中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (26): 6744-6751.doi: 10.12307/2026.845
• 功能性生物材料Functional biomaterials • 上一篇 下一篇
不同3D打印同种异体骨和人工高分子复合多孔支架材料修复大鼠颅骨缺损的比较
刘伟伟1,苟元彬2,崔晓雪1,李 昕1,刘大卫1,史梦柔1,陈 斌1,李智峰2
- 1天津市医药科学研究所,天津市 300020;2天津中津生物发展有限公司,天津市 300380
Comparison of different 3D-printed allogeneic bone and artificial polymer composite porous scaffold materials for repairing cranial bone defects in rats
Liu Weiwei1, Gou Yuanbin2, Cui Xiaoxue1, Li Xin1, Liu Dawei1, Shi Mengrou1, Chen Bin1, Li Zhifeng2
- 1Tianjin Institute of Medical & Pharmaceutical Sciences, Tianjin 300020, China; 2Tianjin Zhongjin Biological Development Co., Ltd., Tianjin 300380, China
摘要:
文题释义:
同种异体骨:指经过脱细胞、脱脂和灭菌处理的异体来源骨基质,保留了天然骨的三维孔隙结构和胶原、生长因子等生物活性成分。同种异体骨的关键优势在于提供仿生微环境,促进细胞迁移和成骨分化。
3D打印技术:也称为增材制造,是通过逐层添加材料的方式来构建三维物体。与传统的减材制造方式不同,3D打印通过计算机辅助设计文件,将材料按需逐层堆积,从而形成一个完整的实体物品,这一过程不仅可以极大提高生产效率,还能显著降低生产成本,尤其在复杂零件和个性化定制方面具有独特优势。
背景:同种异体骨材料具有良好的骨缺损修复作用,但存在免疫排斥反应、价格昂贵及刚性较低等不足。
目的:评价3种3D打印同种异体骨与人工高分子复合多孔支架材料的骨缺损修复能力和组织反应及降解性能。
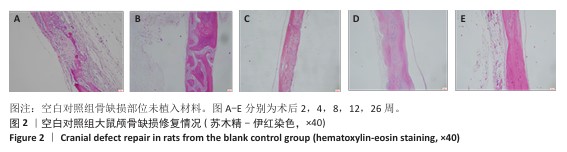
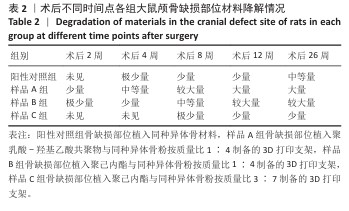
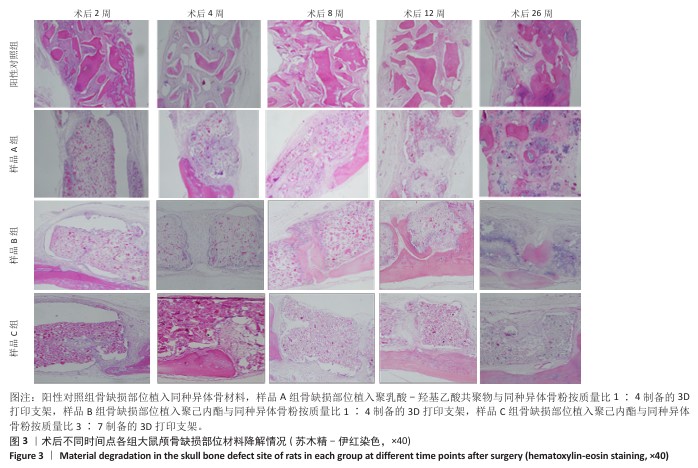
