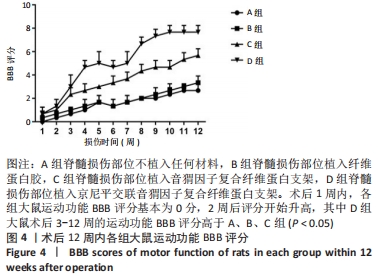
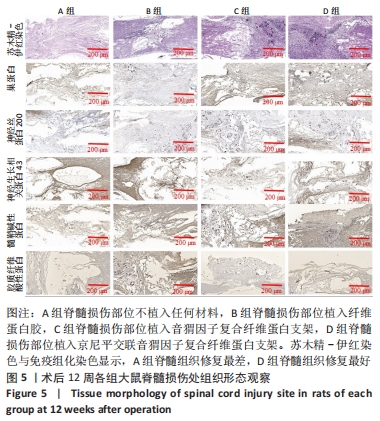
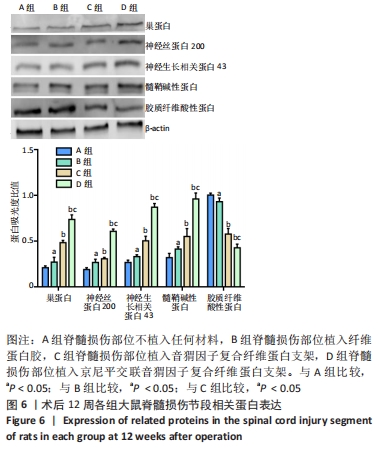
[1] HUBLI M, KRAMER JLK, JUTZELER CR, et al. Application of electrophysiological measures in spinal cord injury clinical trials: a narrative review. Spinal Cord. 2019;57(11):909-923.
[2] RABINSTEIN AA. Traumatic Spinal Cord Injury. Continuum(Minneap Minn). 2018;24(2):551-566.
[3] MULCAHEY MJ, JONES L AT, ROCKHOLD F, et al. Adaptive trial designs for spinal cord injury clinical trials directed to the central nervous system. Spinal Cord. 2020;58(12):1235-1248.
[4] ASSINCK P, DUNCAN GJ, HILTON BJ, et al. Cell transplantation therapy for spinal cord injury. Nat Neurosci. 2017;20(5):637-647.
[5] KUBINOVÁ S, SYKOVÁ E. Biomaterials combined with cell therapy for treatment of spinal cord injury. Regen Med. 2012;7(2):207-224.
[6] ANITUA E, NURDEN P, PRADO R, et al. Autologous fibrin scaffolds: When platelet-and plasma-derived biomolecules meet fibrin. Biomaterials. 2019;192: 440-460.
[7] WEI Y, ZHU G, ZHAO Z, et al. Individualized plasticity autograft mimic with efficient bioactivity inducing osteogenesis. Int J Oral Sci. 2021; 13(1):1-8.
[8] JANMEY PA, WINER JP, WEISEL JW. Fibrin gels and their clinical and bioengineering application[. J R Soc Interface. 2009;6(30):1-10.
[9] MANICKAM B, SREEDHARAN R, ELUMALAI M. ‘Genipin’ – The Natural Water Soluble Cross-linking Agent and Its Importance in the Modified Drug Delivery Systems: An Overview. CCurr Drug Deliv. 2014;11(1): 139-145.
[10] NAM KN, CHOI YS, JUNG HJ, et al. Genipin inhibits the inflammatory response of rat brain microglial cells. Int Immunopharmacol. 2010; 10(4):493-499.
[11] ZHANG Z, HE Q, DENG W, et al. Nasal ectomesenchymal stem cells: Multi-lineage differentiation and transformation effects on fibrin gels - ScienceDirect. Biomaterials. 2015;49:57-67.
[12] CHEN P, SUN Y, ZHOU H, et al. The Effects of Acellular Spinal Cord Scaffolds-Genipin-Sonic Hedgehog on Proliferation and Differentiation of Ectomesenchymal Stem Cells. J Biomater Tiss Eng. 2018;(9):187-192.
[13] VAILLANT C, MONARD D. SHH Pathway and Cerebellar Development. Cerebellum. 2009;8(3):291-301.
[14] CAI C, THORNE J, GRABEL L. Hedgehog Serves as a Mitogen and Survival Factor During Embryonic Stem Cell Neurogenesis. Stem Cells. 2010;26(5):1097-1108.
