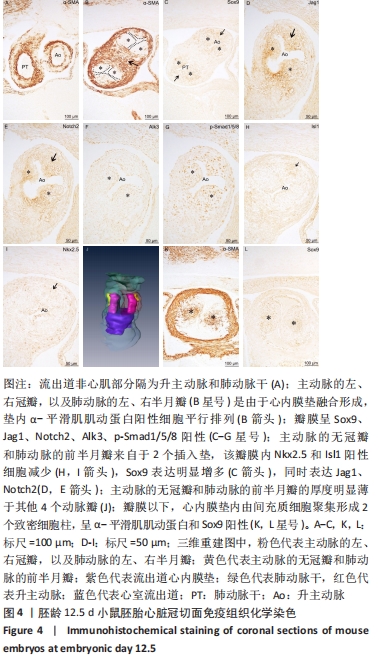
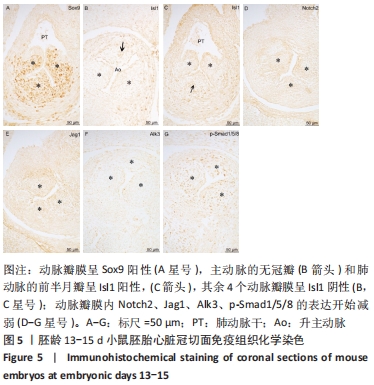
[1] BRAVO-JAIMES K, PRAKASH SK. Genetics in bicuspid aortic valve disease: Where are we? Prog Cardiovasc Dis. 2020;63(4):398-406.
[2] YANG YP, LI HR, CAO XM, et al. Septation of the Intrapericardial Arterial Trunks in the Early Human Embryonic Heart. Chin Med J (Engl). 2018; 131(12):1457-1464.
[3] YAMAGISHI T, NAREMATSU M, NAKAJIMA Y. Msx1 upregulates p27 expression to control cellular proliferation during valvuloseptal endocardial cushion formation in the chick embryonic heart. Anat Rec (Hoboken). 2021;304(8):1732-1744.
[4] CORTES C, FRANCOU A, DE BONO C, et al. Epithelial Properties of the Second Heart Field. Circ Res. 2018;122(1):142-154.
[5] SCHUSSLER O, GHARIBEH L, MOOTOOSAMY P, et al. Cardiac Neural Crest Cells: Their Rhombomeric Specification, Migration, and Association with Heart and Great Vessel Anomalies. Cell Mol Neurobiol. 2021;41(3):403-429.
[6] MIFFLIN JJ, DUPUIS LE, ALCALA NE, et al. Intercalated cushion cells within the cardiac outflow tract are derived from the myocardial troponin T type 2 (Tnnt2) Cre lineage. Dev Dyn. 2018;247(8):1005-1017.
[7] ODELIN G, FAURE E, COULPIER F, et al. Krox20 defines a subpopulation of cardiac neural crest cells contributing to arterial valves and bicuspid aortic valve. Development. 2018;145(1):dev151944.
[8] KURATANI SC, KIRBY ML. Initial migration and distribution of the cardiac neural crest in the avian embryo: an introduction to the concept of the circumpharyngeal crest. Am J Anat. 1991;191(3):215-227.
[9] ANDERSON R, CHAUDHRY B, MOHUN T, et al. Normal and abnormal development of the intrapericardial arterial trunks in humans and mice. Cardiovasc Res. 2012;95(1):108-115.
[10] YANG YP, LI HR, JING Y. Septation and shortening of outflow tract in embryonic mouse heart involve changes in cardiomyocyte phenotype and alpha-SMA positive cells in the endocardium. Chin Med J (Engl). 2004;117(8):1240-1245.
[11] RAMIREZ A, ASTROF S. Visualization and Analysis of Pharyngeal Arch Arteries using Whole-mount Immunohistochemistry and 3D Reconstruction. J Vis Exp. 2020;(157):10.3791/60797.
