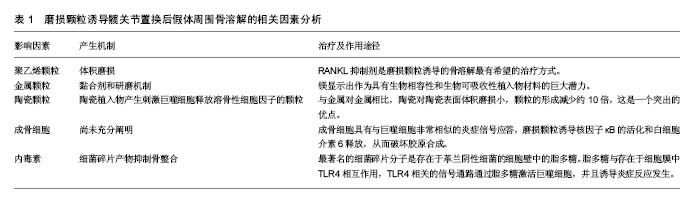
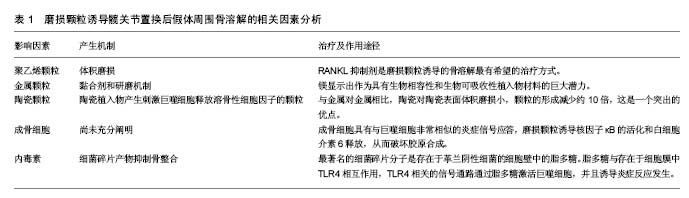
| [1] Greidanus NV, Peterson RC, Masri BA, et al. Quality of life outcomes in revision versus primary total knee arthroplasty. J Arthroplasty. 2011; 26(4): 615-620. [2] Harris WH. Osteolysis and particle disease in hip replacement. A review. Acta Orthop Scand. 1994; 65(1): 113-123.[3] Lewallen EA, Riester SM, Bonin CA, et al. Biological strategies for improved osseointegration and osteoinduction of porous metal orthopedic implants. Tissue Eng Part B Rev. 2014;21(2): 218-230.[4] Gallo J, Raska M, Mrázek F, et al. Bone remodeling, particle disease and individual susceptibility to periprosthetic osteolysis. Physiol Res. 2008; 57(3): 339-349. [5] Omran MTA, Miley DD, McLeod DE, et al. Retrospective assessment of survival rate for short endosseous dental implants. Implant Dentistry. 2015;24(2):185-191. [6] Pedersen AB, Mehnert F, Odgaard A, et al. Existing data sources for clinical epidemiology: The Danish Knee Arthroplasty Register. Clin Epidemiol 2012; 4: 125-35. [7] Paxton EW, Furnes O, Namba RS, et al. Comparison of the Norwegian knee arthroplasty register and a United States arthroplasty registry. J Bone Joint Surg Am 2011; 93(Suppl. 3): 20-30. [8] Illingworth KD, Wachter N, Maloney WJ, et al. Advances in acetabular osteolysis: biomarkers, imaging, and pharmacologic management. Instr Course Lect.2014; 63: 177-186.[9] Glyn-Jones S, Thomas GER, Garfjeld-Roberts P, et al. The John Charnley Award: highly crosslinked polyethylene in total hip arthroplasty decreases long-term wear: a double-blind randomized trial. Clin Orthop Relat Res.2015;473(2): 432-438. [10] Bitar D, Parvizi J. Biological response to prosthetic debris. World J Orthop. 2015; 6(2): 172-189. [11] Rujitanaroj PO, Jao B, Yang J, et al. Controlling fibrous capsule formation through long-term down-regulation of collagen type I (COL1A1) expression by nanofiber-mediated siRNA gene silencing. Acta Biomater. 2013;9(1):4513-4524.[12] Messier SP, Gutekunst DJ, Davis C, et al. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005; 52(7): 2026-2032.[13] Gallo J, Goodman SB, Konttinen YT, et al. Particle disease: biologic mechanisms of periprosthetic osteolysis in total hip arthroplasty. Innate Immun. 2013;19(2): 213-224. [14] Davies AP, Willert HG, Campbell PA, et al. An unusual lymphocytic perivascular infiltration in tissues around contemporary metal-on-metal joint replacements. J Bone Joint Surg Am.2005; 87(1): 18-27.[15] Liu X, Qu X, Wu C, et al. The effect of enoxacin on osteoclastogenesis and reduction of titanium particle-induced osteolysis via suppression of JNK signaling pathway. Biomaterials. 2014;35(22): 5721-5730. [16] Duisabeau L, Combrade P, Forest B. Environmental effect on fretting of metallic materials for orthopaedic implants. Wear. 2004; 256: 805-816.[17] Schramm M, Wirtz DC, Holzwarth U, et al. The Morse taper junction in modular revision hip replacement--a biomechanical and retrieval analysis. Biomed Tech (Berl). 2000; 45(4): 105-109. [18] Jauch SY, Huber G, Sellenschloh K, et al. Micromotions at the taper interface between stem and neck adapter of a bimodular hip prosthesis during activities of daily living. J Orthop Res. 2013; 31(8): 1165-1171. [19] Mistry JB, Chughtai M, Elmallah RK, et al. Trunnionosis in total hip arthroplasty: a review. J Orthop Traumatol. 