中国组织工程研究 ›› 2024, Vol. 28 ›› Issue (2): 295-301.doi: 10.12307/2023.863
• 组织构建综述 tissue construction review • 上一篇 下一篇
Atg介导的自噬、运动和骨骼肌衰老
王京峰,文登台,王士杰,高颖晖
- 鲁东大学,体育学院,山东省烟台市 264025
-
收稿日期:2022-11-24接受日期:2023-01-04出版日期:2024-01-18发布日期:2023-06-30 -
通讯作者:文登台,博士,副教授,硕士研究生导师,鲁东大学,体育学院,山东省烟台市 264025 -
作者简介:王京峰,男,1997年生,山东省泰安市人,汉族,鲁东大学在读硕士研究生,主要从事运动人体科学研究。 -
基金资助:国家自然科学基金项目(32000832),项目负责人:文登台;山东省自然科学基金(ZR2020QC096),项目负责人:文登台
Atg-mediated autophagy, exercise and skeletal muscle aging
Wang Jingfeng, Wen Dengtai, Wang Shijie, Gao Yinghui
- College of Physical Education, Ludong University, Yantai 264025, Shandong Province, China
-
Received:2022-11-24Accepted:2023-01-04Online:2024-01-18Published:2023-06-30 -
Contact:Wen Dengtai, PhD, Associate professor, Master’s supervisor, College of Physical Education, Ludong University, Yantai 264025, Shandong Province, China -
About author:Wang Jingfeng, Master candidate, College of Physical Education, Ludong University, Yantai 264025, Shandong Province, China -
Supported by:National Natural Science Foundation of China, No. 32000832 (to WDT); Natural Science Foundation of Shandong Province, No. ZR2020QC096 (to WDT)
摘要:
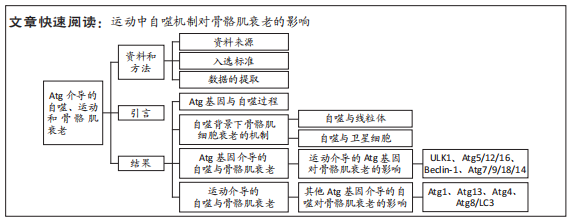
文题释义:
Atg基因(蛋白):Atg基因在进化过程中是高度保守的,从酵母和果蝇到脊椎动物和人类中都可以找到参与自噬的同源基因,Atg蛋白之间相互作用于自噬的连续过程中,参与自噬过程的调节。自噬:巨自噬,通常也称作自噬,是真核生物中进化保守的对细胞内物质进行周转的重要过程。该过程中一些损坏的蛋白或细胞器被双层膜结构的自噬小泡包裹后,送入溶酶体(动物)或液泡(酵母和植物)中进行降解并得以循环利用。营养不足的状态通常会激活自噬,但自噬也与许多生理和病理过程有关,比如发育、分化、神经退行性疾病、应激、感染以及癌症。
背景:运动作为一种可行的非药物治疗方法,有可能逆转随着年龄增长而恶化的骨骼肌衰老。自噬在骨骼肌衰老过程中的作用是不可缺少的。在骨骼肌衰老期间,参与调节自噬的Atg基因以或促进或抑制的方式调节自噬过程,以改善骨骼肌的生理形态。然而自噬在运动调节骨骼肌衰老中的具体分子机制仍令人困惑。
目的:通过对该领域文献的回顾,寻找运动中自噬机制对骨骼肌衰老影响的一般规律。方法:①文献资料法:通过对CNKI及Web of Science数据库有关“Atg基因(蛋白)、自噬、运动以及骨骼肌衰老”等相关文献的检索、查阅和筛选,为全文的分析奠定理论基础。②对比分析法:通过对所得到文献进行阅读分析,比较文献之间的异同点,为论点提供合理的理论支撑;通过对文献的进一步对比分析,理清相关指标间的关系,为全文的分析明确思路。
结果与结论:Atg家族介导的自噬对于延缓骨骼肌衰老是不可或缺的。参与调节自噬的Atg基因以或促进或抑制的方式调节自噬过程,以改善骨骼肌的生理形态及功能。不同的运动模式,如开始运动的年龄、时间或者强度,可能对自噬相关蛋白的表达有异质性的影响,但长期的有氧运动可以调节Atg相关蛋白,诱导骨骼肌自噬,并延缓肌肉质量的损失。
https://orcid.org/0000-0002-9581-6381(王京峰)
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
中图分类号:
引用本文
王京峰, 文登台, 王士杰, 高颖晖. Atg介导的自噬、运动和骨骼肌衰老[J]. 中国组织工程研究, 2024, 28(2): 295-301.
Wang Jingfeng, Wen Dengtai, Wang Shijie, Gao Yinghui. Atg-mediated autophagy, exercise and skeletal muscle aging[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 295-301.
