[1] RAGHU G, COLLARD HR, EGAN JJ, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011;183(6):788-824.
[2] MARTINEZ FJ, COLLARD HR, PARDO A, et al. Idiopathic pulmonary fibrosis. Nat Rev Dis Primers. 2017;3:17074.
[3] DINH PC, PAUDEL D, BROCHU H, et al. Inhalation of lung spheroid cell secretome and exosomes promotes lung repair in pulmonary fibrosis. Nat Commun. 2020;11(1):1064.
[4] LI C, WANG Z, ZHANG J, et al. Crosstalk of mRNA, miRNA, lncRNA, and circRNA and their regulatory pattern in pulmonary fibrosis. Mol Ther Nucleic Acids. 2019;18:204-218.
[5] MEMCZAK S, JENS M, ELEFSINIOTI A, et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature. 2013;495(7441): 333-338.
[6] LU TX, ROTHENBERG ME. MicroRNA. J Allergy Clin Immunol. 2018; 141(4):1202-1207.
[7] NAGOSA S, LEESCH F, PUTIN D, et al. microRNA-184 induces a commitment switch to epidermal differentiation. Stem Cell Reports. 2017;9(6):1991-2004.
[8] XU J, BAI J, ZHANG X, et al. A comprehensive overview of lncRNA annotation resources. Brief Bioinform. 2017;18(2):236-249.
[9] KOPP F, MENDELL JT. Functional classification and experimental dissection of long noncoding RNAs. Cell. 2018;172(3):393-407.
[10] ZHANG W, TANG G, ZHOU S, et al. LncRNA-miRNA interaction prediction through sequence-derived linear neighborhood propagation method with information combination. BMC Genomics. 2019;20(Suppl 11):946.
[11] FU Z, CHEN C, ZHOU Q, et al. LncRNA HOTTIP modulates cancer stem cell properties in human pancreatic cancer by regulating HOXA9. Cancer Lett. 2017;410:68-81.
[12] LIU JQ, DENG M, XUE NN, et al. lncRNA KLF3-AS1 Suppresses Cell Migration and Invasion in ESCC by Impairing miR-185-5p-Targeted KLF3 Inhibition. Mol Ther Nucleic Acids. 2020;20:231-241.
[13] WANG Y, ZHU P, LUO J, et al. LncRNA HAND2-AS1 promotes liver cancer stem cell self-renewal via BMP signaling. EMBO J. 2019;38(17):e101110.
[14] CHEN LL. The expanding regulatory mechanisms and cellular functions of circular RNAs. Nat Rev Mol Cell Biol. 2020;21(8):475-490.
[15] YANG Y, FAN X, MAO M, et al. Extensive translation of circular RNAs driven by N6-methyladenosine. Cell Res. 2017;27(5):626-641.
[16] CHEN J, CHEN T, ZHU Y, et al. circPTN sponges miR-145-5p/miR-330-5p to promote proliferation and stemness in glioma. J Exp Clin Cancer Res. 2019;38(1):398.
[17] JIANG X, XING L, CHEN Y, et al. CircMEG3 inhibits telomerase activity by reducing Cbf5 in human liver cancer stem cells. Mol Ther Nucleic Acids. 2021;23:310-323.
[18] EPSTEIN SHOCHET G, BROOK E, BARDENSTEIN-WALD B, et al. TGF-β pathway activation by idiopathic pulmonary fibrosis (IPF) fibroblast derived soluble factors is mediated by IL-6 trans-signaling. Respir Res. 2020;21(1):56.
[19] WANG G, JIAO H, ZHENG JN, et al. HSP27 regulates TGF-β mediated lung fibroblast differentiation through the Smad3 and ERK pathways. Int J Mol Med. 2017;39(1):183-190.
[20] SUN T, HUANG Z, ZHANG H, et al. TAZ is required for lung alveolar epithelial cell differentiation after injury. JCI Insight. 2019;5(14): e128674.
[21] CHENG F, SHEN Y, MOHANASUNDARAM P, et al. Vimentin coordinates fibroblast proliferation and keratinocyte differentiation in wound healing via TGF-β-Slug signaling. Proc Natl Acad Sci U S A. 2016;113(30): E4320-E4327.