[12] ELEY L, ALQAHTANI AM, MACGROGAN D, et al. A novel source of arterial valve cells linked to bicuspid aortic valve without raphe in mice. Elife. 2018;7:e34110.
[13] ANDERSON RH, WEBB S, BROWN NA, et al. Development of the heart: (3) formation of the ventricular outflow tracts, arterial valves, and intrapericardial arterial trunks. Heart. 2003;89(9):1110-1118.
[14] SOTO-NAVARRETE MT, LÓPEZ-UNZU MÁ, DURÁN AC, et al. Embryonic development of bicuspid aortic valves. Prog Cardiovasc Dis. 2020; 63(4):407-418.
[15] FRANCOU A, SAINT-MICHEL E, MESBAH K, et al. Second heart field cardiac progenitor cells in the early mouse embryo. Biochim Biophys Acta. 2013;1833(4):795-798.
[16] DE BONO C, THELLIER C, BERTRAND N, et al. T-box genes and retinoic acid signaling regulate the segregation of arterial and venous pole progenitor cells in the murine second heart field. Hum Mol Genet. 2018;27(21):3747-3760.
[17] MILOS NC, NORDSTROM DB, ONGARO I, et al. Variations in structure of the outflow tract of the human embryonic heart: A new hypothesis for generating bicuspid aortic semilunar valves. Ann Anat. 2017;211:88-103.
[18] MACGROGAN D, LUNA-ZURITA L, DE LA POMPA JL. Notch signaling in cardiac valve development and disease. Birth Defects Res A Clin Mol Teratol. 2011;91(6):449-459.
[19] HARRISON OJ, VISAN AC, MOORJANI N, et al. Defective NOTCH signaling drives increased vascular smooth muscle cell apoptosis and contractile differentiation in bicuspid aortic valve aortopathy: A review of the evidence and future directions. Trends Cardiovasc Med. 2019;29(2):61-68.
[20] GARG V, MUTH AN, RANSOM JF, et al. Mutations in NOTCH1 cause aortic valve disease. Nature. 2005;437(7056):270-274.
[21] KAUFMAN MH. The Atlas of Mouse Developmet. US: academeic press. 1992:5-8.
[22] VARADKAR P, KRAMAN M, DESPRES D, et al. Notch2 is required for the proliferation of cardiac neural crest-derived smooth muscle cells. Dev Dyn. 2008;237(4):1144-1152.
[23] HIGH FA, JAIN R, STOLLER JZ, et al. Murine Jagged1/Notch signaling in the second heart field orchestrates Fgf8 expression and tissue-tissue interactions during outflow tract development. J Clin Invest. 2009; 119(7):1986-1996.
[24] LIN CJ, LIN CY, CHEN CH, et al. Partitioning the heart: mechanisms of cardiac septation and valve development. Development. 2012;139(18): 3277-3299.
[25] WANG W, HU YF, PANG M, et al. BMP and Notch Signaling Pathways differentially regulate Cardiomyocyte Proliferation during Ventricle Regeneration. Int J Biol Sci. 2021;17(9):2157-2166.
[26] ZHAO HJ, KLAUSEN C, LI Y, et al. Bone morphogenetic protein 2 promotes human trophoblast cell invasion by upregulating N-cadherin via non-canonical SMAD2/3 signaling. Cell Death Dis. 2018;9(2):174.
[27] THIERY JP, SLEEMAN JP. Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol. 2006;7(2):131-142.
[28] 杨艳萍, 赵瑞庆, 李海荣, 等. 小鼠胚胎心脏流出道嵴的发育 [J]. 解剖学报,2010,41(1):80-86.
[29] MACGROGAN D, D’AMATO G, TRAVISANO S, et al. Sequential Ligand-Dependent Notch Signaling Activation Regulates Valve Primordium Formation and Morphogenesis. Circ Res. 2016;118(10):1480-1497.
[30] JAIN R, ENGLEKA K, RENTSCHLER S, et al. Cardiac neural crest orchestrates remodeling and functional maturation of mouse semilunar valves. J Clin Invest. 2011;121(1):422-430.
|