中国组织工程研究 ›› 2021, Vol. 25 ›› Issue (21): 3292-3299.doi: 10.3969/j.issn.2095-4344.3873
• 人工假体 artificial prosthesis • 上一篇 下一篇
假体无菌性松动过程中Co2+对成骨前体细胞的生物学反应
蒋昇源1,李 丹2,姜建浩1,杨上游3,4,杨淑野1
- 1滨州医学院附属医院创伤骨科,山东省滨州市 256600;2 巫山县人民医院,重庆市 404700;3 美国堪萨斯州堪萨斯大学威寄托骨科中心,堪萨斯州威寄托市,美国;4美国堪萨斯州威寄托州立大学生物系,堪萨斯州威寄托市,美国
-
收稿日期:2019-12-06修回日期:2019-12-10接受日期:2020-07-20出版日期:2021-07-28发布日期:2021-01-23 -
通讯作者:杨淑野,医学博士,滨州医学院附属医院创伤骨科,山东省滨州市 256600 杨上游,教授,美国堪萨斯州堪萨斯大学威寄托骨科中心,美国堪萨斯州威寄托州立大学生物系,堪萨斯州威寄托市,美国 -
作者简介:蒋昇源,男,1993年生,山东省龙口市人,汉族,滨州医学院在读硕士,主要从事创伤骨科研究。 李丹,女,1988年生,重庆市巫山县人,汉族,2014年中南大学毕业,主管护师,主要从事临床护理工作。 -
基金资助:山东省医药卫生科技发展计划项目(2016WS0023),项目负责人:杨淑野;滨州医学院科研计划(BY2016KYQD19),项目负责人:杨淑野
Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis
Jiang Shengyuan1, Li Dan2, Jiang Jianhao1, Shang-you Yang3, 4, Yang Shuye1
- 1Department of Traumatic Orthopedics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China; 2Chongqing Wushan County People’s Hospital, Chongqing 404700, China; 3Department of Orthopedic Surgery, the University of Kansas School of Medicine-Wichita, Wichita, KS, USA; 4Department of Biological Sciences, Wichita State University, Wichita, KS, USA
-
Received:2019-12-06Revised:2019-12-10Accepted:2020-07-20Online:2021-07-28Published:2021-01-23 -
Contact:Yang Shuye, MD, Department of Traumatic Orthopedics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China Shang-you Yang, Professor, Department of Orthopedic Surgery, the University of Kansas School of Medicine-Wichita, Wichita, KS, USA; Department of Biological Sciences, Wichita State University, Wichita, KS, USA -
About author:Jiang Shengyuan, Master candidate, Department of Traumatic Orthopedics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China Li Dan, Nurse-in-charge, Chongqing Wushan County People’s Hospital, Chongqing 404700, China Jiang Shengyuan and Li Dan contributed equally to this work. -
Supported by:the Medical and Health Technology Development Plan Project of Shandong Province, No. 2016WS0023 (to YSY); the Scientific Research Plan from the Binzhou Medical University, No. BY2016KYQD19 (to YSY)
摘要:
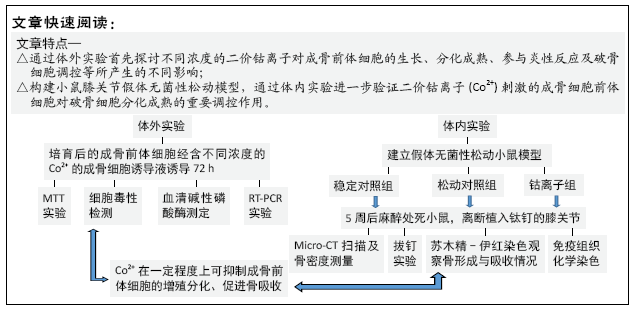
文题释义:
磨损颗粒:人工关节置换后假体关节的微动造成的摩擦可产生磨损颗粒,常见的磨损颗粒包括金属颗粒(钴铬钼、钛)、超高分子量聚乙烯颗粒(UHMWPE)、骨水泥颗粒(PMMA)及陶瓷颗粒等。
假体松动:包括假体感染性松动和无菌性松动。感染性松动是由于术中或术后感染所致,机制明确。无菌性松动机制较为复杂,目前认为是由于磨损颗粒引起的骨溶解、吸收导致假体周围骨性支撑力学结构性能降低所致。
背景:假体无菌性松动是人工关节置换后的主要并发症。目前金属离子已被证实参与人工假体无菌性松动的过程,如何控制和减缓假体松动成为当前研究的热点。
目的:观察在假体松动过程中,不同浓度的二价钴离子(Co2+)刺激成骨前体细胞的生物学反应。
方法:①体外实验:小鼠成骨前体细胞(MC3T3-E1)分别与含不同浓度Co2+的成骨细胞诱导液共同培养72 h,诱导为成骨细胞;应用MTT法检测细胞的增殖能力;通过测定乳酸脱氢酶活性来反映不同浓度Co2+的细胞毒性;测定血清碱性磷酸酶蛋白浓度来检测成骨前体细胞向成骨细胞转化的能力;应用RT-PCR测定相关因子mRNA表达;②体内实验:将小鼠分为3组,稳定对照组将钛钉置入小鼠的胫骨近端、松动对照组置入钛钉和钴铬颗粒、钴离子组置入钛钉和钴铬颗粒并注入经过钴离子刺激的成骨前体细胞,术后立即使用 MicroCT 进行假体周围骨密度测定,5周后再次行假体周围骨密度测定,麻醉处死小鼠并离断患侧膝关节进行拔钉实验,并将拔钉后的组织进行苏木精-伊红染色。通过拔钉力量来测定假体松动程度;通过假体与骨界面之间膜的厚度来反映炎症反应的严重程度;通过抗酒石酸酸性磷酸酶染色观察假体周围组织中破骨细胞数量。