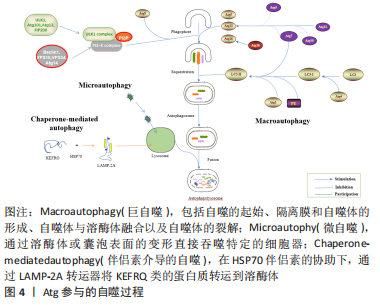
Atg12、微管相关蛋白1轻链3α/β(microtubule Associated protein 1 light chain 3 Alpha/Beta,MAP1LC3A/MAP1LC3B)和LC3是酵母Atg8的人类同源物[9-10],它在吞噬细胞膜的伸长和扩张中起着关键作用。Atg12与Atg5共轭,由Atg7和Atg10介导,然后与Atg16进行非共价作用[11-12]。Atg4的半胱氨酸肽酶在C端裂解LC3,产生LC3-Ⅰ,然后在一个类似泛素化的过程中与磷脂酰乙醇胺(phosphatidyl ethanolamine,PE)相连,期间需要Atg7和Atg3来产生LC3-Ⅱ[1,13]。吞噬细胞在成熟阶段的闭合导致了细胞质成分的分离和自噬囊泡的形成,随着成熟过程的推进,自噬囊泡与内体、囊泡(酵母和植物)或溶酶体(动物细胞)融合形成自噬泡,导致内体及其内容物的分解;随之而来的分解代谢产物,如氨基酸和脂肪酸,被送回细胞质并在细胞代谢中加以利用,提供内部能量生产的来源和分解代谢的初始成分[1,14-15]。
2.2 自噬背景下骨骼肌细胞衰老的机制 近些年来自噬在骨骼肌衰老中的作用备受人们关注(图5),了解在自噬背景下骨骼肌衰老的细胞机制对于明确自噬对于骨骼肌的调节作用尤为重要。
2.2.1 自噬与线粒体 线粒体功能失调被认为是导致生物衰老的主要因素之一,包括氧化能力下降和氧化损伤增加[16]。活性氧作为“氧化还原信使”,在正常生理环境下控制细胞内的信号传递,然而过多的活性氧会对细胞造成不可弥补的损害,并通过线粒体控制的内在凋亡途径导致细胞死亡。为了应对衰老过程中的氧化应激,自噬会消除受损的线粒体,并作为细胞清道夫和抗凋亡的防御措施[17]。线粒体生物生成和自噬之间存在着密切的关系。细胞的适应和抗压能力取决于这两个过程之间的平衡关系[18]。这种平衡的破坏可能是有害的,并导致线粒体损伤的积累,这可能加速人们增龄性的细胞凋亡[19]。线粒体功能障碍对骨骼肌衰老有着显著的影响[20]。最近的研究表明,由于线粒体自噬功能下降,衰老的骨骼肌会出现线粒体功能障碍的堆积[21-22]。保持适当的线粒体功能需要通过线粒体自噬来控制,此过程与通过线粒体融合以及裂解进行的形态调节同时发生[23]。裂解可能阻止健康细胞中线粒体的持续伸长,这可能会导致细胞衰老。自噬清除已受损的细胞器有助于延缓细胞衰老。在线粒体DNA异常患者的成肌纤维细胞中发现了对受损线粒体的选择性破坏(也被称为线粒体吞噬),并且在这些患者的细胞中,已经观察到了高于平均水平的自噬反应,这一点也证明了自噬在延缓骨骼肌衰老中的作用[24]。由上述结果不难看出,自噬与骨骼肌线粒体的功能结构有关,并随时影响骨骼肌衰老的走向。
2.2.2 自噬与卫星细胞 骨骼肌的再生依赖于卫星细胞(肌肉干细胞),尽管它们通常处于休眠状态[25]。随着年龄的增长,肌肉细胞的再生能力下降,卫星细胞停止休眠[26]。长时间的静止状态可以通过自噬来维持,并且干细胞需要自噬来消除静止阶段产生的细胞废物[27]。蛋白质和细胞器的周转是由基础自噬进行的,在静止的卫星细胞中是活跃的[26]。肌肉干细胞的再生速度随着人们的年龄增长而减慢[28],从正常静止状态转变为不可逆的衰老状态,这种减少随着年龄的增长体现的最为明显。生理衰老中卫星细胞自噬的异常或者年轻细胞自噬基因的损伤导致卫星细胞功能和数量的下降,通过蛋白质平衡的丧失、线粒体功能障碍和氧化压力的增加,最终导致衰老[29]。在针对于老年小鼠的研究中发现,基础自噬通量受损导致了受损蛋白质和亚细胞器(包括线粒体)的堆积[26],这些有害废物在卫星细胞中的堆积损害了它们的健康,使它们无法进入真正的静止状态。重组自噬功能可逆转衰老,并恢复老年卫星细胞的再生功能[29]。因此,自噬与肌肉再生有关,是决定肌肉干细胞的静止和衰老命运转变的关键因素之一[3]。
2.3 Atg基因介导的自噬与骨骼肌衰老 在进化保守的Atg基因的参与下,自噬是一个关键的分解代谢过程,影响骨骼肌的物质周转以及能量的产生和消耗。来自转基因动物模型的数据表明,运动产生的自噬对身体锻炼的适应是必要的[30]。由此,研究Atg基因如何在运动中影响骨骼肌意义深远。在此依据Atg基因(表1)的细胞成分将Atg基因分为4类阐述其介导的自噬在骨骼肌中的作用:Atg1/ULK1复合体、Atg5-12-16复合体、吞噬体组装位点、其他细胞成分。

2.3.1 运动介导的Atg基因对骨骼肌衰老的影响
(1)Atg1/ULK1复合物(ULK1):有研究结果表明,短期有氧运动通过人类骨骼肌ULK1刺激自噬信号,这表明自噬的开始是人类对运动的一个重要生理反应。运动是一种常见的AMP依赖的蛋白激酶(adenosine 5‘-monophosphate (AMP)-activated protein kinase,AMPK)激活剂[31-32],在已有的研究中已经证实了这一点。在生物体内进行的研究显示,在耐力运动后,肌肉自噬增加,而来自培养细胞的研究结果表明,AMPK有能力使ULK1 Ser555位点磷酸化[33]。AMPK-Thr172是人类骨骼肌中ULK1 Ser555位点的上游激酶,并且AMPK-Thr172的增加与ULK1 Ser555的磷酸化程度之间存在正相关关系,这进一步证明了运动诱导的人体骨骼肌自噬是由AMPK-ULK1信号通路介导的[34]。在对小鼠肌肉的研究中表明,胰岛素刺激和急性运动会减少肌肉自噬体的数量,而运动训练可能会增加自噬体的合成能力。此外,运动时激活AMPK似乎不足以控制肌肉中自噬体的数量,而胰岛素限制自噬的能力可能是通过哺乳动物中雷帕霉素靶蛋白C1(mammalian target of rapamycin C1, mTORC1)的靶点ULK1介导的[35-36]。此外,ULK1与骨骼肌如何适应运动训练的代谢有关。在比较ULK1和ULK2在运动训练后对于骨骼肌胰岛素的改善作用的实验中发现Ulk1是必要的,而不是ULK2 [37],这进一步证实ULK1在运动时骨骼肌中的调节作用。
(2)Atg5-12-16复合物(Atg5/12/16):传统自噬的关键过程包括LC3的脂化和ATG12-ATG5/ATG16L1复合物的形成。LC3脂化所必需的E3类连接酶活动不能由ATG12-ATG5/ATG16L1复合物的单个成分完成[38]。这种联合作用的模式在体育训练中得到了深刻的体现。例如,在为期8周的阻力训练后,蛋白Atg5、Atg12和Atg16表达的上升[39]。另一方面,Atg12和Atg16似乎受到低级炎症的影响。最近的研究发现,各种分解代谢条件下的肌肉萎缩与促炎症细胞因子及其受体Fn14有关。Fn14敲除小鼠与野生型小鼠相比,泛素化蛋白水平和Atg12的表达急剧减少[40]。在评估高强度训练对与炎症和自噬相关的特定基因表达变化的影响的研究中,同样发现了ATG12、ATG16L1表达的变化(增加)[41]。 尽管一些研究涉及到ATG12-ATG5 /ATG16L1在骨骼肌中的一些变化,但直接涉及ATG12-ATG5 /ATG16L1在骨骼肌中作用机制的研究很少,需要进一步的实验探究。
(3)吞噬体组装位点(Beclin-1、Atg7/9/18):一些研究结果表明,在从事长期有氧运动和阻力运动时,衰老的骨骼肌会表达更多的自噬相关蛋白。与自噬有关的蛋白质的表达因运动的种类和程度不同而有明显的差异[42]。在训练后,老龄小鼠的Beclin-1表达量提高,腓肠肌质量与体质量的比例明显增加(长期的) [43]。这些数据表明,自噬调节能力在老年骨骼肌中减弱。而且当动物接受运动训练时,这种效应被上调。此外,和肌纤维类型转换之间似乎有关联。来自营养匮乏的自噬因子1(nutrient-depleted autophagy factor-1,Naff-1)敲除小鼠的骨骼肌显示出向慢肌纤维(Ⅰ型)的明显转变以及对肌肉疲劳的更大抵抗,Naff-1基因敲除小鼠的肌肉发力能力明显降低[44],这一发现不仅证明了自噬调节因子Naff-1在维持骨骼肌平衡中的作用,而且为研究病态肌纤维的转化提供了新的研究视角。自噬的丧失可促进细胞核的迁移和聚集,抑制Atg9可抑制形态异常的肌纤维萎缩,同样沉默Atg9、Atg5和Atg18可促进肌纤维纵向的减少,也证明了这一点[45]。由于敲除Atg9和Atg18增加了肌纤维的纵向直径,由此更宽的肌肉纤维可能有助于在极性迁移过程中细胞核从内轴横向位移[45-46],这为自噬基因如何控制肌肉纤维的形状变化提供了一个新的视角。Atg7基因敲除小鼠表现出自噬异常,导致了小鼠氧化应激、线粒体功能失调以及细胞器和蛋白质的堆积[47-49],加速了小鼠的衰老。此外,在肌肉特异性Atg7基因敲除小鼠中观察到了异常的线粒体、混乱的肌浆、网状扩张以及不典型的同心膜结构。这些数据表明Atg7基因在维持肌肉质量和肌肉纤维完整性方面的作用。另外,LC3-Ⅱ、p62(自噬受体,也称为SQSTM1蛋白)、Atg5、Atg7和Beclin-1被证明在Rpt3(蛋白酶)缺陷小鼠的自噬途径中蛋白表达升高,这种上调可能是由于补偿性的蛋白质水解激活造成的[50]。
