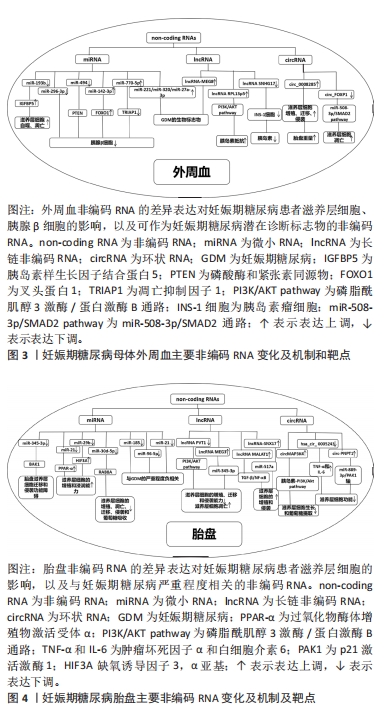
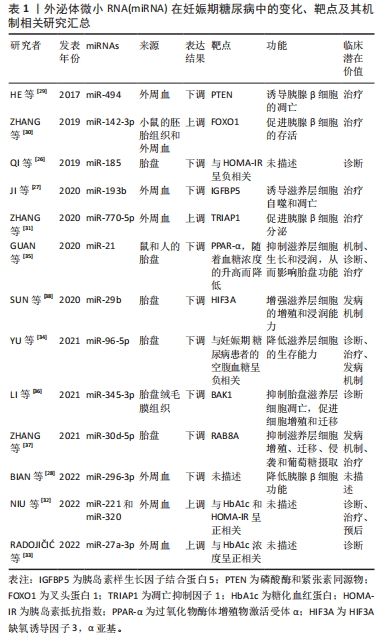
[1] MOON JH, JANG HC. Gestational diabetes mellitus: diagnostic approaches and maternal-offspring complications. Diabetes Metab J. 2022;46(1):3-14.
[2] MCINTYRE HD, CATALANO P, ZHANG C, et al. Gestational diabetes mellitus. Nat Rev Dis Primers. 2019;5(1):47.
[3] ZHAO C, DONG J, JIANG T, et al. Early second-trimester serum miRNA profiling predicts gestational diabetes mellitus. PLoS One. 2011;6(8):e23925.
[4] CHEN LL. The biogenesis and emerging roles of circular RNAs. Nat Rev Mol Cell Biol. 2016;17(4):205-211.
[5] XU YX, PU SD, LI X, et al. Exosomal ncRNAs: novel therapeutic target and biomarker for diabetic complications. Pharmacol Res. 2022;178:106135.
[6] ILJAS JD, GUANZON D, ELFEKY O, et al. Review: Bio-compartmentalization of microRNAs in exosomes during gestational diabetes mellitus. Placenta. 2017;54:76-82.
[7] GILLET V, OUELLET A, STEPANOV Y, et al. miRNA profiles in extracellular vesicles from serum early in pregnancies complicated by gestational diabetes mellitus. J Clin Endocrinol Metab. 2019;104(11):5157-5169.
[8] GAO C, SUN X, LU L, et al. Prevalence of gestational diabetes mellitus in mainland China: a systematic review and meta-analysis. J Diabetes Investig. 2019;10(1):154-162.
[9] VINCE K, PERKOVIC P, MATIJEVIC R. What is known and what remains unresolved regarding gestational diabetes mellitus (GDM). J Perinat Med. 2020;48(8):757-763.
[10] JUAN J, YANG H. Prevalence, prevention, and lifestyle intervention of gestational diabetes mellitus in China. Int J Environ Res Public Health. 2020;17(24):9517.
[11] DOYLE LM, WANG MZ. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells. 2019;8(7):727.
[12] WEI H, CHEN Q, LIN L, et al. Regulation of exosome production and cargo sorting. Int J Biol Sci. 2021;17(1):163-177.
[13] JADLI AS, BALLASY N, EDALAT P, et al. Inside(sight) of tiny communicator: exosome biogenesis, secretion, and uptake. Mol Cell Biochem. 2020;467(1-2):77-94.
[14] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science . 2020;367(6478):eaau6977.
[15] GURUNG S, PEROCHEAU D, TOURAMAINDOU L, et al. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal. 2021;19(1):47.
[16] PEGTEL DM, GOULD SJ. Exosomes. Annu Rev Biochem. 2019;88:487-514.
