem cells, beta-tricalcium phosphate granules and supporting mesh: six-year clinical follow-up results. Stem Cells Transl Med. 2017;6(7):1576-1582.[35] Di Bella C, Farlie P, Penington AJ. Bone regeneration in a rabbit critical-sized skull defect using autologous adipose-derived cells. Tissue Eng Part A. 2008;14(4):483-490.[36] Roldán JC, Schulz P. BMP-7 preserves surface integrity of degradable-ceramic cranioplasty in a göttingen minipig model. Plast Reconstr Surg. 2017;5(3):e1255.[37] Smith DM, Cooper GM, Afifi A M, et al. Regenerative Surgery in Cranioplasty Revisited. Plast Reconstr Surg. 2011;128(5):1053-1060.[38] Sun M, He Y, Zhou T, et al. Adipose extracellular matrix/stromal vascular fraction gel secretes angiogenic factors and enhances skin wound healing in a murine model. Biomed Res Int. 2017;2017:1-11.[39] Liao HT, Marra KG. Application of platelet-rich plasma and platelet-rich fibrin in fat grafting: basic science and literature review. Tissue Eng. 2014;20(4):267-276. |
.jpg)


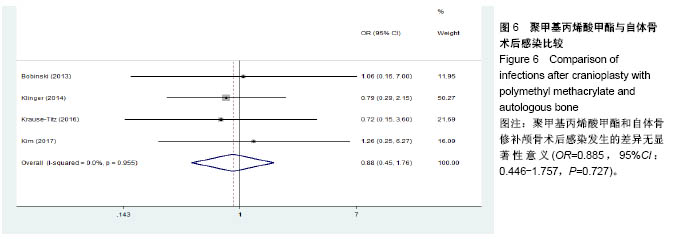
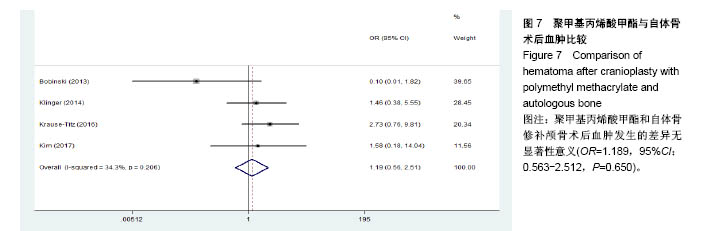
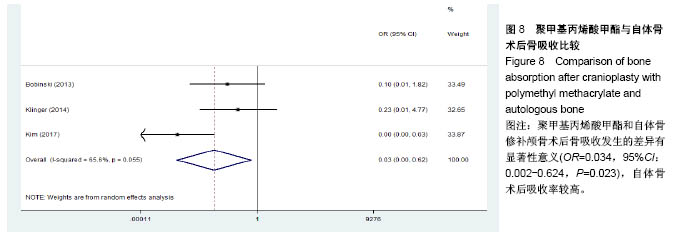
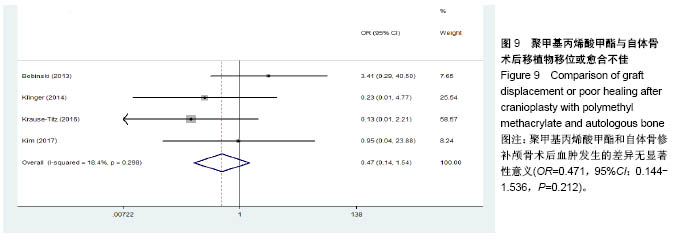
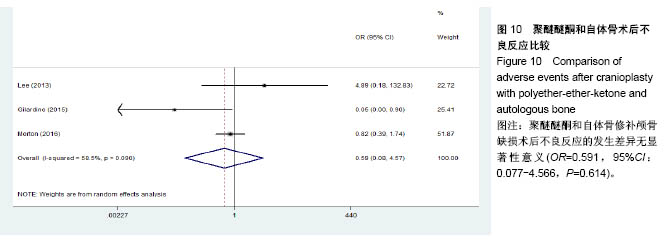
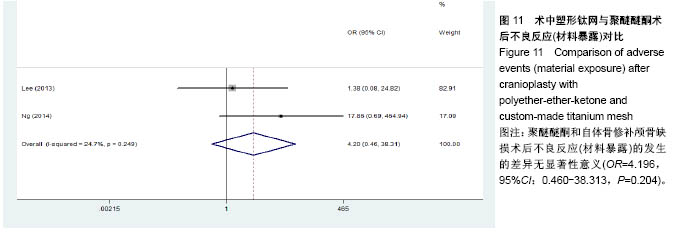
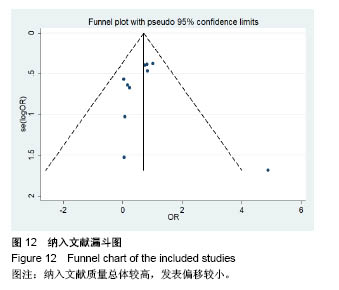
.jpg)