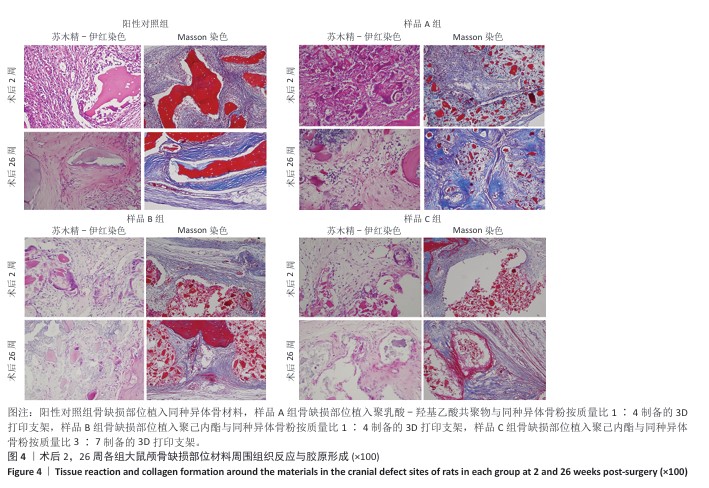
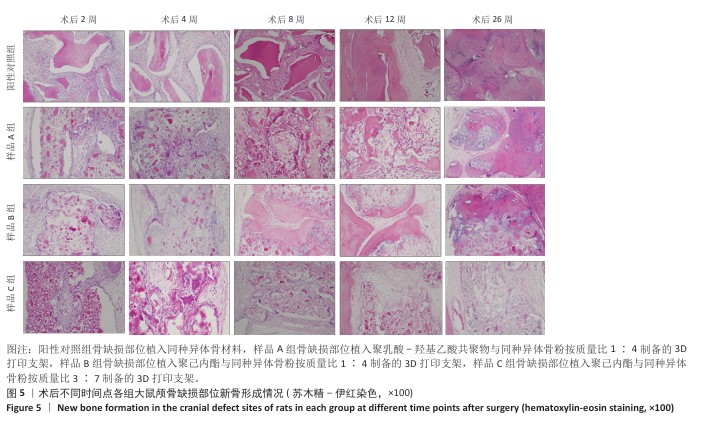
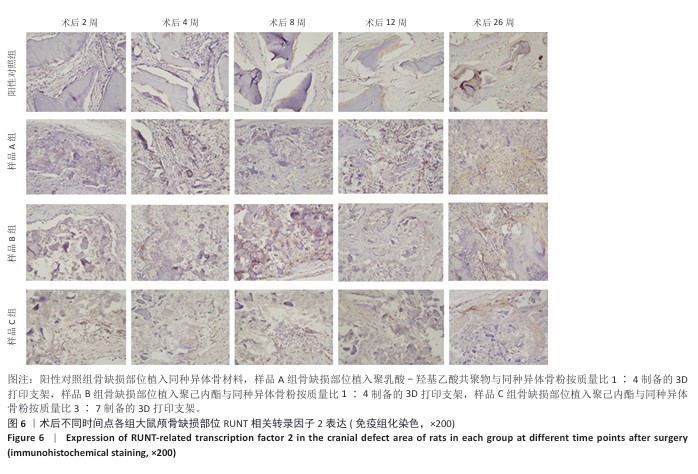
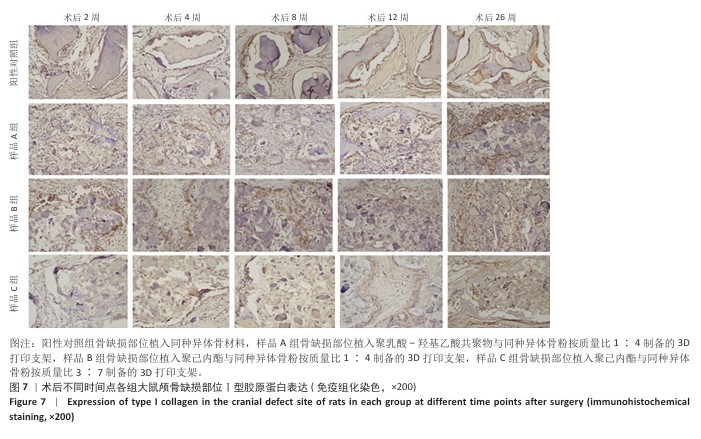
方法:制备3种骨修复材料:①样品A:将聚乳酸-羟基乙酸共聚物与同种异体骨粉按质量比1∶4 混合,基于低温沉积3D打印制备骨修复材料;②样品B:将聚己内酯与同种异体骨粉按质量比1∶4 混合,基于低温沉积3D打印制备骨修复材料;③样品C:将聚己内酯与同种异体骨粉按质量比3∶7混合,基于高温熔融3D打印制备骨修复材料。在35只SD大鼠矢状缝两侧各钻取1个直径为5 mm的圆形骨缺损,其中10个缺损处不进行任何干预(空白对照组),15个缺损部位植入同种异体骨材料(阳性对照组),15个缺损部位植入样品A(样品A组),15个缺损部位植入样品B(样品B组),15个缺损部位植入样品C(样品C组)。术后2,4,8,12,26周取材,苏木精-伊红染色观察材料降解、植入部位组织反应和新骨形成情况,Masson染色观察胶原纤维形成状况,免疫组化染色观察RUNT相关转录因子2和Ⅰ型胶原蛋白表达。
结果与结论:①苏木精-伊红染色:实验周期内,各植入材料均有不同程度降解,其中样品A和B降解较快,其次为阳性对照品,样品C降解较慢。随着时间的延长,各植入材料组的炎症反应均有减轻趋势,但纤维组织增生和新生血管仍较明显;术后26周,样品A组炎症反应减轻明显,其次为样品B组和阳性对照组,样品C组减轻趋势不明显。样品B组术后8周即可见少量新骨形成,样品A组和阳性对照组术后12周可见新骨形成,该3组术后26周可见大量新骨形成;样品C组始终未见明显新骨形成。②Masson染色:术后2周,各植入材料组可见少量排列较紊乱的胶原沉积;术后26周,各植入材料组可见大量排列较规则的胶原沉积。③免疫组化染色:随着时间的延长,各植入材料组RUNT相关转录因子2和Ⅰ型胶原蛋白表达增加,阳性对照组、样品A组、样品B组术后12,26周的RUNT相关转录因子2和Ⅰ型胶原蛋白表达高于样品C组。④结果表明:聚乳酸-羟基乙酸共聚物或聚己内酯与同种异体骨粉低温沉积3D打印的样品比高含量聚己内酯与同种异体骨粉高温熔融打印的样品降解快、炎症反应减轻、成骨因子表达及新骨形成更明显。
https://orcid.org/0009-0003-4777-3940(刘伟伟)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号: