[1] VERZI MP, MCCULLEY DJ, DE VAL S, et al. The right ventricle, outflow tract, and ventricular septum comprise a restricted expression domain within the secondary/anterior heart field. Dev Biol. 2005;287(1):134-145.
[2] YAN L, XIE M, TAN B, et al. The effects of β-catenin on cardiomyogenesis via Islet-1 and MLIP ubiquitination. Exp Biol Med (Maywood). 2022; 247(21):1956-1967.
[3] CHRIST A, MARCZENKE M, WILLNOW TE. LRP2 controls sonic hedgehog-dependent differentiation of cardiac progenitor cells during outflow tract formation. Hum Mol Genet. 2020;29(19):3183-3196.
[4] ROCHAIS F, MESBAH K, KELLY RG. Signaling pathways controlling second heart field development. Circ Res. 2009;104(8):933-942.
[5] LI F, CHONG ZZ, MAIESE K. Winding through the WNT pathway during cellular development and demise. Histol Histopathol. 2006;21(1):103-124.
[6] COHEN ED, WANG Z, LEPORE JJ, et al. Wnt/beta-catenin signaling promotes expansion of Isl-1-positive cardiac progenitor cells through regulation of FGF signaling. J Clin Invest. 2007;117(7):1794-1804.
[7] JAIN R, LI D, GUPTA M, et al. HEART DEVELOPMENT. Integration of Bmp and Wnt signaling by Hopx specifies commitment of cardiomyoblasts. Science. 2015;348(6242):aaa6071.
[8] RIMKUS TK, CARPENTER RL, QASEM S, et al. Targeting the Sonic Hedgehog Signaling Pathway: Review of Smoothened and GLI Inhibitors. Cancers (Basel). 2016;8(2):22.
[9] GODDEERIS MM, SCHWARTZ R, KLINGENSMITH J, et al. Independent requirements for Hedgehog signaling by both the anterior heart field and neural crest cells for outflow tract development. Development. 2007;134(8):1593-1604.
[10] DYER LA, KIRBY ML. Sonic hedgehog maintains proliferation in secondary heart field progenitors and is required for normal arterial pole formation. Dev Biol. 2009;330(2):305-317.
[11] DELLOYE-BOURGEOIS C, RAMA N, BRITO J, et al. Sonic Hedgehog promotes the survival of neural crest cells by limiting apoptosis induced by the dependence receptor CDON during branchial arch development. Biochem Biophys Res Commun. 2014;452(3):655-660.
[12] ROWTON M, GUZZETTA A, RYDEEN AB, et al. Control of cardiomyocyte differentiation timing by intercellular signaling pathways. Semin Cell Dev Biol. 2021;118:94-106.
[13] 李慧超,师亮,杨艳萍,等.Sonic Hedgehog信号通路分子及胰岛素增强子结合蛋白在小鼠胚胎心流出道发育的早期表达[J].山西医科大学学报,2017,48(11):1087-1091.
[14] LIN L, CUI L, ZHOU W, et al. Beta-catenin directly regulates Islet1 expression in cardiovascular progenitors and is required for multiple aspects of cardiogenesis. Proc Natl Acad Sci U S A. 2007;104(22):9313-9318.
[15] WAXSE BJ, SENGUPTA P, HESKETH GG, et al. Myosin VI facilitates connexin 43 gap junction accretion. J Cell Sci. 2017;130(5):827-840.
[16] GHASEMI TAHRIR F, GUPTA M, MYERS V, et al. Role of Bcl2-associated Athanogene 3 in Turnover of Gap Junction Protein, Connexin 43, in Neonatal Cardiomyocytes. Sci Rep. 2019;9(1):7658.
[17] REAUME AG, DE SOUSA PA, KULKARNI S, et al. Cardiac malformation in neonatal mice lacking connexin43. Science. 1995;267(5205):1831-1834.
[18] KAPOOR N, GALANG G, MARBÁN E, et al. Transcriptional suppression of connexin43 by TBX18 undermines cell-cell electrical coupling in postnatal cardiomyocytes. J Biol Chem. 2011;286(16):14073-14079.