2016; 17(1): 1-6. [20] Pastides PS, Dodd M, Sarraf KM, et al. Trunnionosis: A pain in the neck. World J Orthop. 2013; 4(4): 161-166. [21] Morawietz L, Classen RA, Schröder JH, et al. Proposal for a histopathological consensus classification of the periprosthetic interface membrane. J Clin Pathol. 2006; 59(6): 591-597.[22] Mert M, Oztürkmen Y, Ünkar EA, et al. Sciatic nerve compression by an extrapelvic cyst secondary to wear debris after a cementless total hip arthroplasty: A case report and literature review. Int J Surg Case Rep. 2013; 4(10): 805-808. [23] Berend ME, Ritter MA, Hyldahl HC, et al. Implant migration and failure in total knee arthroplasty is related to body mass index and tibial component size. J Arthroplasty. 2008; 23(6) (Suppl. 1): 104-109. [24] Wong J, Steklov N, Patil S, et al. Predicting the effect of tray malalignment on risk for bone damage and implant subsidence after total knee arthroplasty. J Orthop Res. 2011; 29(3): 347-353. [25] Pap G, Machner A, Rinnert T, et al. Development and characteristics of a synovial-like interface membrane around cemented tibial hemiarthroplasties in a novel rat model of aseptic prosthesis loosening. Arthritis Rheum. 2001; 44(4): 956-963.[26] Gouron R, Petit L, Boudot C, et al. Osteoclasts and their precursors are present in the induced-membrane during bone reconstruction using the Masquelet technique. J Tissue Eng Regen Med. 2017;11(2):382-389. [27] Windisch C, Windisch B, Kolb W, et al. Osteodensitometry measurements of periprosthetic bone using dual energy X-ray absorptiometry following total knee arthroplasty. Arch Orthop Trauma Surg. 2012; 132(11): 1595-1601. [28] Banerjee S, Pivec R, Issa K, et al. Large-diameter femoral heads in total hip arthroplasty: an evidence-based review. Am J Orthop. 2014; 43(11): 506-12. [29] Musib MK. Response to ultra-high molecular weight polyethylene particles. Am J Biomed Eng. 2011; 1(1): 7-12.[30] Jiang Y, Jia T, Wooley PH, et al. Current research in the pathogenesis of aseptic implant loosening associated with particulate wear debris. Acta Orthop Belg. 2013;79(1): 1-9.[31] Hodrick JT, Severson EP, McAlister DS, et al. Highly crosslinked polyethylene is safe for use in total knee arthroplasty. Clin Orthop Relat Res. 2008; 466(11): 2806-2812. [32] Liu A, Ingham E, Fisher J, et al. Generation of a large volume of clinically relevant nanometre-sized ultra-high-molecular-weight polyethylene wear particles for cell culture studies. Proc Inst Mech Eng H. 2014;228(4):418-426. [33] Carossino AM, Carulli C, Ciuffi S, et al. Hypersensitivity reactions to metal implants: laboratory options. BMC Musculoskelet Disord. 2016;17(1):486. [34] Archibeck MJ, Jacobs JJ, Roebuck KA, et al. The basic science of periprosthetic osteolysis. Instr Course Lect. 2001;50:185-195.[35] Goodman SB, Gibon E, Yao Z. The basic science of periprosthetic osteolysis. Instr Course Lect. 2013; 62: 201-206.[36] Wang CT, Lin YT, Chiang BL, et al. Over-expression of receptor activator of nuclear factor-kappaB ligand (RANKL), inflammatory cytokines, and chemokines in periprosthetic osteolysis of loosened total hip arthroplasty. Biomaterials. 2010; 31(1): 77-82. [37] Jiang X, Sato T, Yao Z, et al. Local delivery of mutant CCL2 protein-reduced orthopaedic implant wear particle-induced osteolysis and inflammation in vivo. J Orthop Res. 2016;34(1): 58-64. [38] Atkins GJ, Haynes DR, Howie DW, et al. Role of polyethylene particles in peri-prosthetic osteolysis: a review. World J Orthop. 