(4)其他细胞成分(Atg14):Atg14,又被称为Atg14L或Barkor(Beclin-1的一个重要调节器),参与了许多生物过程,包括发育、免疫、肿瘤抑制以及对一些心脏和神经系统疾病的防御[13,51]。由于Atg14 Ser29和ULK1磷酸化之间的正相关关系,ULK1 Ser555激活位点的磷酸化似乎代表了ULK1的激酶活性[52]。有研究表明,在合成代谢和分解代谢情况下,急性运动通过ULK1促进人体骨骼肌的自噬信号。从上述数据可以反映出骨骼肌中自噬信号的增加与Atg14的参与密不可分。缺少ULK1-ULK2、Atg14或Rb1cc1时,骨骼肌中的空泡会堆积[9]。研究人员发现,Atg14敲除小鼠和RB1的诱导卷曲蛋白RB1CC1抗体(rabbit Anti-RB1CC1/FIP200 antibody,Rb1cc1)敲除小鼠的骨骼肌都显示出泛素+SQSTM1+沉积的自噬空泡性肌病(autophagic vacuolar myopathy,AVM)特征,而且Rb1cc1敲除小鼠的骨骼肌还显示出TARDBP/TDP-43+(神经退行性疾病致病基因)病理和其他类似于ULK1/ULK2敲除小鼠描述的肌病的症状[7,53]。大多数糖酵解型的Ⅱ型肌纤维受到衰老(肌肉疏松症)、不运动和长期禁食条件(饥饿)带来的骨骼肌萎缩的影响,但氧化型的Ⅰ型肌纤维却能正常保存下来[54],这是由Ⅲ类磷脂酰肌醇激酶(vacuolar protein sorting 34,Vps34)蛋白水平的下降带来的,该蛋白不依赖于mTORC1,但依赖于STAT3(转录激活蛋白,STAT蛋白质家族成员),以及Vps34/p150/Beclin1/Atg14复合物1 [55],提示自噬基因Beclin1/Atg14在维持氧化型肌纤维中的重要意义[56]。
2.3.2 其他Atg基因介导的自噬与骨骼肌衰老
(1)Atg1/ULK1复合物(Atg1、Atg13):在关键组织中,Atg1的局部激活可以以非细胞自主的方式延缓衰老。Atg1的神经元上调对于诱导衰老过程中的组织间效应和延长寿命是必要的[57]。有研究表明甘氨酸-酪氨酸-苯丙氨酸 (Glycine-Tyrosine-Phenylalanine,Gyf)介导的自噬调节对于维持神经肌肉内稳态和预防组织退行性病变非常重要[58]。据报道,GIGYF2位点的人类突变与帕金森病相关,而Gyf又是GIGYF1(Grb10相互作用的Gyf蛋白1)和GIGYF2的同源基因。Gyf的沉默可以完全抑制Atg1-Atg13激活在刺激自噬通量中的作用,影响泛素化蛋白聚集物的清除。由此可以推测在帕金森病的形成过程中Atg1-Atg13复合物可能会出现缺陷。此外,有结果表明Pink1/Parkin促进衰老肌肉线粒体泛素化的自噬,并以Atg1依赖的方式延长寿命[59]。在间接飞行肌,线粒体泛素化以年龄依赖的方式增加,在间接飞行肌中过表达Pink1或Parkin可以消除线粒体泛素化(PINK1/Parkin介导的线粒体自噬在运动性骨骼肌损伤中的作用),恢复ATP水平和延长寿命,而通过Atg1敲除阻断自噬可以抑制衰老间接飞行肌中的这些作用[60]。令人惊讶的是Pink1和Parkin同样是两个与帕金森病相关的家族基因,这佐证了针对于Atg1缺陷时可能伴随着帕金森疾病产生的推测,为探索帕金森疾病的提供了一个新的研究方向。
ULK1复合体由ULK1(或ULK2)、FIP200、Atg13和Atg101组成,在自噬的初始阶段起作用。先前的研究表明ULK1和FIP200可以在自噬以外的通路中发挥作用[61]。然而,Atg13和Atg101的作用是否与ULK1和FIP200相似仍是未知的。有研究生成了Atg13敲除小鼠。结果发现Atg13缺陷小鼠在子宫内死亡,这与大多数其他类型的Atg缺陷小鼠不同,缺乏Atg13的胚胎表现为生长迟缓和心肌生长缺陷。在培养的成纤维细胞中,缺乏Atg13会阻碍自噬体在上游的形成。此外,Atg13缺失增强了对肿瘤坏死因子α诱导的细胞凋亡的敏感性,而其他Atg蛋白以及ULK1和ULK2的同时缺失则不起作用[61]。这些结果表明Atg13具有自噬和非自噬功能,后者对心脏发育至关重要,可能与FIP200共享,但与ULK1/2不相同。另外,有研究发现了在肌少症发生之前,线粒体质量控制和自噬通量的早期改变会使肌肉微环境恶化。而Atg13和LC3-Ⅱ升高以及P62和溶酶体相关膜蛋白1(lysosome-associated membrane protein 1,LAMP1,又称为CD107a)积累可能是造成自噬通量受损的直接原因[62]。还有就是Atg13在肌痛性脑脊髓炎患者中的表现,研究发现Atg13释放到血清后自噬受损可能是肌痛性脑脊髓炎的病理信号;Atg13在肌痛性脑脊髓炎患者的血清中被强烈上调,表明自噬代谢事件受损[63]。这些研究结果均可以反映出Atg13在自噬以及非自噬调节过程中的特异性,相信对其做进一步的研究将会是有趣的。
(2)其他细胞成分(Atg4、Atg8/LC3、Atg3):Atg4是一种高度特异性的蛋白酶,对自噬起着至关重要的作用[64]。在Atg4b-/-小鼠(自噬缺陷)模型中,首次描述了系统性自噬下降的代谢足迹。不同组织的代谢组中自噬缺乏的影响是高度依赖于组织的,在骨骼肌中比在心脏中更严重,而在肝组织中不明显,在这方面可以认为Atg4B缺乏可能以组织依赖的方式影响自噬[65]。
在酵母中,自噬启动、物质识别、废物吞噬以及囊泡闭合均依赖于Atg8。在高等真核生物中,Atg8已经进化为LC3/GABARAP蛋白家族(LC3A、LC3B、LC3C、GABARAP、GABARAPL1和GABARAPL2)[66]。
在诱导自噬体降解过程中,LC3-Ⅰ被脂化为LC3-Ⅱ,并与物质隔离膜结合,允许自噬体形成。LC3的脂化导致LC3-Ⅱ/LC3-Ⅰ比值增加,该比值是多种组织自噬的常用标记物,包括骨骼肌[67]。MAP1LC3是哺乳动物Atg8同源物之一,同样通过哺乳动物Atg7和Atg3催化的泛素样偶联以及涉及哺乳动物Atg4的脱溶反应进行调控。因此,脂质偶联的MAP1LC3 (MAP1LC3- Ⅱ)也通常被用作自噬结构的标记物[68]。
LC3B是研究最多的家族蛋白,与自噬体的发育和成熟有关,并被用于监测自噬活性[69]。在骨骼肌中,ULK1-ULK2、Atg14或Rb1cc1的缺失会导致囊泡的积累[70-71]。研究发现ULK1-ULK2、Atg14或Rb1cc1的缺失损害了自噬通量,但并不能完全消除Atg8家族成员的脂化以及LC3B+自噬体的生成(Atg5或Atg7的缺失也是如此),这表明LC3B+自噬体的存在(尽管水平降低)对肌肉空泡病理的发生是必需的[72]。当Atg3和Atg7处于非活性状态时,由于与其底物LC3的稳定共价作用而免受氧化侵蚀。当Atg3和Atg7被激活时,由于LC3转移到磷脂酰乙醇胺(脂化),这种相互作用变得短暂,而这一过程对功能性自噬至关重要[73]。在老年小鼠的骨骼肌中,LC3B-Ⅰ与磷脂酰乙醇胺的结合也下降,可能是由于Atg3和Atg12-Atg5蛋白水平下降所导致。在衰老过程中,巨噬细胞也有类似的下降,但对伴侣介导的自噬(chaperone-mediated autophagy,CMA)、线粒体周转以及肌肉与心脏的代谢功能失调有着不同的影响[74]。
很明显,Atg家族介导的自噬在骨骼肌衰老的过程中所扮演的角色不可或缺。并且有许多证据可以证明Atg家族基因的作用具有一定的联合性,大都不是一个Atg基因介导自噬过程,而是相互协调配合。在不同形式的运动干预下,Atg家族基因有着相应的表达,参与调节自噬过程的各个方面。