结果与结论:①体外实验结果:当Co2+浓度升高时,成骨前体细胞的增殖会受到抑制;Co2+对成骨前体细胞表达血清碱性磷酸酶具有显著的抑制作用;Co2+可促进单核细胞趋化蛋白1、肿瘤坏死因子α、白细胞介素6、核因子κB 受体活化因子配基(RANKL)、活化T细胞核因子c1(NFATc1) mRNA表达,抑制骨保护素及成骨细胞特异性转录因子mRNA表达;低浓度Co2+(62 µmol/L)促进低密度脂蛋白受体相关蛋白(Lrp-5)和Runx2 mRNA表达,高浓度Co2+(500 µmol/L)抑制其表达;②体内实验结果:MicroCT 扫描显示钴离子组小鼠骨密度值最低(P ˂ 0.05);钴离子组中,拔钉实验所需要的剪切力较对照组明显降低(P ˂ 0.05);苏木精-伊红染色染色显示,钴离子组假体周围炎性反应膜的形成,Co2+刺激的成体前体细胞可能会加重假体周围炎症反应;③结果证实,成骨细胞在假体无菌性松动中可发挥重要作用,Co2+刺激的成骨细胞前体细胞对破骨细胞分化成熟的具有重要调控作用。
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
中图分类号:
引用本文
蒋昇源, 李 丹, 姜建浩, 杨上游, 杨淑野. 假体无菌性松动过程中Co2+对成骨前体细胞的生物学反应[J]. 中国组织工程研究, 2021, 25(21): 3292-3299.
Jiang Shengyuan, Li Dan, Jiang Jianhao, Shang-you Yang, Yang Shuye. Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3292-3299.
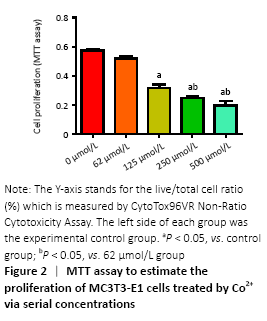
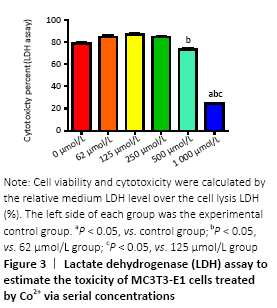
Cells were cultured with Co2+ of different concentrations for 72 hours. MTT cell proliferation assay showed that the experimental groups which included 125, 250, 500 μmol/L Co2+ inhibited the proliferation of MC3T3-E1 cells in varying degrees (P < 0.05; Figure 2). LDH assay showed significant toxicity when Co2+ reached 1 000 μmol/L and Co2+ showed slight toxicity at 500 μmol/L (P < 0.05; Figure 3).
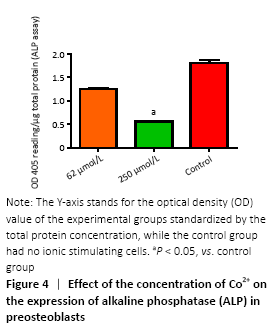
ALP activity (in vitro)
Figure 4 showed the different effects of two concentrations of Co2+ (62 μmol/L and 250 μmol/L) on ALP expression in MC3T3-E1 cells, respectively. When Co2+ was 250 μmol/L, it showed significant inhibition of ALP expression compared with the control group (P < 0.05).
Gene expression in preosteoblasts (in vitro)
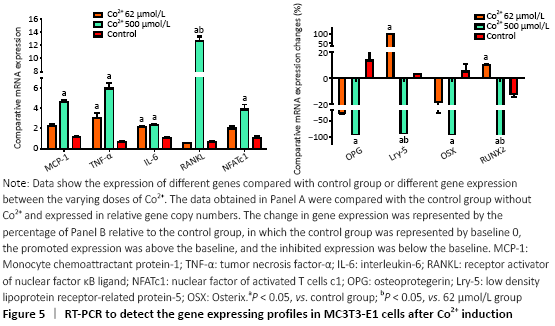
RT-PCR was used to detect the expression of a variety of genes to further investigate the molecular biological mechanisms underlying the effects of different concentrations of Co2+ (62 μmol/L and 500 μmol/L) on MC3T3-E1 cells. The data showed that Co2+ significantly promoted MCP-1 gene expression at 500 μmol/L compared with the control group (P < 0.05). Both groups significantly promoted the expression of TNF-α and IL-6, and they also promoted the expression of RANKL. When Co2+ was at 500 μmol/L, the expression of RANKL was significantly increased compared with that of 62 μmol/L group. The expression of NFATc1 stimulated by Co2+ was significantly enhanced at 500 μmol/L compared with the control group (P < 0.05). The expression levels of OPG and OSX were inhibited in both experimental groups. When Co2+ was at 500 μmol/L, there were significantly differences with the control group; thus, the RANKL/OPG ratio was much higher than the control value (P < 0.05). Same trend with Lrp-5 and Runx2, both of them promoted the expression of Co2+ at 62 μmol/L and inhibited the expression at 500 μmol/L compared with the control group (P < 0.05; Figure 5).