[22] BONIFAZI M, DI VINCENZO M, CAFFARINI M, et al. How the pathological microenvironment affects the behavior of mesenchymal stem cells in the idiopathic pulmonary fibrosis. Int J Mol Sci. 2020;21(21):8140.
[23] SCHAFER MJ, WHITE TA, IIJIMA K, et al. Cellular senescence mediates fibrotic pulmonary disease. Nat Commun. 2017;8:14532.
[24] NEWTON DA, LOTTES RG, RYAN RM, et al. Dysfunctional lactate metabolism in human alveolar type II cells from idiopathic pulmonary fibrosis lung explant tissue. Respir Res. 2021;22(1):278.
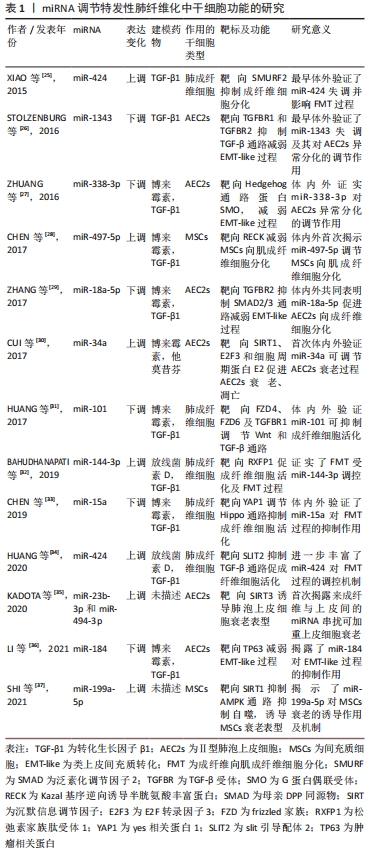
[25] XIAO X, HUANG C, ZHAO C, et al. Regulation of myofibroblast differentiation by miR-424 during epithelial-to-mesenchymal transition. Arch Biochem Biophys. 2015;566:49-57.
[26] STOLZENBURG LR, WACHTEL S, DANG H, HARRIS A. miR-1343 attenuates pathways of fibrosis by targeting the TGF-β receptors. Biochem J. 2016;473(3):245-256.
[27] ZHUANG Y, DAI J, WANG Y, et al. MiR-338* targeting smoothened to inhibit pulmonary fibrosis by epithelial-mesenchymal transition. Am J Transl Res. 2016;8(7):3206-3213.
[28] CHEN X, SHI C, WANG C, et al. The role of miR-497-5p in myofibroblast differentiation of LR-MSCs and pulmonary fibrogenesis. Sci Rep. 2017;7: 40958.
[29] ZHANG Q, YE H, XIANG F, et al. miR-18a-5p Inhibits Sub-pleural Pulmonary Fibrosis by Targeting TGF-β Receptor II. Mol Ther. 2017; 25(3):728-738.
[30] CUI H, GE J, XIE N, et al. miR-34a promotes fibrosis in aged lungs by inducing alveolarepithelial dysfunctions. Am J Physiol Lung Cell Mol Physiol. 2017;312(3):L415-L424.
[31] HUANG C, XIAO X, YANG Y, et al. MicroRNA-101 attenuates pulmonary fibrosis by inhibiting fibroblast proliferation and activation. J Biol Chem, 2017;292(40):16420-16439.
[32] BAHUDHANAPATI H, TAN J, DUTTA JA, et al. MicroRNA-144-3p targets relaxin/insulin-like family peptide receptor 1 (RXFP1) expression in lung fibroblasts from patients with idiopathic pulmonary fibrosis. J Biol Chem. 2019;294(13):5008-5022.
[33] CHEN Y, ZHAO X, SUN J, et al. YAP1/Twist promotes fibroblast activation and lung fibrosis that conferred by miR-15a loss in IPF. Cell Death Differ. 2019;26(9):1832-1844.
[34] HUANG Y, XIE Y, ABEL PW, et al. TGF-β1-induced miR-424 promotes pulmonary myofibroblast differentiation by targeting Slit2 protein expression. Biochem Pharmacol. 2020;180:114172.