2011;2(10): 93-101.[39] Witte F, Hort N, Vogt C, et al. Degradable biomaterials based on magnesium corrosion. Curr Opin Solid State Mater Sci. 2008; 12(5-6): 63-72. [40] Feyerabend F, Fischer J, Holtz J, et al. Evaluation of short-term effects of rare earth and other elements used in magnesium alloys on primary cells and cell lines. Acta Biomater. 2010; 6(5): 1834-1842. [41] Pichler K, Kraus T, Martinelli E, et al. Cellular reactions to biodegradable magnesium alloys on human growth plate chondrocytes and osteoblasts. Int Orthop. 2014; 38(4): 881-889. [42] Posada OM, Tate RJ, Meek RM, et al. In vitro analyses of the toxicity, immunological, and gene expression effects of cobalt-chromium alloy wear debris and Co ions derived from metal-on-metal hip implants. Lubricants. 2015;(3): 539-568.[43] Tsaousi A, Jones E, Case CP. The in vitro genotoxicity of orthopaedic ceramic (Al2 O3) and metal (CoCr alloy) particles. Mutat Res. 2010;697(1-2):1-9. [44] O’Neill SC, Queally JM, Devitt BM, et al. The role of osteoblasts in peri-prosthetic osteolysis. Bone Joint J. 2013; 95(8): 1022-1026.[45] Shi Y, Xie Y, Pan H, et al. Plasma-sprayed titanium patterns for enhancing early cell responses. J Ther Spray Technol. 2016;25(5): 946-958. [46] Syggelos SA, Aletras AJ, Smirlaki I, et al. Extracellular matrix degradation and tissue remodeling in periprosthetic loosening and osteolysis: focus on matrix metalloproteinases, their endogenous tissue inhibitors, and the proteasome. Biomed Res Int. 2013;2013:230805.[47] Bonsignore LA, Anderson JR, Lee Z, et al. Adherent lipopolysaccharide inhibits the osseointegration of orthopedic implants by impairing osteoblast differentiation. Bone. 2013; 52(1): 93-101. [48] Nalepka JL, Lee MJ, Kraay MJ, et al. Lipopolysaccharide found in aseptic loosening of patients with inflammatory arthritis. Clin Orthop Relat Res. 2006;451: 229-235. [49] Morra M, Cassinelli C, Bollati D, et al. Adherent endotoxin on dental implant surfaces: a reappraisal. J Oral Implantol. 2015;41(1): 10-16. [50] Bi Y, Seabold JM, Kaar SG, et al. Adherent endotoxin on orthopedic wear particles stimulates cytokine production and osteoclast differentiation. J Bone Miner Res. 2001; 16(11): 2082-2091. [51] Hirayama T, Tamaki Y, Takakubo Y, et al. Toll-like receptors and their adaptors are regulated in macrophages after phagocytosis of lipopolysaccharide-coated titanium particles. J Orthop Res. 2011;29(7): 984-992.[52] Sims NA, Martin TJ. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep. 2014;3:481.[53] Rajamannan NM, Cicek M, Hawse JR, et al. In vitro cell culture model of calcification: molecular regulation of myofibroblast differentiation to an osteoblast phenotype //Molecular biology of valvular heart disease. Springer London, 2014: 13-20. [54] Morra M, Cassinelli C, Bollati D, et al. Adherent endotoxin on dental implant surfaces: a reappraisal. J Oral Implantol. 2015;41(1): 10-16. [55] US Food and Drug Administration. Guideline on validation of the Limulus amebocyte lysate test as an end-product endotoxin test for human and animal parenteral drugs, biological products and medical devices 1987 [displayed 29 March 2010].[56] Sukur E, Akman YE, Ozturkmen Y, et al. Particle disease: a current review of the biological mechanisms in periprosthetic osteolysis after hip arthroplasty. Open Orthop J. 2016;10: 241. |
.jpg)
.jpg)