General situation and surgical results of experimental mice (in vivo)
The mice in each group grew and moved well, and generally responded well. No mice died in each group. The uneven distribution of cobalt and chromium particles around the prosthesis and the knee joint was observed after the knee joint was severed, and no significant anatomical changes were observed in the gross morphology of the knee joint (Figure 6).

MicroCT evaluation results (in vivo)
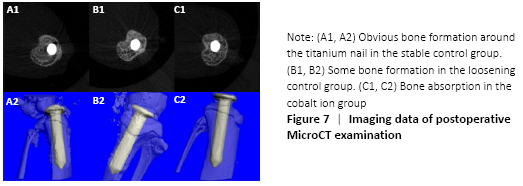
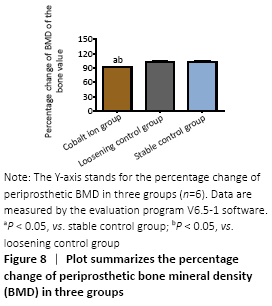
MicroCT scans showed that mice in the stable control group had good fixation and no displacement, bone ingrowth around the titanium nail could be seen obviously. There was visible bone resorption around the titanium nail in the cobalt ion group (Figure 7), and the percentage of changes of bone mineral density before and after treatment was significantly lower than that in the loose control group and the stable control group (P < 0.05; Figure 8).
Stability evaluation of titanium nail prosthesis-nail pulling test (in vivo)
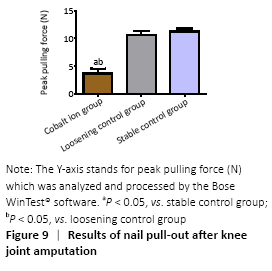
The blade holding the distal tibia of the mice which were fixed well by bone cement was able to withstand at least 100 Newtons (N) tension. In the stable control group, the maximum shear force needed at the moment of pullout was (11.58±2.47) N and (10.92±2.44) N in the loose control group, while in the cobalt ion group, the pullout force of the titanium nail was further reduced to (5.93±1.91) N. There were significant differences between the experimental group and the control group (P < 0.05; Figure 9).
Observation of histomorphology (in vivo)
Hematoxylin-eosin staining showed that there was obvious bone ingrowth and few inflammatory cells infiltration around the titanium nail in the stable control group. There was obvious inflammatory reaction membrane formation and a large number of inflammatory cell infiltration around the prosthesis in the cobalt ion group (Figure 10A-C). The comparison of periprosthetic membrane thickness between the above groups was calculated by computer image analysis software. The thickness of inflammatory reaction membrane around prosthesis in cobalt ion group was significantly thicker than that other two control groups (P < 0.05; Figure 10D). Differentiation and quantity of osteoclasts at the titanium nail prosthesis-bone interface were evaluated by TRAP staining, which showed that the degree of them in the surrounding tissues of the cobalt ion group was significantly higher than that of the control group (P < 0.05; Figure 11).
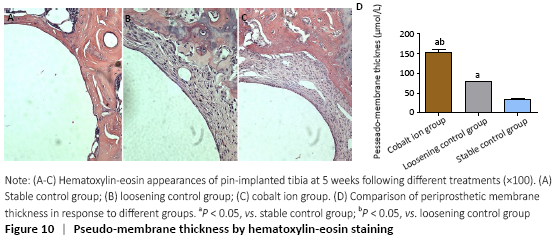

Immunohistochemical assessments (in vivo)
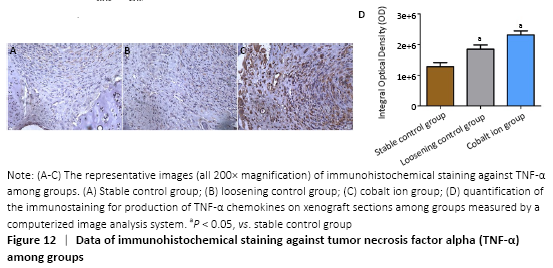
The expression of the positive-staining of TNF-α among groups is shown in the Figure 12A-C. There was almost no expression of TNF-α in the stable control group, but the cobalt ion group was the most obvious. All staining conditions were summarized, and it was concluded that the cobalt ion group and loosening control group were significantly higher than the stable control group in staining (P < 0.05; Figure 12D).