2.4 运动介导的自噬与骨骼肌衰老 虽然不同的运动模式,如开始运动的年龄、运动时间和运动强度,可能对自噬相关蛋白的表达有异质性的影响,但长期的有氧运动可以调节自噬相关蛋白,诱导骨骼肌自噬,并延缓肌肉质量的损失。虽然短期的低强度运动(干预期小于12周,频率小于每周3次)可能不会超过运动诱导自噬的阈值,但高强度运动有可能引起过度自噬[75-77]。人们发现,急性阻力运动可以激活Vps34,而自噬与运动后的蛋白质分解代谢有关[78-79]。然而在针对于人类的一项研究中发现,急性阻力运动减少了LC3的脂化(LC3-Ⅰ到LC3-Ⅱ的转化和骨骼肌中p62水平的降低),表明自噬可能在肌肉中被下调[80]。此类发现在骨骼肌的线粒体中也有体现,其中BNIP3(Bcl-2/E1B-19 kDa,
Bcl-2蛋白家族,线粒体促凋亡蛋白)表达增加,为自噬清除线粒体堆积废物提供了额外的证据[81-82]。此外,在中等强度耐力运动后的大鼠中发现了较高的Atg7、Beclin1和LC3蛋白水平,表明了适度的有氧运动增加了老年小鼠的自噬水平(Atg7和Beclin1)[83]。并且,有研究表明,9周的阻力运动训练促进了自噬,减少了老年大鼠骨骼肌的凋亡,避免了肌肉质量和力量的损失[84]。
体育锻炼可能会损害细胞结构,需要自噬机制的作用来清除和修复受损的细胞成分。有研究提取了股外侧肌肉(200 km跑)样本,发现了自噬蛋白Atg4、LC3和Atg12表达[85]。此外,还发现自噬蛋白的水平在进行超级马拉松之后表达更高[86]。虽然体育运动有益于身体健康,但高强度的运动,如超耐力运动,可能会使机体承受代谢压力进而损害线粒体的功能[6]。在这种情况下,自噬被激活,以消除受损的蛋白质和细胞器,并在能量不足时提供氨基酸[87-88]。从长远来看,精确控制运动量对于最大限度地发挥运动的益处至关重要。

| [1] SHIRAKABE A, IKEDA Y, SCIARRETTA S, et al. Aging and Autophagy in the Heart. Circ Res. 2016;118(10):1563-1576. [2] RUBINSZTEIN DC, MARINO G, KROEMER G. Autophagy and Aging. Cell. 2011;146:682-695. [3] XIA Q, HUANG X, HUANG J, et al. The Role of Autophagy in Skeletal Muscle Diseases. Front Physiol. 2021;12:638983. [4] RYTER SW, CHOI AM. Autophagy in lung disease pathogenesis and therapeutics. Redox Biol. 2015;4:215-225. [5] STJEPANOVIC G, BASKARAN S, LIN MG, et al. Vps34 Kinase Domain Dynamics Regulate the Autophagic PI 3-Kinase Complex. Mol Cell. 2017;67(3):528-534.e3. [6] DIKIC I, ELAZAR Z. Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol. 2018;19(6):349-364. [7] GALLUZZI L, BAEHRECKE EH, BALLABIO A, et al. Molecular definitions of autophagy and related processes. EMBO J. 2017;36(13):1811-1836. [8] LAMB CA, YOSHIMORI T, TOOZE SA. The autophagosome: origins unknown, biogenesis complex. Nat Rev Mol Cell Biol. 2013;14(12):759-774. [9] NAKATOGAWA H. Two ubiquitin-like conjugation systems that mediate membrane formation during autophagy. Essays Biochem. 2013;55:39-50. [10] ZHANG S, YAZAKI E, SAKAMOTO H, et al. Evolutionary diversification of the autophagy-related ubiquitin-like conjugation systems. Autophagy. 2022;18(12):2969-2984. [11] ROMANOV J, WALCZAK M, IBIRICU I, et al. Mechanism and functions of membrane binding by the Atg5-Atg12/Atg16 complex during autophagosome formation. EMBO J. 2012;31(22):4304-4317. [12] WALCZAK M, MARTENS S. Dissecting the role of the Atg12-Atg5-Atg16 complex during autophagosome formation. Autophagy. 2013;9(3):424-425. [13] ZHU M, ZHAN M, XI CJ, et al. Molecular characterization and expression of the autophagy-related gene Atg14 in WSSV-infected Procambarus clarkii. Fish Shellfish Immunol. 2022;125:200-211. [14] OHSUMI Y. Historical landmarks of autophagy research. Cell Res. 2014;24:9-23. [15] SAKAKIBARA K, EIYAMA A, SUZUKI SW, et al. Phospholipid methylation controls Atg32-mediated mitophagy and Atg8 recycling. EMBO J. 2015;34(21):2703-2719. [16] SON JM, LEE C. Mitochondria: multifaceted regulators of aging. BMB Rep. 2019;52(1): 13-23. [17] LYAMZAEV KG, TOKARCHUK AV, PANTELEEVA AA, et al. Induction of autophagy by depolarization of mitochondria. Autophagy. 2018;14(5):921-924. [18] PALIKARAS K, TAVERNARAKIS N. Mitochondrial homeostasis: the interplay between mitophagy and mitochondrial biogenesis. Exp Gerontol. 2014;56:182-188. [19] GONZALEZ-FREIRE M, DE CABO R, BERNIER M, et al. Reconsidering the Role of Mitochondria in Aging. J Gerontol A Biol Sci Med Sci. 2015;70(11):1334-1342. [20] 张雪,漆正堂,丁树哲.线粒体介导骨骼肌重塑在运动抵御癌症恶病质中的作用研究进展[J].上海体育学院学报,2021,45(1):78-85. [21] GOUSPILLOU G, SGARIOTO N, KAPCHINSKY S, et al. Increased sensitivity to mitochondrial permeability transition and myonuclear translocation of endonuclease G in atrophied muscle of physically active older humans. FASEB J. 2014;28(4):1621-1633. [22] O’LEARY MF, VAINSHTEIN A, IQBAL S, et al. Adaptive plasticity of autophagic proteins to denervation in aging skeletal muscle. Am J Physiol Cell Physiol. 