[35] KADOTA T, YOSHIOKA Y, FUJITA Y, et al. Extracellular vesicles from fibroblasts induce epithelial-cell senescence in pulmonary fibrosis. Am J Respir Cell Mol Biol. 2020;63(5):623-636.
[36] LI J, PAN C, TANG C, et al. miR-184 targets TP63 to block idiopathic pulmonary fibrosis by inhibiting proliferation and epithelial-mesenchymal transition of airway epithelial cells. Lab Invest. 2021; 101(2):142-154.
[37] SHI L, HAN Q, HONG Y, et al. Inhibition of miR-199a-5p rejuvenates aged mesenchymal stem cells derived from patients with idiopathic pulmonary fibrosis and improves their therapeutic efficacy in experimental pulmonary fibrosis. Stem Cell Res Ther. 2021;12(1):147.
[38] DU Y, HAO X, LIU X. Low expression of long noncoding RNA CDKN2B-AS1 in patients with idiopathic pulmonary fibrosis predicts lung cancer by regulating the p53-signaling pathway. Oncol Lett. 2018;15(4):4912-4918.
[39] GOKEY JJ, SNOWBALL J, SRIDHARAN A, et al. MEG3 is increased in idiopathic pulmonary fibrosis and regulates epithelial cell differentiation. JCI Insight. 2018;3(17):e122490.
[40] LI X, YU T, SHAN H, et al. lncRNA PFAL promotes lung fibrosis through CTGF by competitively binding miR-18a. FASEB J. 2018;32(10):5285-5297.
[41] JIANG H, CHEN Y, YU T, et al. Inhibition of lncRNA PFRL prevents pulmonary fibrosis by disrupting the miR-26a/smad2 loop. Am J Physiol Lung Cell Mol Physiol. 2018;315(4):L563-L575.
[42] QIAN W, CAI X, QIAN Q, et al. lncRNA ZEB1-AS1 promotes pulmonary fibrosis through ZEB1-mediated epithelial-mesenchymal transition by competitively binding miR-141-3p. Cell Death Dis. 2019;10(2):129.
[43] QIAN W, CAI X, QIAN Q. Sirt1 antisense long non-coding RNA attenuates pulmonary fibrosis through sirt1-mediated epithelial-mesenchymal transition. Aging (Albany NY). 2020;12(5):4322-4336.
[44] HUANG C, LIANG Y, ZENG X, et al. Long noncoding RNA FENDRR exhibits antifibrotic activity in pulmonary fibrosis. Am J Respir Cell Mol Biol. 2020;62(4):440-453.
[45] ZHANG Y, YAO XH, WU Y, et al. LncRNA NEAT1 regulates pulmonary fibrosis through miR-9-5p and TGF-β signaling pathway. Eur Rev Med Pharmacol Sci. 2020;24(16):8483-8492.
[46] LIU P, ZHAO L, GU Y, et al. LncRNA SNHG16 promotes pulmonary fibrosis by targeting miR-455-3p to regulate the Notch2 pathway. Respir Res. 2021;22(1):44.
[47] YI H, LUO D, XIAO Y, et al. Knockdown of long non‑coding RNA DLEU2 suppresses idiopathic pulmonary fibrosis by regulating the microRNA‑369‑3p/TRIM2 axis. Int J Mol Med. 2021;47(5):80.
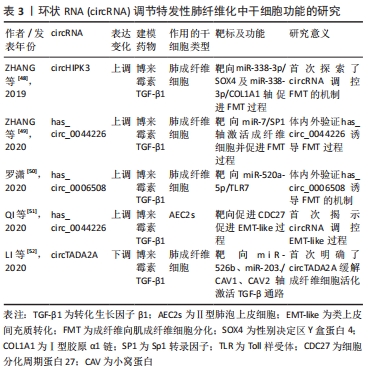
[48] ZHANG JX, LU J, XIE H, et al. circHIPK3 regulates lung fibroblast-to-myofibroblast transition by functioning as a competing endogenous RNA. Cell Death Dis. 2019;10(3):182.
[49] ZHANG L, CHI X, LUO W, et al. Lung myofibroblast transition and fibrosis is regulated by circ0044226. Int J Biochem Cell Biol. 2020;118:105660.
[50] 罗潇.环状RNA的差异性表达及hsa_circ_0006508在特发性肺纤维化中的作用机制研究[D].太原:山西医科大学,2020.
[51] QI F, LI Y, YANG X, et al. Hsa_circ_0044226 knockdown attenuates progression of pulmonary fibrosis by inhibiting CDC27. Aging (Albany NY). 2020;12(14):14808-14818.
[52] LI J, LI P, ZHANG G, et al. CircRNA TADA2A relieves idiopathic pulmonary fibrosis by inhibiting proliferation and activation of fibroblasts. Cell Death Dis. 2020;11(7):553.
[53] BAI J, DENG J, HAN Z, et al. CircRNA_0026344 via exosomal miR-21 regulation of Smad7 is involved in aberrant cross-talk of epithelium-fibroblasts during cigarette smoke-induced pulmonary fibrosis. Toxicol Lett. 2021;347:58-66.
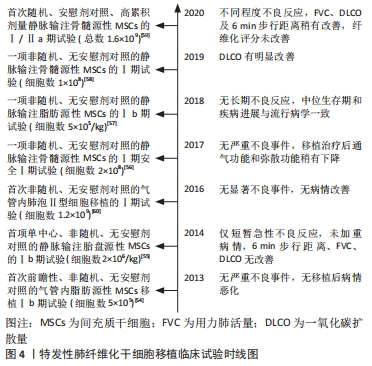
[54] TZOUVELEKIS A, PASPALIARIS V, KOLIAKOS G, et al. A prospective, non-randomized, no placebo-controlled, phase Ib clinical trial to study the safety of the adipose derived stromal cells-stromal vascular fraction in idiopathic pulmonary fibrosis. J Transl Med. 2013;11:171.
[55] CHAMBERS DC, ENEVER D, ILIC N, et al. A phase 1b study of placenta-derived mesenchymal stromal cells in patients with idiopathic pulmonary fibrosis. Respirology. 2014;19(7):1013-1018.
[56] GLASSBERG MK, MINKIEWICZ J, TOONKEL RL, et al. Allogeneic human mesenchymal stem cells in patients with idiopathic pulmonary fibrosis via intravenous delivery (AETHER): a phase i safety clinical trial. Chest. 2017;151(5):971-981.
[57] NTOLIOS P, MANOLOUDI E, TZOUVELEKIS A, et al. Longitudinal outcomes of patients enrolled in a phase Ib clinical trial of the adipose-derived stromal cells-stromal vascular fraction in idiopathic pulmonary fibrosis. Clin Respir J. 2018;12(6):2084-2089.
[58] FISHMAN JE, KIM GJ, KYEONG NY, et al. Intravenous stem cell dose and changes in quantitative lung fibrosis and DLCO in the AETHER trial: a pilot study. Eur Rev Med Pharmacol Sci. 2019;23(17):7568-7572.
[59] AVERYANOV A, KOROLEVA I, KONOPLYANNIKOV M, et al. First-in-human high-cumulative-dose stem cell therapy in idiopathic pulmonary fibrosis with rapid lung function decline. Stem Cells Transl Med. 2020;9(1):6-16.
[60] SERRANO-MOLLAR A, GAY-JORDI G, GUILLAMAT-PRATS R, et al. Safety and tolerability of alveolar type II cell transplantation in idiopathic pulmonary fibrosis. Chest. 2016;150(3):533-543.
[61] LOPEZ-RODRIGUEZ E, GAY-JORDI G, KNUDSEN L, et al. Improved alveolar dynamics and structure after alveolar epithelial type II cell transplantation in bleomycin induced lung fibrosis. Front Med (Lausanne). 2021;8:640020.
[62] LENNOX KA, BEHLKE MA. Cellular localization of long non-coding RNAs affects silencing by RNAi more than by antisense oligonucleotides. Nucleic Acids Res. 2016;44(2):863-877.
[63] JANAS MM, SCHLEGEL MK, HARBISON CE, et al. Selection of GalNAc-conjugated siRNAs with limited off-target-driven rat hepatotoxicity. Nat Commun. 2018;9(1):723.
[64] WAN C, ALLEN TM, CULLIS PR. Lipid nanoparticle delivery systems for siRNA-based therapeutics. Drug Deliv Transl Res. 2014;4(1):74-83.