| [1] Holt G, Murnaghan C, Reilly J, et al. The biology of aseptic osteolysis. Clin Orthop Relate Res. 2007;460:240-252. [2] Pajarinen J, Lin TH, Nabeshima A, et al. Mesenchymal stem cells in the aseptic loosening of total joint replacements. J Biomed Mater Res Part A. 2017;105(4):1195-1207. [3] Inacio MC, Ake CF, Paxton EW, et al. Sex and risk of hip implant failure: assessing total hip arthroplasty outcomes in the United States. JAMA Intern Med. 2013;173(6):435-441. [4] Saeed K, Dryden M, Bassetti M, et al. Prosthetic joints : shining lights on challenging blind spots. Int J Antimicrob Agents. 2017;49(2): 153-161. [5] Jiang Y, Jia T, Gong W, et al. Effects of Ti, PMMA, UHMWPE, and Co-Cr wear particles on differentiation and functions of bone marrow stromal cells. J Biomed Mater Res Part A. 2013;101(10):2817-2825. [6] Yang SY, Zhang K, Bai L, et al. Polymethylmethacrylate and titanium alloy particles activate peripheral monocytes during periprosthetic inflammation and osteolysis. J Orthop Res. 2011;29(5):781-786. [7] Lochner K, Fritsche A, Jonitz A, et al. The potential role of human osteoblasts for periprosthetic osteolysis following exposure to wear particles. Int J Mol Med. 2011;28(6):1055-1063. [8] Chen W, Li Z, Guo Y, et al. Wear particles impair antimicrobial activity via suppression of reactive oxygen species generation and ERK1/2 phosphorylation in activated macrophages. Inflammation. 2015;38(3):1289-1296. [9] Jonitz-Heincke A, Lochner K, Schulze C, et al. Contribution of human osteoblasts and macrophages to bone matrix degradation and proinflammatory cytokine release after exposure to abrasive endoprosthetic wear particles. Mol Med Rep. 2016;14(2):1491-500. [10] Tereza D, Jiri G, Eva K. The role of the chemokine system in tissue response to prosthetic by-products leading to periprosthetic osteolysis and aseptic loosening. Front Immunol. 2017;8:1026. [11] Wang W, Ferguson DJP, Quinn JMW, et al. Biomaterial particle phagocytosis by bone-resorbing osteoclasts. Bone Joint J. 1997; 79-B(5):849-856. [12] Ollivere B, Wimhurst J A, Clark IM, et al. Current concepts in osteolysis. J Bone Joint Surg Br Vol. 2012;94(1):10-15. [13] Lass R, Alexander G, Kolb A, et al. Comparison of synovial fluid, urine, and serum ion levels in metal-on-metal total hip arthroplasty at a minimum follow-up of 18 years. J Orthop Res. 2014;32(9):1234-1240. [14] Lombardi JAV, Barrack RL, Berend KR, et al. The hip society: algorithmic approach to diagnosis and management of metal-on-metal arthroplasty. Bone Joint J. 2012;94-B(11 Supple A):14-18. [15] Keegan GM, Learmonth ID, Case CP. Orthopaedic metals and their potential toxicity in the arthroplasty patient: a review of current knowledge and future strategies. J Bone Joint Surg Br Vol. 2007;89(5): 567. [16] Hailer NP, Blaheta RA, Dahlstrand H, et al. Elevation of circulating HLA DR\r, +\r, CD8\r, +\r, T-cells and correlation with chromium and cobalt concentrations 6 years after metal-on-metal hip arthroplasty. Acta Orthop. 2011;82(1):6-12. [17] Caicedo MS, Desai R, Mcallister K, et al. Soluble and particulate Co-Cr-Mo alloy implant metals activate the inflammasome danger signaling pathway in human macrophages: a novel mechanism for implant debris reactivity. J Orthop Res. 2009;27(7):847-854. [18] Jian-Hao J, Peng L, Gang-Qiang D, et al. Cobalt-chromium particles inducing preosteoblasts may aggravate periprosthetic inflammation. Zhongguo Zuzhi Gongcheng Yanjiu. 2019;23(26):4101-4108. [19] Yang S, Zhang K, Li F, et al. Biological responses of preosteoblasts to particulate and ion forms of Co-Cr alloy. J Biomed Mater Res Part A. 2015;103(11):3564-3571. [20] Yang SY, Yu H, Gong W, et al. Murine model of prosthesis failure for the long-term study of aseptic loosening. J Orthop Res. 2007;25(5): 603-611. [21] Zhang L, Jia TH, Chong ACM, et al. Cell-based osteoprotegerin therapy for debris-induced aseptic prosthetic loosening on a murine model. Gene Ther. 2010;17(10):1262-1269. [22] Goodman SB, Gibon E, Pajarinen J, et al. Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J R Soc Interface. 2014;11(93):20130962. [23] Nakamura-Ota M, Hamanaka R, Yano H, et al. A new murine osteoblastic cell line immortalized with the SV40 large T antigen. Cell Tissue Banking. 2014;15(3):373-380. [24] Cadosch D, Gautschi OP, Chan E, et al. Titanium induced production of chemokines CCL17/TARC and CCL22/MDC in human osteoclasts and osteoblasts. J Biomed Mater Res Part A. 2010;92A(2): 475-483. [25] Chiu R, Smith KE, Ma GK, et al. Polymethylmethacrylate particles impair osteoprogenitor viability and expression of osteogenic transcription factors Runx2, osterix, and Dlx5. J Orthop Res. 2010; 28(5):571-577. [26] Nakamura Y, Hasebe A, Takahashi K, et al. Oligomerization-induced conformational change in the c-terminal region of nel-like molecule 1 (NELL1) protein is necessary for the efficient mediation of murine MC3T3-E1 cell adhesion and spreading. J Biol Chem. 2014; 289(14):9781-9794. [27] Hallab NJ, Anderson S, Caicedo M, et al. Effects of soluble metals on human peri-implant cells. 2005;74A(1):124-140. [28] Gallo J, Goodman SB, Konttinen YT, et al. Osteolysis around total knee arthroplasty: A review of pathogenetic mechanisms. Acta Biomater. 2013;9(9):8046-8058. [29] Haleem-Smith H, Argintar E, Bush C, et al. Biological responses of human mesenchymal stem cells to titanium wear debris particles. J Orthop Res. 2012;30(6):853-863. [30] Dalal A, Pawar V, Mcallister K, et al. Orthopedic implant cobalt-alloy particles produce greater toxicity and inflammatory cytokines than titanium alloy and zirconium alloy-based particles in vitro, in human osteoblasts, fibroblasts, and macrophages. J Biomed Mater Res Part A. 2012;100A(8):2147-2158. [31] Mandelin J, Li TF, Liljeström M, et al. Imbalance of RANKL/RANK/OPG system in interface tissue in loosening of total hip replacement. Bone Joint J. 2003;85(8):1196-1201. [32] Zijlstra WP, Bulstra SK, Raay J, et al. Cobalt and chromium ions reduce human osteoblast-like cell activity in vitro, reduce the OPG to RANKL ratio, and induce oxidative stress. J Orthop Res. 2012;30(5): 740-747. [33] Millett PJ, Allen MJ, Bostrom MPG. Effects of alendronate on particle-induced osteolysis in a rat model. J Bone Joint Surg. 2002; 84-A(2):236-249. [34] Ren WP, Yang SY, Wooley PH. A novel murine model of orthopaedic wear-debris associated osteolysis. Scand J Rheumatol. 2004;33(5): 349-357. |
| [1] | 张同同, 王中华, 文 杰, 宋玉鑫, 刘 林. 3D打印模型在颈椎肿瘤手术切除与重建中的应用[J]. 中国组织工程研究, 2021, 25(9): 1335-1339. |
| [2] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [3] | 徐东紫, 张 婷, 欧阳昭连. 心脏组织工程领域全球专利竞争态势分析[J]. 中国组织工程研究, 2021, 25(5): 807-812. |
| [4] | 吴子健, 胡昭端, 谢有琼, 王 峰, 李 佳, 李柏村, 蔡国伟, 彭 锐. 3D打印技术与骨组织工程研究文献计量及研究热点可视化分析[J]. 中国组织工程研究, 2021, 25(4): 564-569. |
| [5] | 常文辽, 赵 杰, 孙晓亮, 王 锟, 吴国锋, 周 剑, 李树祥, 孙 晗. 人工骨膜的材料选择、理论设计及生物仿生功能[J]. 中国组织工程研究, 2021, 25(4): 600-606. |
| [6] | 刘 旒, 周箐竹, 龚 桌, 刘博言, 杨 斌, 赵 娴. 胶原/无机材料构建组织工程骨的特点及制造技术[J]. 中国组织工程研究, 2021, 25(4): 607-613. |
| [7] | 刘 飞, 崔宇韬, 刘 贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究, 2021, 25(4): 614-620. |
| [8] | 李晓壮, 段 浩, 王伟舟, 唐志宏, 王旸昊, 何 飞. 骨组织工程材料治疗骨缺损疾病在体内实验中的应用[J]. 中国组织工程研究, 2021, 25(4): 626-631. |
| [9] | 张振坤, 李 喆, 李 亚, 王莹莹, 王亚苹, 周馨魁, 马珊珊, 关方霞. 海藻酸盐基水凝胶/敷料在创面愈合中的应用:持续、动态与顺序释放[J]. 中国组织工程研究, 2021, 25(4): 638-643. |
| [10] | 陈佳娜, 邱燕玲, 聂敏海, 刘旭倩. 组织工程支架材料修复口腔颌面部软组织缺损[J]. 中国组织工程研究, 2021, 25(4): 644-650. |
| [11] | 邢 浩, 张永红, 王 栋. 长骨大段骨缺损修复方法的优势与不足[J]. 中国组织工程研究, 2021, 25(3): 426-430. |
| [12] | 张立书, 刘安琪, 何小宁, 金 岩, 李 蓓, 金 钫. Alpl基因影响骨髓间充质干细胞治疗溃疡性结肠炎[J]. 中国组织工程研究, 2021, 25(25): 3970-3975. |
| [13] | 王秋霏, 顾 叶, 彭育沁, 薛 峰, 巨 荣, 朱 锋, 王熠军, 耿德春, 徐耀增. 人工假体磨损颗粒作用下Wnt/β-catenin信号通路对成骨细胞的影响[J]. 中国组织工程研究, 2021, 25(24): 3894-3901. |
| [14] | 杨彩会, 刘启成, 董 明, 王丽娜, 左美娜, 陆 颖, 牛卫东. 丝氨酸/苏氨酸蛋白激酶促进慢性根尖周炎模型小鼠的骨破坏[J]. 中国组织工程研究, 2021, 25(23): 3654-3659. |
| [15] | 王 皓, 陈明学, 李俊康, 罗旭江, 彭礼庆, 李 获, 黄 波, 田广招, 刘舒云, 眭 翔, 黄靖香, 郭全义, 鲁晓波. 脱细胞猪皮基质构建组织工程半月板支架[J]. 中国组织工程研究, 2021, 25(22): 3473-3478. |
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
Cytological experiment and randomized controlled animal experiment.