[19] CHAKRABORTY S, YUTZEY KE. Tbx20 regulation of cardiac cell proliferation and lineage specialization during embryonic and fetal development in vivo. Dev Biol. 2012;363(1):234-246.
[20] AI Z, FISCHER A, SPRAY DC, et al. Wnt-1 regulation of connexin43 in cardiac myocytes. J Clin Invest. 2000;105(2):161-171.
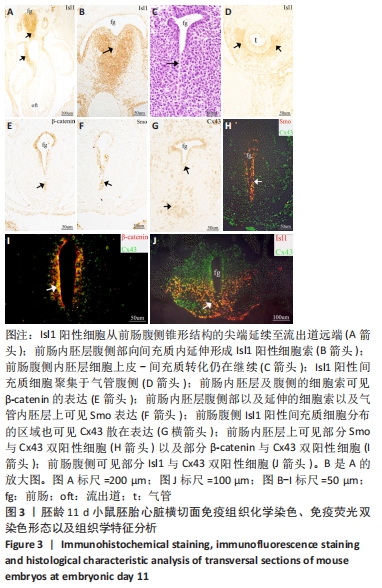
[21] 师亮,李慧超,景雅,等.鼠胚呼吸内胚层相关第二生心区发育中的上皮-间充质转化[J].解剖学报,2018,49(4):480-485.
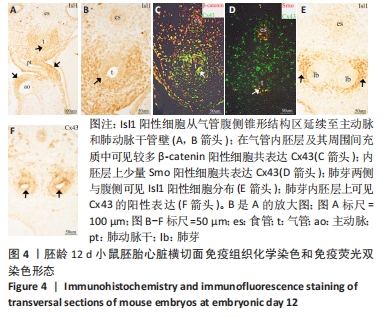
[22] 李行,景雅,李云华,等.Cx43敲除小鼠胚胎心脏流出道内第二生心区和心脏神经嵴来源间充质细胞减少[J].中国组织工程研究, 2021,25(13):2018-2024.
[23] YAMAGISHI H. Cardiac Neural Crest. Cold Spring Harb Perspect Biol. 2021;13(1):a036715.
[24] SHAO Q, ESSELTINE JL, HUANG T, et al. Connexin43 is Dispensable for Early Stage Human Mesenchymal Stem Cell Adipogenic Differentiation But is Protective against Cell Senescence. Biomolecules. 2019;9(9):474.
[25] SCHUSSLER O, GHARIBEH L, MOOTOOSAMY P, et al. Cardiac Neural Crest Cells: Their Rhombomeric Specification, Migration, and Association with Heart and Great Vessel Anomalies. Cell Mol Neurobiol. 2021;41(3):403-429.
[26] MIYAMOTO M, GANGRADE H, TAMPAKAKIS E. Understanding Heart Field Progenitor Cells for Modeling Congenital Heart Diseases. Curr Cardiol Rep. 2021;23(5):38.
[27] KLAUS A, SAGA Y, TAKETO MM, et al. Distinct roles of Wnt/beta-catenin and Bmp signaling during early cardiogenesis. Proc Natl Acad Sci U S A. 2007;104(47):18531-18536.
[28] UENO S, WEIDINGER G, OSUGI T, et al. Biphasic role for Wnt/beta-catenin signaling in cardiac specification in zebrafish and embryonic stem cells. Proc Natl Acad Sci U S A. 2007;104(23):9685-9690.
[29] LICKERT H, KUTSCH S, KANZLER B, et al. Formation of multiple hearts in mice following deletion of beta-catenin in the embryonic endoderm. Dev Cell. 2002;3(2):171-181.
[30] GODDEERIS MM, RHO S, PETIET A, et al. Intracardiac septation requires hedgehog-dependent cellular contributions from outside the heart. Development. 2008;135(10):1887-1895.
[31] BRITO JM, TEILLET MA, LE DOUARIN NM. An early role for sonic hedgehog from foregut endoderm in jaw development: ensuring neural crest cell survival. Proc Natl Acad Sci U S A. 2006;103(31):11607-11612.
[32] ZHANG C, LI Y, CAO J, et al. Hedgehog signalling controls sinoatrial node development and atrioventricular cushion formation. Open Biol. 2021;11(6):210020. |