[17] SHAO J, ZARO J, SHEN Y. Advances in exosome-based drug delivery and tumor targeting: from tissue distribution to intracellular fate. Int J Nanomedicine. 2020;15:9355-9371.
[18] LI SP, LIN ZX, JIANG XY, et al. Exosomal cargo-loading and synthetic exosome-mimics as potential therapeutic tools. Acta Pharmacol Sin. 2018;39(4):542-551.
[19] YANG D, ZHANG W, ZHANG H, et al. Progress, opportunity, and perspective on exosome isolation - efforts for efficient exosome-based theranostics. Theranostics. 2020;10(8):3684-3707.
[20] SIMON C, GREENING DW, BOLUMAR D, et al. Extracellular vesicles in human reproduction in health and disease. Endocr Rev. 2018;39(3):292-332.
[21] GHAFOURIAN M, MAHDAVI R, AKBARI JONOUSH Z, et al. The implications of exosomes in pregnancy: emerging as new diagnostic markers and therapeutics targets. Cell Commun Signal. 2022;20(1):51.
[22] HERRERA-VAN OOSTDAM AS, SALGADO-BUSTAMANTE M, LOPEZ JA, et al. Placental exosomes viewed from an ‘omics’ perspective: implications for gestational diabetes biomarkers identification. Biomark Med. 2019;13(8):675-684.
[23] JIN J, MENON R. Placental exosomes: a proxy to understand pregnancy complications. Am J Reprod Immunol. 2018;79(5):e12788.
[24] CORREIA DE SOUSA M, GJORGJIEVA M, DOLICKA D, et al. Deciphering miRNAs’ Action through miRNA Editing. Int J Mol Sci. 2019;20(24):6249.
[25] LU TX, ROTHENBERG ME. MicroRNA. J Allergy Clin Immunol. 2018;141(4):1202-1207.
[26] QI S, WANG X. Decreased expression of miR-185 in serum and placenta of patients with gestational diabetes mellitus. Clin Lab. 2019. doi:10.7754/Clin.Lab. 2019.190445.
[27] JI Y, ZHANG W, YANG J, et al. MiR-193b inhibits autophagy and apoptosis by targeting IGFBP5 in high glucose-induced trophoblasts. Placenta. 2020;101:185-193.
[28] BIAN G, XUE Y, LIU Y, et al. Role of lncRNA-MEG8/miR-296-3p axis in gestational diabetes mellitus. Nephrology (Carlton). 2022;27(12):994-1002.
[29] HE Y, BAI J, LIU P, et al. miR-494 protects pancreatic β-cell function by targeting PTEN in gestational diabetes mellitus. EXCLI J. 2017;16:1297-307.
[30] ZHANG T, JI C, SHI R. miR-142-3p promotes pancreatic β cell survival through targeting FOXO1 in gestational diabetes mellitus. Int J Clin Exp Pathol. 2019;12(5):1529-1538.
[31] ZHANG YL, CHEN XQ. Dysregulation of microRNA-770-5p influences pancreatic-β-cell function by targeting TP53 regulated inhibitor of apoptosis 1 in gestational diabetes mellitus. Eur Rev Med Pharmacol Sci. 2020;24(2):793-801.
[32] NIU S, YU K, WANG W, et al. The expression and clinical value of miR-221 and miR-320 in the plasma of women with gestational diabetes mellitus. Clin Lab. 2022. doi:10.7754/Clin.Lab. 2021.210927.
[33] RADOJICIC O, DOBRIJEVIC Z, ROBAJAC D, et al. Gestational diabetes is associated with an increased expression of miR-27a in peripheral blood mononuclear cells. Mol Diagn Ther. 2022;26(4):421-435.
[34] YU X, LIU Z, FANG J, et al. miR-96-5p: a potential diagnostic marker for gestational diabetes mellitus. Medicine (Baltimore). 2021;100(21):e25808.
[35] GUAN CY, TIAN S, CAO JL, et al. Down-regulated miR-21 in gestational diabetes mellitus placenta induces PPAR-α to inhibit cell proliferation and infiltration. Diabetes Metab Syndr Obes. 2020;13:3009-3034.
[36] LI Y, ZHUANG J. miR-345-3p serves a protective role during gestational diabetes mellitus by targeting BAK1. Exp Ther Med. 2021;21(1):2.