Time and settings
This study was completed at Clinical Laboratory, Binzhou Medical College in May, 2018.
Materials
MC3T3-E1 cells were purchased from ATCC (CRL-2594), and divalent cobalt ion (Co2+) was purchased from Sigma-Aldrich (Cat.#C8861). Eighteen female severe combined immunodeficient mice, aged 6 to 8 weeks, were provided by Shandong Experimental Animal Center, license No. SCXK (Lu) 20140007. Animal experiment was approved by the Ethics Committee of the Binzhou Medical University Hospital, approval number: 2018-003-01. Related information of cobalt ion is shown in Table 1.
Methods
Culture and induction of preosteoblasts (in vitro)
MC3T3-E1 cells (ATCC, Manassas, VA, USA) were cultured in Alpha Minimum Essential Medium at 37% and 5% CO2 atmosphere, which observed under a microscope, then the cells were fused and fell off to form cell suspension. After centrifugation and trypsin removal, the cells were subcultured in a ratio of 1∶3 and the third generation cells were taken for experiment. The MC3T3-E1 cells were cultured in Alpha Minimum Essential Medium containing 5% fetal bovine serum (FBS) (Invitrogen, Grand Island, NY, USA), 100 U/mL penicillin (Invitrogen), 100 mg/mL streptomycin (Invitrogen), 10 mmol/L β-glycerolphosphoric (Sigma-Aldrich, St. Louis, MO, USA), 50 μg/mL L-ascorbicacid (Sigma-Aldrich), 100 nmol/L dexamethasone (Sigma-Aldrich) and different concentrations of Co2+ (Sigma-Aldrich) for 3 days[18-19].
Determination of MTT and lactate dehydrogenase (LDH) (in vitro)
MC3T3-E1 cells were seeded in 96-well culture plates and per well contains 8×103 cells. Osteoblasts inducing fluid (Sigma-Aldrich) mixed with 0, 62, 125, 250, 500, and 1 000 μmol/L Co2+ were added respectively. After 72 hours of differentiation induction, 20 μL MTT solution was added to each well to culture for 4 hours. After the supernatant was removed, 150 μL dimethyl sulfoxide (Invitrogen) was added and the absorbance of each well at OD450nm was measured using a microplate reader. Cyto Tox 96®Non-Ratio Cytotoxicity Assay (Promega, Madison, WI, USA) was used to calculate the proportion of dead cells and viable cells. LDH assay was used to detect the toxicity of Co2+ at different concentrations to MC3T3-E1 cells.
Alkaline phosphatase (ALP) activity determination (in vitro)
The culture medium containing 200 μL preosteoblasts (MC3T3-E1) was inoculated into 48-well culture plate. After adding the osteogenic induction solution, the culture medium was co-cultured with two concentrations of Co2+ (62 μmol/L and 250 μmol/L) for 72 hours. The concentration of ALP protein in solution was determined by ALP Kit (Sigma-Aldrich) and the OD value was measured at 405 nm by spectrophotometer to reflect the content of ALP protein.
Real-time polymerase chain reaction (PCR) (in vitro)
RT-PCR was used to reveal the effects of different concentrations of Co2+ (62, 250 μmol/L) on the expression of monocyte chemoattractant protein-1 (MCP-1), TNF-α, IL-6, receptor activator of nuclear factor κB ligand (RANKL), osteoprotegerin (OPG), nuclear factor of activated T cells c1 (NFATc1), Runx2, osteoblast specific transcription factor Osterix (Osx) and low density lipoprotein receptor-related protein-5 (Lrp5) in MC3T3-E1 cells. Firstly, RNA was extracted from MC3T3-E1 cells which reacted with two different concentrations of Co2+ to determine the amount and concentration of RNA. After preparing the reverse transcription mixture, the reaction mixture was placed in Veriti 96-well Thermal Cycler (Applied Biosystems, Foster City, CA, USA) to prepare the cDNA. The detected genes were amplified by StepOnePlus® RT-PCR system (Applied Biosystems) and standardized with their respective housekeeping gene 18S.
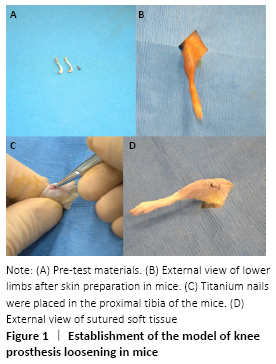
Establishment of a model of joint loosening in mice (in vivo)
Eighteen female severe combined immunodeficient mice, aged 6-8 weeks, were fed adaptively for one week. The mice were isolated for two weeks before the experiment. Subsequently, a model of aseptic loosening of mouse knee prosthesis was established (Pin-model) and the specific preparation method could be referred to the previous literature[20-21]. The standard model was that the titanium nail was completely implanted into the tibial medullary cavity of mice and the postoperative activity of the knee was not affected, which could be determined by postoperative X-ray examination and physiological activity of mice.