[15] SCHEFF SW, SAUCIER DA, CAIN ME. A statistical method for analyzing rating scale data: the BBB locomotor score. J Neurotaum. 2002;19(10): 1251-1260.
[16] COLLINGER JL, FOLDES S, BRUNS TM, et al. Neuroprosthetic technology for individuals with spinal cord injury. J Spinal Cord Med. 2013;36(4): 258-272.
[17] SONG YH, AGRAWAL NK, GRIFFIN JM, et al. Recent advances in nanotherapeutic strategies for spinal cord injury repair. Adv Drug Deliver Rev. 2019;148: 38-59.
[18] ZHAO Y, WANG Y, GONG J, et al. Chitosan degradation products facilitate peripheral nerve regeneration by improving macrophage-constructed microenvironments. Biomaterials. 2017;134:64-77.
[19] FAWCETT JW. The extracellular matrix in plasticity and regeneration after CNS injury and neurodegenerative disease. Prog Brain Res. 2015; 218:213.
[20] KHABOUSHAN AS, SHAKIBAEI M, KAJBAFZADEH AM, et al. Prenatal Neural Tube Anomalies: A Decade of Intrauterine Stem Cell Transplantation Using Advanced Tissue Engineering Methods. Stem Cell Rev Rep. 2021:1-16. doi: 10.1007/s12015-021-10150-w.
[21] STRALEY KS, FOO CWP, HEILSHORN SC. Biomaterial Design Strategies for the Treatment of Spinal Cord Injuries. J Neurotraum. 2009;27(1): 1-19.
[22] JI WC, ZHANG XW, QIU YS. Selected suitable seed cell, scaffold and growth factor could maximize the repair effect using tissue engineering method in spinal cord injury. World J Exp Med. 2016;6(3):58-62.
[23] HU XC, LU YB, YANG YN, et al. Progress in clinical trials of cell transplantation for the treatment of spinal cord injury: how many questions remain unanswered? Neural Regen Res. 2021;16(3):405-413.
[24] WU GH, SHI HJ, CHE MT, et al. Recovery of paralyzed limb motor function in canine with complete spinal cord injury following implantation of MSC-derived neural network tissue. Biomaterials. 2018;181:15-34.
[25] CANONICO S. The use of human fibrin glue in the surgical operations. Acta Biomed. 2003; 74 Suppl 2:21-25.
[26] SCHULZ PM, GEHRINGER W, NÖHRING S, et al. Biochemical characterization, stability, and pathogen safety of a new fibrinogen concentrate (fibryga®). Biologicals. 2018;52:72-77.
[27] KATTULA S, BYRNES JR, WOLBERG AS. Fibrinogen and Fibrin in Hemostasis and Thrombosis. Arterioscl Throm Vas. 2017;37(3):e13.
[28] DE MELO B G, JODAT YA, CRUZ EM, et al. Strategies to use fibrinogen as bioink for 3D bioprinting fibrin-based soft and hard tissues. Acta Biomater. 2020;3:117.
[29] SQUIRE JM, PARRY DA. Fibrous Protein Structures: Hierarchy, History and Heroes. Subcell Biochem. 2017;82:1-33.
[30] LAIDMÄE I, ĒRGLIS K, CĒBERS A, et al. Salmon fibrinogen and chitosan scaffold for tissue engineering: in vitro and in vivo evaluation. J Mater Sci Mater Med. 2018;29(12):182.
[31] DE MELO BAG, JODAT YA, CRUZ EM, et al. Strategies to use fibrinogen as bioink for 3D bioprinting fibrin-based soft and hard tissues. Acta Biomater. 2020;117:60-76.
[32] KANG Z, WANG Y, XU J, et al. An RGD-containing peptide derived from wild silkworm silk fibroin promotes cell adhesion and spreading. Polymers. 2018;10(11):1193.
[33] BENCHERIF SA, GSIB O, EGLES C. Fibrin: An underrated biopolymer for skin tissue engineering. J Mol Biol Biotech. 2017;2(1):123-126.
[34] YOO JS, KIM YJ, KIM SH, et al. Study on Genipin: A New Alternative Natural Crosslinking Agent for Fixing Heterograft Tissue. Korean J Thorac Cardiovasc Surg. 2011;44:254-257.
[35] BIGI A, COJAZZI G, PANZAVOLTA S, et al. Stabilization of gelatin films by crosslinking with genipin. Biomaterials. 2002;23(24):4827-4832.
|