[37] ZHANG L, LI K, TIAN S, et al. Down-regulation of microRNA-30d-5p is associated with gestational diabetes mellitus by targeting RAB8A. J Diabetes Complications. 2021;35(8):107959.
[38] SUN DG, TIAN S, ZHANG L, et al. The miRNA-29b is downregulated in placenta during gestational diabetes mellitus and may alter placenta development by regulating trophoblast migration and invasion through a HIF3A-dependent mechanism. Front Endocrinol (Lausanne). 2020;11:169.
[39] LI Y, CHENG X, LI D. LncRNA RPL13p5 gene expression promotes insulin resistance in patients with gestational diabetes. Ann Palliat Med. 2021;10(10):11024-11034.
[40] LI J, DU B, GENG X, et al. lncRNA SNHG17 is downregulated in gestational diabetes mellitus (GDM) and has predictive values. Diabetes Metab Syndr Obes. 2021;14:831-838.
[41] WANG Q, LU X, LI C, et al. Down-regulated long non-coding RNA PVT1 contributes to gestational diabetes mellitus and preeclampsia via regulation of human trophoblast cells. Biomed Pharmacother. 2019;120:109501.
[42] ZHANG H. Mechanism associated with aberrant lncRNA MEG3 expression in gestational diabetes mellitus. Exp Ther Med. 2019;18(5):3699-3706.
[43] GUIYU S, QUAN N, RUOCHEN W, et al. LncRNA-SNX17 promotes htr-8/svneo proliferation and invasion through miR-517a/IGF-1 in the placenta of diabetic macrosomia. Reprod Sci. 2022;29(2):596-605.
[44] ZHANG Y, QU L, NI H, et al. Expression and function of lncRNA MALAT1 in gestational diabetes mellitus. Adv Clin Exp Med. 2020;29(8):903-910.
[45] SHAFABAKHSH R, MIRHOSSEINI N, CHAICHIAN S, et al. Could circRNA be a new biomarker for pre-eclampsia? Mol Reprod Dev. 2019;86(12):1773-1780.
[46] MAHMOUDI E, CAIRNS MJ. CircRNA and ageing. Subcell Biochem. 2023;102:249-270.
[47] CHEN H, ZHANG S, WU Y, et al. The role of circular RNA circ_0008285 in gestational diabetes mellitus by regulating the biological functions of trophoblasts. Biol Res. 2021; 54(1):14.
[48] LI M, HUANG Y, XI H, et al. Circ_FOXP1 promotes the growth and survival of high glucose-treated human trophoblast cells through the regulation of miR-508-3p/SMAD family member 2 pathway. Endocr J. 2022;69(9):1067-1078.
[49] WANG H, SHE G, ZHOU W, et al. Expression profile of circular RNAs in placentas of women with gestational diabetes mellitus. Endocr J. 2019;66(5):431-441.
[50] YAN L, FENG J, CHENG F, et al. Circular RNA expression profiles in placental villi from women with gestational diabetes mellitus. Biochem Biophys Res Commun. 2018; 498(4):743-750.
[51] WANG H, ZHOU W, SHE G, et al. Downregulation of hsa_circ_0005243 induces trophoblast cell dysfunction and inflammation via the β-catenin and NF-κB pathways. Reprod Biol Endocrinol. 2020;18(1):51.
[52] CHEN X, HUANG J, PENG Y, et al. The role of circRNA polyribonucleotide nucleoside transferase 1 on gestational diabetes mellitus. Cell Mol Biol (Noisy-le-grand). 2022; 68(6):148-154.
[53] DU R, WU N, BAI Y, et al. circMAP3K4 regulates insulin resistance in trophoblast cells during gestational diabetes mellitus by modulating the miR-6795-5p/PTPN1 axis. J Transl Med. 2022;20(1):180.
[54] YE Z, WANG S, HUANG X, et al. Plasma exosomal miRNAs associated with metabolism as early predictor of gestational diabetes mellitus. Diabetes. 2022;71(11):2272-2283.
[55] FILARDI T, CATANZARO G, GRIECO GE, et al. Identification and validation of miR-222-3p and miR-409-3p as plasma biomarkers in gestational diabetes mellitus sharing validated target genes involved in metabolic homeostasis. Int J Mol Sci. 2022;23(8):4276.