The Pin-model mice were randomly divided into three groups: (1) stable control group: titanium nail implantation (n=6); (2) loosening control group: titanium nail implantation+intra-articular injection of cobalt-chromium particles+MC3T3-E1 cells not stimulated by Co2+ (n=6); (3) cobalt ion group: titanium nail implantation+intra-articular injection of cobalt-chromium particles+MC3T3-E1 cells stimulated by Co2+ (n=6). MC3T3-E1 cells were induced by placing them in normal osteogenic induction fluid and osteogenic induction fluid containing 500 μmol/L Co2+, respectively. One week after operation, 10 μL suspension containing two kinds of MC3T3-E1 cells was injected into the joint cavity of the loosening control group and the cobalt ion group (Figure 1).
MicroCT bone scan (in vivo)
The proximal tibia of 18 mice were performing microCT (vivaCT 40, SCANCO Medical, Brüttisellen, Switzerland) scanning to ensure that the titanium nails were located in the bone marrow cavity of the tibia after the operation, and the location of the titanium nail and the bone mineral density value of the proximal tibia were recorded postoperatively and 5 weeks after surgery, respectively.
Pull-out test (in vivo)
Mice were executed and the knee joints were amputated at the prosthesis joint to remove the tibia with titanium nails five weeks after operation. The cartilage around the tibial plateau was scraped with the surgical blade to expose the tail cap of the titanium nail. Bose 3200 ElectroForce load frame (Bose Corporation, Eden Prairie, MN, USA) was used for nail pulling experiment. The output data are analyzed and processed by Bose WinTest® (Bose Corporation) software.
Histological morphological evaluation and image analysis (in vivo)
After the nail pulling experiment, tibial tissue around the titanium nail was collected and decalcified to make paraffin sections. Hematoxylin-eosin staining was used to observe the degree of inflammation, bone formation and bone resorption in the tissue around the titanium nail under optical microscope. The number of osteoclasts was observed by TRAP staining. Six regions were randomly selected in each section to determine the number of osteoclasts with positive staining, and the positive cells of TRAP staining were counted according to Image-pro software (Media Cybernetics, Silver Spring, MD, USA).
Immunohistochemical assessments (in vivo)
Immunohistochemical staining was performed to detect proinflammatory cytokines (TNF-α) within the implanted tissue. The mice were sacrificed 5 weeks after surgery and the mouse limb bone chips were harvested and fixed in 10% formalin as soon as taken out from mice. The specimens were decalcified in 12% EDTA for 10 days followed by histology process to prepare 6 µm unstained sections. Immunohistochemical staining kits, polyclonal antibodies against human and mouse TNF-α were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Digital images were taken under a microscope and then analyzed using image-pro PIus software (version 7.0, Media Cybernetics, Silver Spring, MD, USA). Each picture was randomly selected from six regions to be counted.
Statistical analysis
Data are expressed as the mean ± SEM. For statistical analysis, rank sum test was performed using the GraphPad Prism 5.0 software package. Significance was set at P < 0.05.
Preosteoblasts are the intermediate form of bone marrow mesenchymal stem cells differentiating into mature osteoblasts, which are abundant in bone marrow stroma and easily affected by wear particles in the process of transformation to osteoblasts. Previous studies have used MC3T3-E1 cells as preosteoblast models[25-26].
In vitro, to better study the biological effects of metal ions on the preosteoblasts, we established the optimal concentration of bivalent cobalt ions which is 62- 1 000 μmol/L for the first time[19, 27]. MTT cell proliferation assay after 72 hours of co-culture with different concentrations of Co2+ showed the degree of proliferation of MC3T3-E1 cells would be significantly inhibited compared with the control group When Co2+ ≥ 125 μmol/L. LDH which exists in almost all tissues and cells plays an important role in the process of energy metabolism and transformation, and cell damage caused by various factors can cause LDH overflow and increase the activity of it. Our experiment showed obvious toxicity when Co2+ at 1 000 μmol/L, and slight toxicity at 500 μmol/L. The above data showed that high concentration of bivalent cobalt ions in the humoral microenvironment produced a large number of toxic reactions to the preosteoblasts, inhibiting their proliferation and leading to their apoptosis. The inhibition of ALP expression which predicts the degree of maturation of preosteoblasts into osteoblasts indicates that the function of preosteoblasts is damaged. Our study suggests that the concentrations of Co2+ (250 μmol/L) can inhibit the expression of ALP compared with the control group. The above studies demonstrated that Co2+ can damage the function of preosteoblasts, osteoblasts and osteogenesis.
Chemokines play an important role in the process of inflammation.MCP-1 as a potent chemokine can promote the differentiation and activation of osteoclasts[28]. In our experiment, Co2+ of high concentration (500 μmol/L) can promote the expression of MCP-1, which indicates that Co2+ is easier to recruit monocytes/macrophages and osteoclasts. Previous studies have shown that pro-inflammatory cytokines such as TNF-α, IL-1 and IL-6 are usually secreted in wear particles-mediated osteolysis, which accelerates osteolytic effect of osteoclasts[29]. Dalal et al.[30] believe that cobalt-chromium particles of any type promote the release of inflammatory factors, such as TNF-α, IL-1β, IL-8 and IL-6 by osteoblasts, fibroblasts, and monocytes/macrophages. We studied the effects of Co2+ on TNF-α and IL-6 genes and the results showed that Co2+ at different concentrations promoted the release of inflammatory factors which was consistent with the IHC results in vivo. The system of RANKL/RANK/OPG is closely related to the differentiation of osteoclasts. Osteoblasts and bone marrow stromal cells express a certain amount of RANKL in physiological state and secrete a corresponding amount of OPG to maintain a balance between osteogenesis and osteolysis[31].