2013;304(5):C422-430. [23] VAKIFAHMETOGLU-NORBERG H, OUCHIDA AT, NORBERG E. The role of mitochondria in metabolism and cell death. Biochem Biophys Res Commun. 2017;482(3):426-431. [24] LI P, MA Y, YU C, et al. Autophagy and Aging: Roles in Skeletal Muscle, Eye, Brain and Hepatic Tissue. Front Cell Dev Biol. 2021;9:752962. [25] 金晶,冯祎中,黄伟,等.运动通过激活卫星细胞功能延缓和改善骨骼肌衰减症的研究进展[J].体育科学,2019,39(8):73-80. [26] GARCIA-PRAT L, MUNOZ-CANOVES P, MARTINEZ-VICENTE M. Dysfunctional autophagy is a driver of muscle stem cell functional decline with aging. Autophagy. 2016;12:612-613. [27] Revuelta M, Matheu A. Autophagy in stem cell aging. Aging Cell. 2017;16:912-915. [28] 王震,蔺海旗,何霏,等.运动激活骨骼肌卫星细胞:增龄性肌衰减症及肌肉损伤修复的运动预防和治疗[J].中国组织工程研究,2021,25(23):3752-3759. [29] GARCÍA-PRAT L, MARTÍNEZ-VICENTE M, PERDIGUERO E, et al. Autophagy maintains stemness by preventing senescence. Nature. 2016;529(7584):37-42. [30] 钱帅伟,丁树哲.运动性细胞自噬是调节骨骼肌代谢稳态的内置机制[J].体育科学, 2015,35(10):55-65. [31] KJØBSTED R, MUNK-HANSEN N, BIRK JB, et al. Enhanced Muscle Insulin Sensitivity After Contraction/Exercise Is Mediated by AMPK. Diabetes. 2017;66(3):598-612. [32] 张坦,孙易,丁树哲.运动介导AMPK调控线粒体质量控制的机制研究进展[J].中国体育科技,2018,54(6):97-102 [33] KIM J, KUNDU M, VIOLLET B, et al. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol. 2011;13(2):132-141. [34] MøLLER AB, VENDELBO MH, CHRISTENSEN B, et al. Physical exercise increases autophagic signaling through ULK1 in human skeletal muscle. J Appl Physiol (1985). 2015;118(8):971-979. [35] FRITZEN AM, MADSEN AB, KLEINERT M, et al. Regulation of autophagy in human skeletal muscle: effects of exercise, exercise training and insulin stimulation. J Physiol. 2016;594(3):745-761. [36] 张鑫愉,周晓勐,牛燕媚,等.mTOR复合物在有氧运动改善小鼠骨骼肌糖代谢过程中的作用[J].中国运动医学杂志,2018,37(12):1017-1023. [37] DRAKE JC, WILSON RJ, CUI D, et al. Ulk1, Not Ulk2, Is Required for Exercise Training-Induced Improvement of Insulin Response in Skeletal Muscle. Front Physiol. 2021;12: 732308. [38] CHANGOTRA H, KAUR S, YADAV SS, et al. ATG5: A central autophagy regulator implicated in various human diseases. Cell Biochem Funct. 2022; 40(7):650-667. [39] MEJÍAS-PEÑA Y, ESTÉBANEZ B, RODRIGUEZ-MIGUELEZ P, et al. Impact of resistance training on the autophagy-inflammation-apoptosis crosstalk in elderly subjects. Aging (Albany NY). 2017;9(2):408-418. [40] TAJRISHI MM, SATO S, SHIN J, et al. The TWEAK-Fn14 dyad is involved in age-associated pathological changes in skeletal muscle. Biochem Biophys Res Commun. 2014;446(4):1219-1224. [41] KOSTRZEWA-NOWAK D, TRZECIAK-RYCZEK A, WITYK P, et al. Post-Effort Changes in Autophagy- and Inflammation-Related Gene Expression in White Blood Cells of Healthy Young Men. Cells. 2021;10(6):1406. [42] WANG C, LIANG J, REN Y, et al. A Preclinical Systematic Review of the Effects of Chronic Exercise on Autophagy-Related Proteins in Aging Skeletal Muscle. Front Physiol. 20224;13:930185. [43] KIM YA, KIM YS, OH SL, et al. Autophagic response to exercise training in skeletal muscle with age. J Physiol Biochem. 2013;69(4):697-705. [44] CHANG NC, NGUYEN M, BOURDON J, et al. Bcl-2-associated autophagy regulator Naf-1 required for maintenance of skeletal muscle. Hum Mol Genet. 2012;21(10):2277-2287. [45] ZHUANG X, CHUNG KP, CUI Y, et al. ATG9 regulates autophagosome progression from the endoplasmic reticulum in Arabidopsis. Proc Natl Acad Sci U S A. 2017;114(3):E426-E435. [46] KULEESHA, FENG L, WASSER M. Spatial pattern analysis of nuclear migration in remodelled muscles during Drosophila metamorphosis. BMC Bioinformatics. 2017; 18(1):329. [47] COSGROVE BD, GILBERT PM, PORPIGLIA E, et al. