[1] HAJARI VR, BENKE AP, GOEL EO. Advances in High-Frequency Surgical Device Design and Safety. Darpan International Research Analysis. 2024;12(3):269-282.
[2] VILOS GA, RAJAKUMAR C. Electrosurgical generators and monopolar and bipolar electrosurgery. J Minim Invasive Gynecol. 2013;20(3):279-287.
[3] BAI Y, YANG F, LIU C, et al. Expert consensus on the clinical application of high‐frequency electrosurgery in digestive endoscopy (2020, Shanghai). J Dig Dis. 2022;23(1):2-12.
[4] HAIPO C, JIAPING H, CHENGLI S, et al. Influence of different sizes on the temperature field of bipolar high-frequency electric knife. J Funct Mater Cailiao. 2018;49(9):9122-9126.
[5] DUNDEKOVÁ S, ŠKORÍK V, HADZIMA B, et al. The influence of the temperature on the corrosion resistance of aisi 316l stainless steel. ANNALS of Faculty Engineering Hunedoara-International Journal of Engineering. 2015;13(1):203-206.
[6] BARUA A, PRADHAN S, PRIYADARSHINI M, et al. Recent advancement in tungsten heavy alloy processing for different industrial applications. J Mater Eng Perform. 2024;34:8232-8252.
[7] RYU H, SEO MH, ROGERS JA. Bioresorbable metals for biomedical applications: from mechanical components to electronic devices. Adv Healthc Mater. 2021; 10(17):2002236.
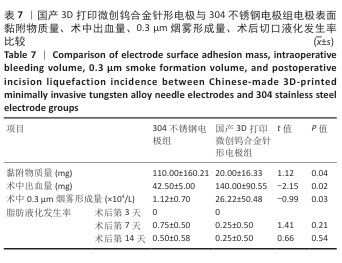
[8] 付和林,郝汝飞,郑靖,等.高频电刀电凝模式下304不锈钢和钨两种电极表面的组织粘附行为[J].中国表面工程,2016,29(3):96-102.
[9] WANG J, LIU Z, WU Y, et al. Cutting Performance and Tool Wear of AlCrN-and TiAlN-Coated Carbide Tools during Milling of Tantalum–Tungsten Alloy. Machines. 2024;12(3):170.
[10] 李顺,张周然,郑昆鹏,等.TiVTa 系低活化多主元合金的微观结构及相稳定性[J].材料工程,2023,51(8):181-189.
[11] WU M, DIAO G, XU Z, et al. Investigation of mechanical properties and wear resistance of A2/B2 type medium-entropy alloy matrix reinforced with tungsten particles by in-situ reaction. Metals. 2023;13(4):656.
[12] 王利剑,陈文革.钨铜合金表面粗糙度对抗电弧烧蚀性能的影响[J].粉末冶金材料科学与工程,2016,21(5):802-808.
[13] KURT MŞ, ARSLAN ME, YAZICI A, et al. Tribological, biocompatibility, and antibiofilm properties of tungsten–germanium coating using magnetron sputtering. J Mater Sci Mater Med. 2021;32(1):6.
[14] AHERWAR A, BAHRAMINASAB M. Biocompatibility evaluation and corrosion resistance of tungsten added Co-30Cr-4Mo-1Ni alloy. Biomed Mater Eng. 2017; 28(6):687-701.
[15] 蔡天智,陈婧婧.2015 年我国内窥镜贸易分析[J].中国医疗器械信息,2016, 22(6):21-25.
[16] 尤颢,杨谦.电外科手术设备的进展[J].中国医疗器械杂志,2012,36(4): 285-287.
[17] CHAROENKWAN K, CHOTIROSNIRAMIT N, RERKASEM K. Scalpel versus electrosurgery for abdominal incisions. Cochrane Database Syst Rev. 2017;6(6): CD005987.
[18] 杨勤玲,冯爱芳,鱼晓清.切口脂肪液化的诊治及护理研究综述[C]//第三届中华护理学会科技奖颁奖大会暨第三届护理学术年会论文集,2013:467-471.
[19] 宋静.手术切口脂肪液化换药方法的研究进展[J].全科护理,2016,14(27): 2838-2840.
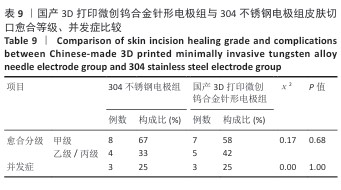
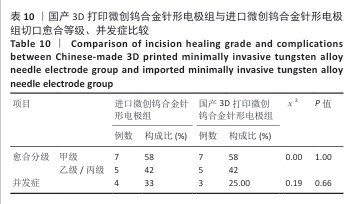
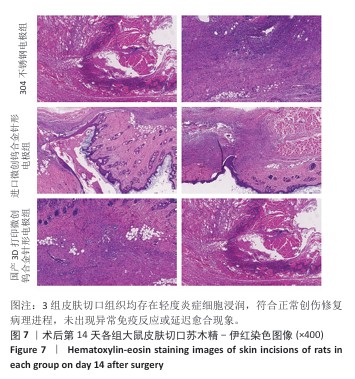
[20] 纪光伟,吴远志,王旭,等.高频电刀对腹部切口愈合影响的实验和临床研究[J].中华医学杂志,2002,82(17):1199-1202.
[21] 李巍,张艳秋.钨针电刀在耳内镜鼓膜修补手术中的应用[J].中华耳科学杂志,2021,19(2):248.
[22] MUKESHKUMAR MM. A comparative study of colorado micro dissection needle versus scalpel in maxillofacial surgery. Rajiv Gandhi University of Health Sciences (India), 2016.
[23] FURUDATE K, KATO J, HORIMOTO K, et al. Evaluation of degeneration of excision margin tissue using a Colorado MicroDissection Needle: A report of two cases. Australas J Dermatol. 2023;64(3):e301-e303.
[24] RIDEOUT B, SHAW GY. Tonsillectomy using the Colorado microdissection needle: a prospective series and comparative technique review. South Med J. 2004;97(1):11-17.
[25] ZHENG W, ZHU S, ZHANG Y, et al. Novel application of microdissection tungsten needle in total thyroidectomy with central neck dissection for papillary thyroid carcinoma. Front Surg. 2022;9:896275.
[26] 陈懿,田武国,张刚,等.微创钨针在开放性甲状腺手术中喉返神经和甲状旁腺保护的临床应用[J].中华内分泌外科杂志,2020,14(1):23-27.
[27] MISHRA P, BHOLA N, AGARWAL A, et al. To assess and compare the outcomes of the cutaneous neck dissection incision taken by colorado microdissection needle, surgical blade and cutting electrocautery in patients with oral squamous cell carcinoma: A prospective study. Natl J Maxillofac Surg. 2024;15(2):233-238.
[28] AL-ZYOUD W, HADDADIN D, HASAN SA, et al. Biocompatibility testing for implants: A novel tool for selection and characterization. Materials. 2023;16(21):6881.
[29] MIRI Z, FARÈ S, MA Q, et al. Updates on polyurethane and its multifunctional applications in biomedical engineering. Prog BiomedEng. 2023;5(4):042001.
[30] XING M. Compatibility of Composite Biomaterials in Sports Injury Repair. Adv Mater Sci Eng. 2021;2021(1):4954325.
[31] PRUKSAPONG C, JANKAJORN S, BURUSAPAT C, et al. Comparison of Colorado Needle Electrocautery and Traditional Scalpel for Lower Eyelid Blepharoplasty Incision: A Randomized Controlled Trial. Plast Reconstr Surg Glob Open. 2024; 12(11):e6325.
|