Our study showed that RANKL was significantly up-regulated, while OPG was significantly inhibited when MC3T3-E1 cells were co-cultured with Co2+, demonstrating that Co2+ could promote osteoclast formation and lead to osteolysis in vitro. The low RANKL/OPG ratio may confirm the chemical staining results of TRAP+ cells in the bivalent cobalt group and another function of preosteoblasts is to regulate osteoclast production by adjusting the ratio of RANKL/OPG[32], which can also provide a solution for the treatment of aseptic prosthesis loosening in the future. The effect of NFATc1 on osteoblasts is still controversial but our experiment showed that two different concentrations of Co2+ were likely to up-regulate the effect of NFATc1 on osteoblasts. Signaling proteins such as Runx2, Lrp5 and Osx also participate in the transformation of preosteoblasts into osteoblasts. In the current study, the expression of Osx gene was inhibited by different concentrations of Co2+ and the gene expression of Lrp5 and Runx2 was reduced by high concentrations of Co2+ (500 μmol/L). It further indicated that Co2+ could significantly damage the maturation and differentiation of osteoblasts from preosteoblasts, resulting in the reduction of the number of osteoblasts and the impairment of their functions. However, it should be noted that low concentrations of Co2+ (62 μmol/L) promoted the expression of Lrp5 and Runx2 genes, which need further study to explain. Through the RT-PCR data, the effects of bivalent cobalt ions on the differentiation, maturation and regulation of osteoclasts were further explained at the gene level, and the molecular biological basis of aseptic loosening of some artificial prostheses was revealed to a certain extent.
In order to study the mechanism and treatment of aseptic prosthesis loosening, many researchers have explored and established many animal models to simulate this pathological process[33-34]. However, these animal models are all short-term osteolysis models and lack the corresponding mechanical stress stimulation, so they cannot really simulate the biological process of prosthesis loosening. We constructed a long-term aseptic loosening animal model of joint prosthesis by implanting titanium nails in the proximal tibia of mice (Pin-model). Considering that wear particles are essential for aseptic prosthesis loosening, we injected cobalt and chromium particles suspension into the tibial bone marrow cavity of mice in the loosening control group and cobalt ion group before the implantation of titanium nails. MicroCT scan and three-dimensional reconstruction can compare the changes of the position of the titanium nail and the bone mineral density around the titanium nail after operation and 5 weeks after operation. The pull-out test can evaluate the strength of the prosthesis-bone interface and also reflect the stability of the implant. Our experimental results showed that the bone mineral density around the prosthesis in the cobalt ion group was significantly lower than that the control group, and the average force required for pulling out the nails was 5.93 N, which was also significantly lower than that the control group. Histological staining showed that a large number of inflammatory cells infiltrated around the prosthesis in the cobalt ion group, and the amount of mature osteoclasts was much higher than that of the control group. These vitro experiments have confirmed that Co2+ can inhibit the transformation of preosteoblasts to osteoblasts, affect bone formation and promote osteoclast formation leading to bone dissolution.
The deficiency existing in this experiment is mainly the experiment selects the cobalt ion concentration in the range of 62-1 000 μmol/L, we can just only estimate the ion concentration of instant around the prosthesis roughly, and whether other ions as chromium ion, titanium, molybdenum ions and aluminum ions also involved is not known, it will be our next research direction. Another limitation is that the body weight, life span and exercise intensity of mice are still quite different from that of humans, so this experimental animal model still cannot truly simulate the decades-long clinical prosthesis loosening process, which still needs to be further improved.
In this study, in vivo and in vitro experiments confirmed that preosteoblasts play an important role in aseptic loosening of prosthesis. Co2+ can inhibit the function of preosteoblasts to a certain extent, and promote the formation of osteoclasts and bone resorption by promoting the release of cytokines such as MCP-1, TNF, IL-6 and RANKL. In a word, Co2+ participates in the transformation of preosteoblasts and regulates their functions but the response of preosteoblasts to different concentrations of Co2+ during aseptic loosening of prosthesis may be different.
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程

文题释义:#br# 磨损颗粒:人工关节置换后假体关节的微动造成的摩擦可产生磨损颗粒,常见的磨损颗粒包括金属颗粒(钴铬钼、钛)、超高分子量聚乙烯颗粒(UHMWPE)、骨水泥颗粒(PMMA)及陶瓷颗粒等。#br# 假体松动:包括假体感染性松动和无菌性松动。感染性松动是由于术中或术后感染所致,机制明确。无菌性松动机制较为复杂,目前认为是由于磨损颗粒引起的骨溶解、吸收导致假体周围骨性支撑力学结构性能降低所致。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||