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat Med. 2014;20(3):255-264. [48] CHAKKALAKAL JV. JONES KM, BASSON MA, et al. The aged niche disrupts muscle stem cell quiescence. Nature. 2012;490(7420):355-360. [49] SOUSA-VICTOR P, GUTARRA S, GARCÍA-PRAT L, et al. Geriatric muscle stem cells switch reversible quiescence into senescence. Nature. 2014;506(7488):316-321. [50] KITAJIMA Y, SUZUKI N, YOSHIOKA K, et al. Inducible Rpt3, a Proteasome Component, Knockout in Adult Skeletal Muscle Results in Muscle Atrophy. Front Cell Dev Biol. 2020;8:859. [51] ZHANG H, GE S, NI B, et al. Augmenting ATG14 alleviates atherosclerosis and inhibits inflammation via promotion of autophagosome-lysosome fusion in macrophages. Autophagy. 2021;17(12):4218-4230. [52] PARK JM, SEO M, JUNG CH, et al. ULK1 phosphorylates Ser30 of BECN1 in association with ATG14 to stimulate autophagy induction. Autophagy. 2018;14(4):584-597. [53] LI D, VOGEL P, LI-HARMS X, et al. ATG14 and RB1CC1 play essential roles in maintaining muscle homeostasis. Autophagy. 2021;17(9):2576-2585. [54] NICHENKO AS, SORENSEN JR, SOUTHERN WM, et al. Lifelong Ulk1-Mediated Autophagy Deficiency in Muscle Induces Mitochondrial Dysfunction and Contractile Weakness. Int J Mol Sci. 2021;22(4):1937. [55] SANTOS-ARAUJO S, BOMFIM L, ARARIPE LO, et al. Silencing of ATG6 and ATG8 promotes increased levels of triacylglycerol (TAG) in the fat body during prolonged starvation periods in the Chagas disease vector Rhodnius prolixus. Insect Biochem Mol Biol. 2020; 127:103484. [56] YAMADA E, BASTIE CC, KOGA H, et al. Mouse skeletal muscle fiber-type-specific macroautophagy and muscle wasting are regulated by a Fyn/STAT3/Vps34 signaling pathway. Cell Rep. 2012;1(5):557-569. [57] ULGHERAIT M, RANA A, RERA M, et al. AMPK modulates tissue and organismal aging in a non-cell-autonomous manner. Cell Rep. 2014;8(6):1767-1780. [58] KIM M, SEMPLE I, KIM B, et al. Drosophila Gyf/GRB10 interacting GYF protein is an autophagy regulator that controls neuron and muscle homeostasis. Autophagy. 2015; 11(8):1358-1372. [59] SI H, MA P, LIANG Q, et al. Overexpression of pink1 or parkin in indirect flight muscles promotes mitochondrial proteostasis and extends lifespan in Drosophila melanogaster. Plos One. 2019;14(11):e0225214. [60] IMAI Y. PINK1-Parkin signaling in Parkinson’s disease: Lessons from Drosophila. Neurosci Res. 2020;159:40-46. [61] KAIZUKA T, MIZUSHIMA N. Atg13 Is Essential for Autophagy and Cardiac Development in Mice. Mol Cell Biol. 2015;36(4):585-595. [62] LIU HW, CHANG YC, CHAN YC, et al. Dysregulations of mitochondrial quality control and autophagic flux at an early age lead to progression of sarcopenia in SAMP8 mice. Biogerontology. 2020;21(3):367-380. [63] GOTTSCHALK G, PETERSON D, KNOX K, et al. Elevated ATG13 in serum of patients with ME/CFS stimulates oxidative stress response in microglial cells via activation of receptor for advanced glycation end products (RAGE). Mol Cell Neurosci. 2022;120:103731. [64] YANG M, CHANG Z, JI H. Characterization and expression analysis of ATG4 paralogs in response to the palmitic acid induced-ER stress in Ctenopharyngodon idellus kidney cells. Comp Biochem Physiol B Biochem Mol Biol. 2021;252:110525. [65] MARTINEZ-GARCIA GG, PEREZ RF, FERNANDEZ AF, et al. Autophagy Deficiency by Atg4B Loss Leads to Metabolomic Alterations in Mice. Metabolites. 2021;11(8):481. [66] SCHAAF MB, KEULERS TG, VOOIJS MA, et al. LC3/GABARAP family proteins: autophagy-(un)related functions. FASEB J. 2016;30(12):3961-3978. [67] FRITZEN AM, FRØSIG C, JEPPESEN J, et al.Role of AMPK in regulation of LC3 lipidation as a marker of autophagy in skeletal muscle. Cell Signal. 