[56] ZHANG L, ZHANG T, SUN D, et al. Diagnostic value of dysregulated microribonucleic acids in the placenta and circulating exosomes in gestational diabetes mellitus. J Diabetes Investig. 2021;12(8):1490-1500.
[57] ZHANG Q, YE X, XU X, et al. Placenta-derived exosomal miR-135a-5p promotes gestational diabetes mellitus pathogenesis by activating PI3K/AKT signalling pathway via SIRT1. J Cell Mol Med. 2023;27(23):3729-3743.
[58] ZHANG L, WU Q, ZHU S, et al. Chemerin-induced down-regulation of placenta-derived exosomal miR-140-3p and miR-574-3p promotes umbilical vein endothelial cells proliferation, migration, and tube formation in gestational diabetes mellitus. Cells. 2022;11(21):3457.
[59] HERRERA-VAN OOSTDAM AS, TORO-ORTIZ JC, LOPEZ JA, et al. Placental exosomes isolated from urine of patients with gestational diabetes exhibit a differential profile expression of microRNAs across gestation. Int J Mol Med. 2020;46(2):546-560.
[60] TANG GY, YU P, ZHANG C, et al. The neuropeptide-related HERC5/TAC1 interactions may be associated with the dysregulation of lncRNA GAS5 expression in gestational diabetes mellitus exosomes. Dis Markers. 2022;2022:8075285.
[61] JIANG B, ZHANG J, SUN X, et al. Circulating exosomal hsa_circRNA_0039480 is highly expressed in gestational diabetes mellitus and may be served as a biomarker for early diagnosis of GDM. J Transl Med. 2022;20(1):5.
[62] ZHANG W, ZHAO X, LI L. Downregulationof circ_0001578 promotes gestational diabetes mellitus by inducing placental inflammation via the NF-κB and JNKs pathways. Front Endocrinol (Lausanne). 2022;13:657802.
[63] ZHANG L, ZENG M, TANG F, et al. Circ-PNPT1 contributes to gestational diabetes mellitus (GDM) by regulating the function of trophoblast cells through miR-889-3p/PAK1 axis. Diabetol Metab Syndr. 2021;13(1):58.
[64] ZHU Y, TIAN F, LI H, et al. Profiling maternal plasma microRNA expression in early pregnancy to predict gestational diabetes mellitus. Int J Gynaecol Obstet. 2015;130(1): 49-53.
[65] CAO YL, JIA YJ, XING BH, et al. Plasma microRNA-16-5p, -17-5p and -20a-5p: novel diagnostic biomarkers for gestational diabetes mellitus. J Obstet Gynaecol Res. 2017; 43(6):974-981.
[66] YOFFE L, POLSKY A, GILAM A, et al. Early diagnosis of gestational diabetes mellitus using circulating microRNAs. Eur J Endocrinol. 2019;181(5):565-577.
[67] ZHANG W, CAO D, WANG Y, et al. LncRNA MEG8 is upregulated in gestational diabetes mellitus (GDM) and predicted kidney injury. J Diabetes Complications. 2021;35(1):107749.
[68] YANG H, YE W, CHEN R, et al. Circulating expression of Hsa_circRNA_102893 contributes to early gestational diabetes mellitus detection. Sci Rep. 2020;10(1):19046.
[69] WU H, WU S, ZHU Y, et al. Hsa_circRNA_0054633 is highly expressed in gestational diabetes mellitus and closely related to glycosylation index. Clin Epigenetics. 2019;11(1):22.
[70] CHEN X, BA Y, MA L, et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008;18(10):997-1006.
[71] SARKER S, SCHOLZ-ROMERO K, PERZE A, et al. Placenta-derived exosomes continuously increase in maternal circulation over the first trimester of pregnancy. J Transl Med. 2014;12:204.
[72] LV Q, DENG J, CHEN Y, et al. Engineered human adipose stem-cell-derived exosomes loaded with miR-21-5p to promote diabetic cutaneous wound healing. Mol Pharm. 2020;17(5):1723-1733.
[73] SHI R, ZHAO L, CAI W, et al. Maternal exosomes in diabetes contribute to the cardiac development deficiency. Biochem Biophys Res Commun. 2017;483(1):602-608.
[74] SU T, ZHANG P, ZHAO F, et al. Exosomal microRNAs mediating crosstalk between cancer cells with cancer-associated fibroblasts and tumor-associated macrophages in the tumor microenvironment. Front Oncol. 2021;11:631703. |