[65] CROOKE ST, VICKERS TA, LIANG XH. Phosphorothioate modified oligonucleotide-protein interactions. Nucleic Acids Res. 2020;48(10): 5235-5253.
[66] LIU YF, HUANG S, NG TK, et al. Longitudinal evaluation of immediate inflammatory responses after intravitreal AAV2 injection in rats by optical coherence tomography. Exp Eye Res. 2020;193:107955.
[67] ROTH TL, PUIG-SAUS C, YU R, et al. Reprogramming human T cell function and specificity with non-viral genome targeting. Nature. 2018; 559(7714):405-409.
[68] RAN FA, HSU PD, WRIGHT J, et al. Genome engineering using the CRISPR-Cas9 system. Nat Protoc. 2013;8(11):2281-2308.
[69] TRAN NT, SOMMERMANN T, GRAF R, et al. Efficient CRISPR/Cas9-Mediated Gene Knockin in Mouse Hematopoietic Stem and Progenitor Cells. Cell Rep. 2019;28(13):3510-3522.e5.
[70] GUIOT J, CAMBIER M, BOECKX A, et al. Macrophage-derived exosomes attenuate fibrosis in airway epithelial cells through delivery of antifibrotic miR-142-3p. Thorax. 2020;75(10):870-881.
[71] SUN X, KANG Y, XUE S, et al. In vivo therapeutic success of MicroRNA-155 antagomir in a mouse model of pulmonary fibrosis induced by bleomycin. Korean J Intern Med. 2021;36(Suppl 1): S160-S169.
[72] XU F, XU F, XIE S, et al. MicroRNA-448 overexpression inhibits fibroblast proliferation and collagen synthesis and promotes cell apoptosis via targeting ABCC3 through the JNK signaling pathway. J Cell Physiol. 2020;235(2):1374-1385.
[73] BEG MS, BRENNER AJ, SACHDEV J, et al. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Invest New Drugs. 2017;35(2):180-188.
[74] VAN DER REE MH, DE VREE JM, STELMA F, et al. Safety, tolerability, and antiviral effect of RG-101 in patients with chronic hepatitis C: a phase 1B, double-blind, randomised controlled trial. Lancet. 2017; 389(10070):709-717.
[75] ZHOU Y, CHAI X. Protective effect of bicyclol against pulmonary fibrosis via regulation of microRNA-455-3p in rats. J Cell Biochem. 2020;121(1): 651-660.
[76] TONG J, WU Z, WANG Y, et al. Astragaloside IV Synergizing with Ferulic Acid Ameliorates Pulmonary Fibrosis by TGF-β1/Smad3 Signaling. Evid Based Complement Alternat Med. 2021. doi: 10.1155/2021/8845798.
[77] KARNER H, WEBB CH, CARMONA S, et al. Functional Conservation of LncRNA JPX Despite Sequence and Structural Divergence. J Mol Biol. 2020;432(2):283-300.
[78] YANG Y, TAI W, LU N, et al. lncRNA ZFAS1 promotes lung fibroblast-to-myofibroblast transition and ferroptosis via functioning as a ceRNA through miR-150-5p/SLC38A1 axis. Aging (Albany NY). 2020;12(10): 9085-9102.
[79] QIAN W, CAI X, QIAN Q, et al. Angelica sinensis polysaccharide suppresses epithelial-mesenchymal transition and pulmonary fibrosis via a DANCR/AUF-1/FOXO3 regulatory axis. Aging Dis. 2020,11(1):17-30.
[80] CHEN H, WANG J, LI R, et al. Astaxanthin attenuates pulmonary fibrosis through lncITPF and mitochondria-mediated signal pathways. J Cell Mol Med. 2020;24(17):10245-10250.
[81] SCHMIDT CA, GIUSTO JD, BAO A, et al. Molecular determinants of metazoan tricRNA biogenesis. Nucleic Acids Res. 2019;47(12):6452-6465.
[82] ZENG H, GAO H, ZHANG M, et al. atractylon treatment attenuates pulmonary fibrosis via regulation of the mmu_circ_0000981/miR-211-5p/TGFBR2 axis in an ovalbumin-induced asthma mouse model. Inflammation. 2021. doi: 10.1007/s10753-021-01463-6.
|