2016;28(6):663-674. [68] ZHU J, WANG KZ, CHU CT. After the banquet: mitochondrial biogenesis, mitophagy, and cell survival. Autophagy. 2013;9(11):1663-1676. [69] CERULLI RA, SHEHAJ L, BROWN H, et al. Stapled Peptide Inhibitors of Autophagy Adapter LC3B. Chembiochem. 2020;21(19):2777-2785. [70] NEMAZANYY I, BLAAUW B, PAOLINI C, et al. Defects of Vps15 in skeletal muscles lead to autophagic vacuolar myopathy and lysosomal disease. EMBO Mol Med. 2013;5(6):870-890. [71] WANG B, MAXWELL BA, JOO JH, et al.ULK1 and ULK2 Regulate Stress Granule Disassembly Through Phosphorylation and Activation of VCP/p97. Mol Cell. 2019;74(4):742-757.e8. [72] LI D, VOGEL P, LI-HARMS X, et al. ATG14 and RB1CC1 play essential roles in maintaining muscle homeostasis. Autophagy. 2021;17(9):2576-2585. [73] FRUDD K, BURGOYNE T, BURGOYNE JR. Oxidation of Atg3 and Atg7 mediates inhibition of autophagy. Nat Commun. 2018;9(1):95. [74] ZHOU J, CHONG SY, LIM A, et al. Changes in macroautophagy, chaperone-mediated autophagy, and mitochondrial metabolism in murine skeletal and cardiac muscle during aging. Aging-Us. 2017;9(2):583-599. [75] WANG C, LIANG J, REN Y, et al. A Preclinical Systematic Review of the Effects of Chronic Exercise on Autophagy-Related Proteins in Aging Skeletal Muscle. Front Physiol. 2022; 13:930185. [76] 崔新雯,张一民,汪赞,等.长期高强度间歇训练对中年大鼠骨骼肌蛋白合成和降解的影响[J].中国体育科技,2021,57(2):12-19. [77] 李方晖,艾竞一,李涛,等.高强度间歇训练对大鼠骨骼肌细胞自噬的影响及其调节机制[J].体育科学,2019,39(2):29-38. [78] GLYNN EL, FRY CS, DRUMMOND MJ, et al. Muscle protein breakdown has a minor role in the protein anabolic response to essential amino acid and carbohydrate intake following resistance exercise. Am J Physiol Regul Integr Comp Physiol. 2010;299(2):R533-540. [79] MACKENZIE MG, HAMILTON DL, MURRAY JT, et al. mVps34 is activated by an acute bout of resistance exercise. Biochem Soc Trans. 2007;35(Pt 5):1314-1316. [80] FRY CS, DRUMMOND MJ, GLYNN EL, et al. Skeletal muscle autophagy and protein breakdown following resistance exercise are similar in younger and older adults. J Gerontol A Biol Sci Med Sci. 2013;68(5):599-607. [81] LO VERSO F, CARNIO S, VAINSHTEIN A, et a. Autophagy is not required to sustain exercise and PRKAA1/AMPK activity but is important to prevent mitochondrial damage during physical activity. Autophagy. 2014;10(11): 1883-1894. [82] VAINSHTEIN A, TRYON LD, PAULY M, et al. Role of PGC-1α during acute exercise-induced autophagy and mitophagy in skeletal muscle. Am J Physiol Cell Physiol. 2015;308: C710-C719. [83] ROCCHI A, HE C. Activating Autophagy by Aerobic Exercise in Mice. J Vis Exp. 2017;(120):55099. [84] LUO L, LU AM, WANG Y, et al. Chronic resistance training activates autophagy and reduces apoptosis of muscle cells by modulating IGF-1 and its receptors, Akt/mTOR and Akt/FOXO3a signaling in aged rats. Exp Gerontol. 2013;48(4):427-436. [85] JAMART C, BENOIT N, RAYMACKERS JM, et al. Autophagy-related and autophagy-regulatory genes are induced in human muscle after ultraendurance exercise. Eur J Appl Physiol. 2012;112(8):3173-3177. [86] JAMART C, FRANCAUX M, MILLET GY, et al. Modulation of autophagy and ubiquitin-proteasome pathways during ultra-endurance running. J Appl Physiol (1985). 2012; 112(9):1529-1537. [87] SANDRI M. Protein breakdown in muscle wasting: role of autophagy-lysosome and ubiquitin-proteasome. Int J Biochem Cell Biol. 2013;45(10):2121-2129. [88] FERRARO E, GIAMMARIOLI AM, CHIANDOTTO S, et al. Exercise-induced skeletal muscle remodeling and metabolic adaptation: redox signaling and role of autophagy. Antioxid Redox Signal. 2014;21(1):154-176. |
| [1] | 谢 鹏, 张 江, 邓小磊, 魏 波, 侯德才. 肌少症小鼠模型构建的系统回顾[J]. 中国组织工程研究, 2024, 28(2): 263-266. |
| [2] | 郭项英, 彭子富, 何亦敏, 房洪波, 姜 宁. MiRNA-122在运动改善非酒精性脂肪肝中的作用[J]. 中国组织工程研究, 2024, 28(2): 272-279. |
| [3] | 常万鹏, 张钟文, 杨钰琳, 訾 阳, 杨梦琦, 杜冰玉, 王 楠, 于少泓. 康复外骨骼机器人对脑卒中下肢运动功能障碍疗效的Meta分析[J]. 中国组织工程研究, 2024, 28(2): 321-328. |
| [4] | 孙 园, 王庆博, 皮亦华, 陆春敏, 徐传仪, 张 艳. 早期和晚期有氧运动对野百合碱诱导肺动脉高压大鼠右心衰竭的影响[J]. 中国组织工程研究, 2024, 28(2): 177-185. |
| [5] | 尹林伟, 黄夏荣, 屈萌艰, 杨 璐, 王金玲, 贾飞阳, 廖 阳, 周 君. 跑台运动对老年大鼠骨质疏松及wnt/β-catenin信号通路的影响[J]. 中国组织工程研究, 2024, 28(2): 231-236. |
| [6] | 杨 裴. 不同运动方式对人体 DNA 损伤、DNA 甲基化和端粒长度的影响[J]. 中国组织工程研究, 2024, 28(1): 147-152. |
| [7] | 马岁录, 何志军, 刘 涛, 李 岩, 何元旭, 何 波, 王威威, 魏晓涛. 中药单体调控“细胞自噬”防治皮瓣坏死[J]. 中国组织工程研究, 2024, 28(1): 153-158. |
| [8] | 邓 锐, 黄科铭, 罗 建 , 陈 功, 冯 健 , 黄维义, 魏 刚. 血红素氧合酶1介导阿托伐他汀在巨噬细胞极化和胆固醇蓄积中的作用[J]. 中国组织工程研究, 2024, 28(1): 62-67. |
| [9] | 刘金玉, 张晗硕, 崔洪鹏, 潘灵之, 赵博然, 李 菲, 丁 宇. 脊柱微创治疗脊髓型颈椎病的有限元生物力学分析及精准化运动康复方案[J]. 中国组织工程研究, 2023, 27(9): 1359-1364. |
| [10] | 王 继, 张 敏, 杨中亚, 张 龙. 体力活动干预2型糖尿病肌少症的研究现状[J]. 中国组织工程研究, 2023, 27(8): 1272-1277. |
| [11] | 聂晨晨, 苏凯奇, 高 静, 凡勇福, 阮晓迪, 袁 洁, 段昭远, 冯晓东. 环状RNA调控脑缺血发病的作用与机制[J]. 中国组织工程研究, 2023, 27(8): 1286-1291. |
| [12] | 梁家琪, 刘恒旭, 阳金鑫, 杨 椅, 邓旭辉, 谭明健, 罗 炯. 运动与肠道菌健康效益的关系[J]. 中国组织工程研究, 2023, 27(8): 1292-1299. |
| [13] | 黄林科, 韦林华, 蒋 捷, 刘 倩, 陈蔚蔚. 雌激素与跑台运动干预卵巢切除模型小鼠骨量和关节软骨的变化[J]. 中国组织工程研究, 2023, 27(8): 1166-1171. |
| [14] | 毕耕超, 张彦龙, 李秋月, 胡龙威, 张 愉. 不同高度和间距下跳深动作中膝关节力学和周围肌肉的激活特征[J]. 中国组织工程研究, 2023, 27(8): 1211-1218. |
| [15] | 阮 凌, 王光华, 吴荣平, 晋 战, 吕镇庆, 张 楠, 李寿邦. 运动强度与高脂饮食模型大鼠脂代谢紊乱和氧化应激的相关性[J]. 中国组织工程研究, 2023, 27(8): 1149-1155. |
1.1.1 检索人及检索时间 第一作者在2021年11月进行检索。
1.1.2 检索文献时限 2010-01-01/2022-12-31。
1.1.3 检索数据库 Web of Science(WOS)数据库,https://www.webofscience.com;CNKI(中国知网)数据库,https://www.cnki.net/。
1.1.4 检索词 自噬与骨骼肌衰老,自噬与运动,运动与骨骼肌衰老,自噬、运动与骨骼肌衰老,Atg基因与骨骼肌衰老,Atg基因、运动和骨骼肌衰老,自噬与骨骼肌线粒体、自噬与骨骼肌卫星细胞;Autophagy and skeletal muscle aging,Autophagy and exercise,Exercise and skeletal muscle aging,Autophagy,exercise and skeletal muscle aging,Atg genes and skeletal muscle aging,Atg genes,exercise and skeletal muscle aging,Autophagy and skeletal muscle mitochondria,Autophagy and skeletal muscle satellite cells。
1.1.5 检索文献类型 研究原著,综述,述评,经验交流,病例报告,荟萃分析等。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以使用WOS数据库检索文献为例,在确定文章方向之后,围绕文章关键词“Atg基因、自噬、运动和骨骼肌衰老”进行关键词的组合搜索,例如输入核心词“Autophagy and skeletal muscle aging”若搜索文献量过少,则选择减少限制词aging再次搜索相关领域文献。在确定文献阅读范围之后,通过文献的阅读逐步排除非相关文献。在阅读期间可利用CitespaceV软件将所确定文献进行进一步的精简,更精准地把握相关文献与所撰写文章的契合度。具体操作步骤:在WOS数据库中输入关键词“Autophagy and skeletal muscle aging”搜索文献,将所有文献以纯文本形式,以及限定全记录和参考文献,下载并以download.txt的格式保存到已经建好的date文件夹中,利用CitespaceV软件将文献数据做可视化分析,锁定需要浏览的与文章相关的文献范围。图1,2。


1.2 入选标准
1.2.1 纳入标准 对应搜索关键词的所有文献。
1.2.2 排除标准 与核心关键词有明显出入的文献。
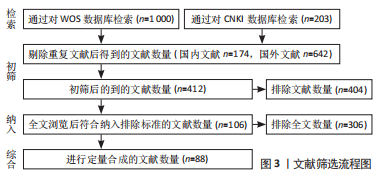
1.3 数据的提取 1 203篇剔除重复文献后得到816篇,其中国内文献174篇、国外文献642篇;阅读文题排除404篇,初筛得到412篇;浏览全文后排除306篇,复筛纳入106篇;最后进行综合分析,纳入88篇文献进行总结。文献筛选流程图,见图3。
 #br#
#br#
文题释义:
Atg基因(蛋白):Atg基因在进化过程中是高度保守的,从酵母和果蝇到脊椎动物和人类中都可以找到参与自噬的同源基因,Atg蛋白之间相互作用于自噬的连续过程中,参与自噬过程的调节。自噬:巨自噬,通常也称作自噬,是真核生物中进化保守的对细胞内物质进行周转的重要过程。该过程中一些损坏的蛋白或细胞器被双层膜结构的自噬小泡包裹后,送入溶酶体(动物)或液泡(酵母和植物)中进行降解并得以循环利用。营养不足的状态通常会激活自噬,但自噬也与许多生理和病理过程有关,比如发育、分化、神经退行性疾病、应激、感染以及癌症。
Atg家族介导的自噬对于延缓骨骼肌衰老是不可或缺的。参与调节自噬的Atg基因以或促进或抑制的方式调节自噬过程,以改善骨骼肌的生理形态及功能。目前仍有Atg家族的部分基因在骨骼肌中的作用还未得到探究,尤其是在运动干预之下,例如Atg2、Atg10、Atg17、Atg101等。运动作为一种可行的非药物治疗方法可以改善骨骼肌随年龄增长的恶化,但是一些特殊疾病除外,如糖尿病、癌症等。不同的运动模式,如开始运动的年龄、时间或者强度,可能对自噬相关蛋白的表达有异质性的影响,但长期的有氧运动可以调节Atg相关蛋白,诱导骨骼肌自噬,并延缓肌肉质量的损失。从长远来看,精确控制运动量对于最大限度地发挥运动的益处至关重要。然而运动引起的适宜自噬的阈值还有待实验的推敲,而对于Atg基因分子机制的实验探究可能会为该量度提供一个很好的方向。相信在不久的将来针对于Atg家族基因联合运动在骨骼肌衰老中的研究将会陆续的展开,Atg家族体系的作用将会不断完善,这将会是一个充满期待且有趣的过程。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||